Opioid Tolerance Treatment Market – Global Market Size, Strategic Growth Drivers, Risk Assessment Framework, Regulatory Landscape Review, Competitive Intensity Mapping & Long-Term Industry Outlook to 2032
Overview
Opioid Tolerance Treatment Market was valued at USD 1.88 Bn in 2025 and is expected to reach USD 2.4 Bn by 2032, growing at a CAGR of 3.51% during the forecast period.
Opioid Tolerance Treatment Market Overview:
Opioids are drugs used to treat severe or persistent pain, such as chronic headaches and backaches, as well as by people recovering from surgery or dealing with severe cancer-related pain, as well as by adults and children who have suffered serious injuries in falls, car accidents, or other incidents or while playing sports. Opioids are classified as drugs as well. Depending on the patient, the circumstance, and the type and amount of pain, doctors may prescribe a variety of opioids in a range of strengths that can be taken in a variety of ways. 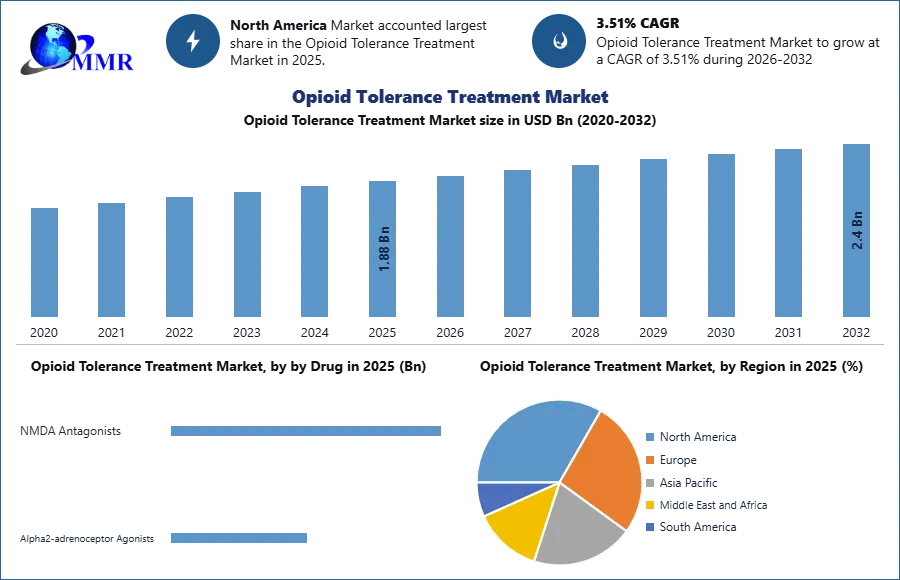 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
The report explores the Opioid Tolerance Treatment Market's segments (Drug, Opioid Tolerance, Route of Administration, and End-User Region). Data has been provided by market participants, and regions (North America, Asia Pacific, Europe, Middle East & Africa, and South America). The MMR market report provides a thorough analysis of the rapid advances that are currently taking place across all industry sectors. Facts and figures, illustrations, and presentations are used to provide key data analysis for the historical period from 2020 to 2024. The report investigates the Opioid Tolerance Treatment Market’s drivers, limitations, prospects, and barriers. This MMR report includes investor recommendations based on a thorough examination of the Opioid Tolerance Treatment Market’s contemporary competitive scenario.
Opioid Tolerance Treatment Market Dynamics:
Opioid therapy is widely used in the treatment of persistent cancer and severe pain. Their excellent efficacy is one factor in their widespread use. When these are used for a longer period of time, there are occasionally unfavorable implications identified. Therapy has less of an impact, and people respond to it less enthusiastically. Such patients need NMDA antagonists or greater doses. Occasionally, alpha2-adrenoceptor agonists are used.
When someone regularly uses opioids, they may have the condition known as opioid tolerance. The body adapts to the presence of opioids over time and no longer reacts to them the same way. This may result in the need for higher doses of opioids to have the same impact as before. For those who depend on opioids for pain treatment or other medical needs, opioid tolerance is a serious issue because it can result in addiction and potentially lethal overdoses.
According to a thorough research the industry for opioid tolerance treatment will extend the opportunities available to market players with moderate market development. The evaluation period for the growth, from 2022 to 2029, would also show an increase in market valuation.
Opioids are powerful painkillers that are administered to patients with chronic cancer and excruciating pain. These opioids are often only taken temporarily. Patients develop an opioid tolerance if these opioids are prescribed or taken more frequently or for a longer period of time, which prevents them from experiencing the desired analgesic or therapeutic response. Higher opioid doses are needed to treat people in such circumstances.
Other opioid therapy alternatives, like NMDA antagonists and alpha2-adrenoceptor agonists, can be used to treat opioid-tolerant patients. These medications give opioid-tolerant patients or those with opioid tolerance the therapeutic response they need.
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 04 February 2026 | Vertex Pharmaceuticals | The FDA granted priority review for Suzetrigine (VX-548), a highly selective NaV1.8 inhibitor designed for acute pain management. | By offering opioid-level efficacy without the central nervous system side effects, this drug reduces the risk of opioid tolerance and dependency development in post-surgical patients. |
| 12 December 2025 | Ensysce Biosciences, Inc. | Ensysce announced the completion of its clinical end-of-Phase 2 meeting with the FDA for PF614-MPAR, a combination product with Multi-Pill Abuse Resistance. | This next-generation technology prevents respiratory depression from overdose, allowing for safer opioid titration in patients who have developed significant physiological tolerance. |
| 15 August 2025 | Ensysce Biosciences, Inc. | The company officially launched the Phase 3 clinical trial for PF614, a Trypsin-Activated Abuse Protected (TAAP) oxycodone prodrug. | The $5.3 million NIDA-funded study focuses on providing extended-release analgesia that is resistant to manipulation, addressing a key gap in long-term pain management for tolerant