PDX Model Market Size – Industry Structure Evaluation, Demand Drivers Analysis, Regional Growth Analysis and Identification, Competitive Positioning Review & Global Market Size Forecast to 2029
Overview
PDX Model Market size was valued at USD 177.32 Mn. in 2022 and the total PDX Model revenue is expected to grow at 13.8% from 2023 to 2029, reaching nearly USD 438.29 Mn.
PDX Model Market Overview:
Patient-Derived Xenograft/PDX model creation has been more popular in recent years, as expectation increase that such systems will be more relevant than a mouse and cell line models, and hence more informative and predictive preclinical therapeutic models. Recent studies have demonstrated that personalized PDX avatars may predict and even lead to cancer patients' tailored treatment. However, the capacity of PDX models to function as true mouse avatars is dependent on the conservation of each case's distinct molecular properties.
Many tumor forms have high intra-tumor complexity and geographic dispersion of different clones and metastases, making customized avatar modeling difficult. However, there is evidence that PDX models are stable across several passes once formed, providing for a consistent, trustworthy system. Despite this limitation, PDX models remain useful models for a specific disease subtype if global gene expression patterns are preserved.
Factors such as rising demand for personalized medication increased R&D efforts in the pharmaceutical industry, and ongoing governmental and private sector support for cancer research are driving the patient-derived xenograft models market forward. Before clinical development and testing in people, the mouse model of cancer has regularly been used to assess the in vivo efficacy of novel anti-cancer treatments. According to GLOBOCAN statistics issued in 2020, 19.3 million cancer cases and 10 million cancer deaths were recorded in 2020. As a result, both the public and business sectors are increasing their support for cancer research.
As a result, the increased emphasis on the discovery of new treatment options, combined with the rising cancer burden, is expected to increase the patient-derived xenograft models market. However, the high cost of tailored PDX models is expected to restrain market growth throughout the forecast period.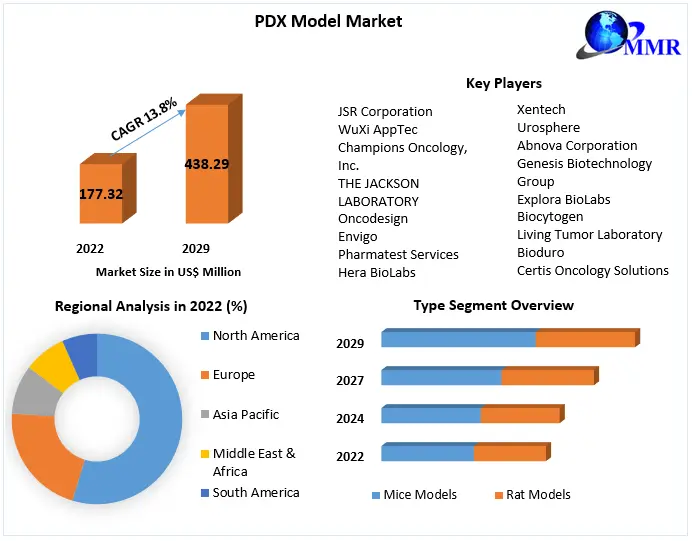 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
PDX Model Market Dynamics:
Rising demand for PDX models for pharmacology and biomarker studies
Traditionally, tumor xenograft and, to a lesser extent, syngeneic models were used for pharmacology, biomarker, and pharmacokinetics/pharmacodynamics research for oncology operations. With the huge growth in the availability and cost of PDX models supplied by both academic institutions and contract research businesses, PDX models' value in normal research operations has increased. A cursory review of cancer discovery programs published in the last three years reveals that PDX models are being used at various levels of preclinical discovery and translational research.
However, there is a growing trend in the PDX Model industry to incorporate PDX model readout as a critical component of the needed data package for both internal usage and regulatory submission. It is possible to trace the use of PDX models in drug development back several decades. One of the early papers using cancer medications and PDX models, for example, investigated numerous chemotherapeutic agents at their respective MTDs in PDX models obtained from 34 patients and found 92% accuracy in predicting effectiveness and 97% accuracy in predicting no-response.
Additional research suggests that the predictive value can vary depending on tumor histology and location, stage of illness from which the models are created, PDX model quality, sample size, and dosing regimen. In addition to selecting models that are histologically, molecularly, and genetically relevant to clinical patients, medication exposure is a crucial element in increasing the translatability of preclinical data.
The growing prevalence of Chronic Diseases rising the demand for Personalized Medicines
Personalized medicine is the process of creating customized medications to provide individualized treatment and care based on personal and genetic variations. Animal models are used to create these medications. Immunodeficient models are transplanted with human tissues in this technique, and the disease expression is monitored. The model is then treated with drugs/gene treatments to determine the best treatment, resulting in appropriate or customized medications.
The discovery of medicines for chronic diseases like cancer has received a significant amount of attention. Nowadays, cancer is a major burden on healthcare systems across various geographic regions. According to Worldwide cancer data issued in 2020, around 18 million cancer cases and 10 million cancer deaths were recorded in 2020. To address the increased occurrence of cancer and provide effective therapies to patients, in vitro, and in vivo techniques are being developed. Cancer is one of the leading causes of death globally. The disease has developed rapidly and is expected to increase substantially throughout the forecast period.
Every year, around 400 000 children are diagnosed with cancer. The most common cancers vary by nation. As a result, the rising global prevalence of cancer drives the market by increasing the rate of diagnosis and the demand for personalized medication. The discovery of PDX models, which have strong predictive value for the effectiveness of established and innovative anti-cancer therapies, is a noteworthy breakthrough in this area. Because of this benefit, these models are increasingly being utilized in preclinical research.
| Rank | Cancer | Cases in 2020 | % of all cancers |
| All cancers | 18,094,716 | ||
| 1 | Breast | 2,261,419 | 12.5 |
| 2 | Lung | 2,206,771 | 12.2 |
| 3 | Colorectal | 1,931,590 | 10.7 |
| 4 | Prostate | 1,414,259 | 7.8 |
| 5 | Stomach | 1,089,103 | 6.0 |
| 6 | Liver | 905,677 | 5.0 |
| 7 | Cervix uteri | 604,127 | 3.3 |
| 8 | Oesophagus | 604,100 | 3.3 |
| 9 | Thyroid | 586,202 | 3.2 |
| 10 | Bladder | 573,278 | 3.2 |
Unaffordable personalized PDX models a retrains for PDX Model Market growth
A specimen of the patient's tumor is implanted and grown in many immunocompromised models to develop individualized PDX models. It is then extracted, fragmented, and re-implanted in a wider group of models that have been treated with various medications and pharmacological combinations. Given the costs of developing and maintaining these models, as well as the use of pricey medications in these models, PDX trials can cost thousands of dollars, even for a single patient. However, utilizing PDX models in government-funded fundamental research might be too expensive. These are the key factors restricting the global adoption of PDX models.
Growing adoption of Humanized PDX models
Even though xenografts develop well in immunodeficient models, these models are largely used for PDX model creation. These models, however, are not appropriate for examining stroma-tumor interactions, which are primarily responsible for tumor development and progression in a patient. Humanized PDX models assist with overcoming these challenges and are gaining industry popularity.
Immunodeficient mice (such as NOD SCID mice) are now being reconstructed with bone marrow or peripheral blood cells, along with patient tumor implants, to generate humanized xenograft models. These models provide an excellent platform for investigating the role of xenogeneic immune cells in overall anti-tumor immunity. They also aid in the research of immunotherapy medications aimed at activating the immune system's effector arm. However, there is a danger of graft versus host illness with humanized xenografts. This constraint can be overcome by confining the experimental setup and analysis to a 6-10 week time range.
PDX Model Market Segment Analysis:
By Type, the Mice Model segment dominated the PDX Model Market with the largest market share in 2022 and is expected to maintain its dominance at the end of the forecast period. The dominance is mostly due to factors like simplicity of acquisition, ease of manipulation, and the lower facility needed for maintaining mouse models under observation compared to rats, which is increasing their adoption in the creation of PDX models.
Patient-derived xenografts (PDX) in immunodeficient mice are useful in vivo models for investigating the pathophysiology of various types of malignant tumors and for antineoplastic drug discovery. Precision medicine has benefited from the use of PDX models. Because PDX resembles the basic properties of human tumors, it provides a good platform for precisely predicting treatment responses.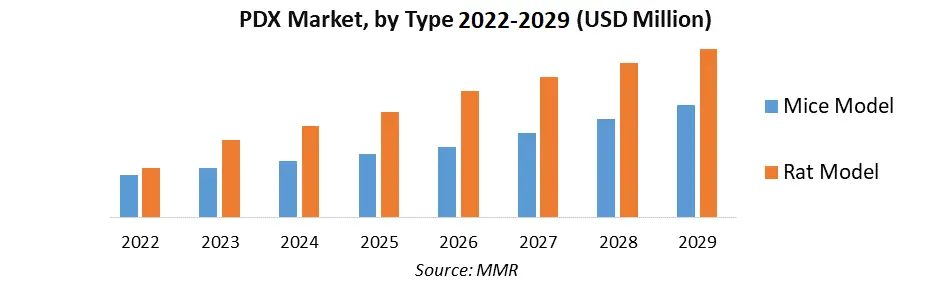
The emergence of immunodeficient mice has greatly aided the use of PDX models in medical research. For in vivo investigations, tumor researchers have mostly relied on xenograft models established by engrafting cultivated cell lines obtained from tumor tissues into immunodeficient mice. Because such models, like in vitro models, retain tumor cells' ability to proliferate indefinitely, they have been used to predict the clinical relevance of studies on proliferating cells or agents that exert anti-tumor effects by damaging and killing tumor cells, such as DNA-damaging agents and agents that target driver mutations.
However, because these models are formed of a limited population of tumor cells, only those capable of adapting to culture conditions, they do not reflect tumor variety and heterogeneity, which are major segment restrictions.
By Tumor Type, the Respiratory Tumor Model held the largest market share in 2022 and is expected to grow at a CAGR of 12.8% during the forecast period. The growing research investments in the field of oncology, as well as the increasing focus of market participants on producing lung cancer PDX models, are significant factors contributing to the segment's growth. Cancer is the world's second biggest cause of mortality. To preserve their competitive edge and enter new regional markets, various organizations are focusing on creating breakthrough therapies for the treatment of chronic diseases. Likewise, a rise in financing for cancer research businesses is expected to enhance the research industry.
Lung cancer is the leading cause of death globally. The most prevalent cancer among NSCLC (non-small cell lung cancer) is LUSC (lung squamous cell carcinoma), which is difficult to treat in many cases since it is commonly identified at an advanced stage. LUSC is significantly linked to smoking and is frequently coupled with other comorbidities. Current targeted treatments are restricted in their ability to treat human lung squamous cell carcinoma (LUSC) due to its numerous somatic mutations and lack of a clear dominant driving mutation. These mutational variations, which exclude the use of conventional targeted treatments or the combination of known therapeutic modalities, would need the development of a preclinical animal model of this malignancy in order to get superior clinical results.
PDX models produced from patients have been identified as potentially helpful preclinical models for customized precision medicine. However, whether the use of LUSC PDX models is acceptable for therapeutic use remains debatable.
| The age-adjusted incidence of lung cancer by histological type | |
| Histological type | Incidence per 100,000 per year |
| All types | 66.9 |
| Adenocarcinoma | 22.1 |
| Squamous cell carcinoma | 14.4 |
| Small-cell carcinoma | 9.8 |
PDX Model Market Regional Insights:
North American market dominated the patient-derived xenograft/ PDX models market in 2022 and is expected to grow at a CAGR of 13.2% during the forecast period. The dominance is attributed to a rise in funding initiatives and awareness among patients regarding personalized medicine, followed by Europe. Patient-derived tumor xenografts are recognized as very relevant preclinical cancer models.
As per American Cancer Society estimates, in 2018, there were 1,735,350 new cases of cancer diagnosed in the United States. Also, in 2018, the National Institute of Cancer expands the repository of cancer research models to include more research models. Hence, the North American region is expected to grow at a faster rate due to rising preclinical activities by pharmaceutical companies, and growing cancer research in this region.
When compared to cell line-derived xenograft models, PDX models are expected to have excellent predictability of treatment effectiveness while keeping tumor inhomogeneity. Large-scale PDX libraries are now being built in Europe and the United States. The US National Cancer Institute revealed intentions in 2016 to transition its anti-cancer drug screening system from the "NCI-60 Human Tumor Cell Lines Screen" to PDX-based models, which increases model adoption and drives regional growth.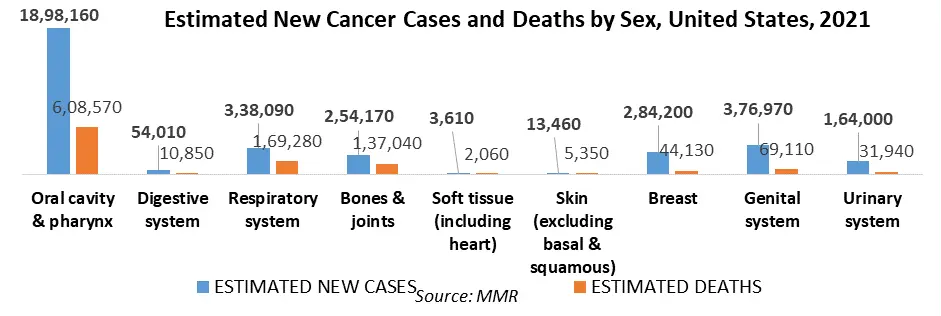
In Europe and the United States, large-scale PDX libraries are being built and used for drug development and biomarker screening. "EurOPDX" was organized in 2013 by 16 European universities and governmental entities. More than 1500 PDX types, including uncommon tumors, have been identified to date. Additionally, the Jackson Institute (USA) has begun to make available on its website the genetic and histological information of over 450 different types of PDXs. Simultaneously, the institution has started the industrial use of PDXs by commercializing several PDX mice models.
Colorectal cancer (CRC) is the third most common cancer, affecting 10% of men and 9% of women globally. CRC is the fourth leading cause of cancer death globally, and the second leading cause in Europe. The disease's intrinsic complexity, defined by many genetic abnormalities in interrelated signaling cascades, influences both therapeutic outcomes and medication development.
CRC is treated with surgery, targeted chemotherapy, and radiation. However, therapeutic success is heavily reliant on selecting the suitable therapeutic modality for the particular patient, who will gain the most from an acceptable and successful therapy. Treating decades, the only medications available for CRC and metastatic CRC patients were 5-fluorouracil (5-FU) regulated by leucovorin (LV). As of now, 5-FU is used as first-line therapy in conjunction with oxaliplatin or irinotecan. The emergence of specific anti-cancer medications such as bevacizumab and cetuximab increased CRC patients' survival.
However, identifying resistance profiles and stratifying responding individuals remains a significant clinical problem in CRC care. This necessitates the development of adequate and dependable preclinical models that replicate cancer pathway dynamics and closely match the clinical scenario. In this setting, therapeutically relevant in vivo models, such as patient-derived xenograft models, are necessary (PDX). PDX has been shown to retain the morphological and molecular properties of the original heterogeneous patient tumor, making it a superior model system for translational research.
Global PDX Model Market Scope: Inquire before buying
| PDX Model Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2022 | Forecast Period: | 2023-2029 |
| Historical Data: | 2018 to 2022 | Market Size in 2022: | US $ 177.32 Mn. |
| Forecast Period 2023 to 2029 CAGR: | 13.8% | Market Size in 2029: | US $ 438.29 Mn. |
| Segments Covered: | by Type | Mice Models Rat Models |
|
| by Tumor Type | Gastrointestinal Tumor Models Gynecological Tumor Models Respiratory Tumor Models Urological Tumor Models Hematological Tumor Models Other Tumor Models |
||
| by Application | Preclinical Drug Development Biomarker Analysis Basic Cancer Research |
||
| by End-User | Pharmaceutical & Biotechnology Companies Contract Research Organizations (CROs) Academic & Research Institutions |
||
PDX Model Market, by Region
North America (United States, Canada and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan and Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A)
South America (Brazil, Argentina Rest of South America)
PDX Model Market Key Players
1. JSR Corporation
2. WuXi AppTec
3. Champions Oncology, Inc.
4. THE JACKSON LABORATORY
5. Charles River Laboratories
6. Oncodesign
7. Envigo
8. Pharmatest Services
9. Hera BioLabs
10. EPO Berlin-Buch GmbH
11. Xentech
12. Urosphere
13. Abnova Corporation
14. Genesis Biotechnology Group
15. Explora BioLabs
16. Biocytogen
17. Living Tumor Laboratory
18. Bioduro
19. Aragen Life Sciences w.e.f,
20. Shanghai Lide Biotech Co., Ltd.
21. Certis Oncology Solutions
22. InnoSer
23. IVRS AB
24. BEIJING IDMO Co. Ltd.
25. Shanghai ChemPartner Co., Ltd.
FAQs:
1. What is a patient-derived xenograft (PDX) model?
Patient-derived xenografts (PDX) are cancer models in which tumor tissue or cells are put into an immune-deficient or humanized mouse. PDX models are the most translational research platform for assessing effectiveness because they imitate human tumor biology and allow for natural disease development.
2. What is a humanized mouse model?
Humanized mice are immunocompromised mice into whom human immune systems have been engrafted by peripheral blood mononuclear cells (PBMCs) or hematopoietic stem cells (HSCs). Humanized mice are useful instruments for testing treatment candidates in vivo in a context similar to human physiology.
3. What is expected to drive the growth of the PDX market during the forecast period?
A major driver in the PDX market is the growing prevalence of Chronic Diseases rising the demand for Personalized Medicines.
4. What is the projected market size & growth rate of the PDX Model Market?
Ans. Patient-Derived Xenograft/ PDX Model Market size was valued at USD 177.32 Mn. in 2022 and the total PDX Model revenue is expected to grow at 13.8% from 2023 to 2029, reaching nearly USD 438.29 Mn.
5. What segments are covered in the PDX Model Market report?
Ans. The segments covered are Type, Tumor Type, Application, End User, and Region.
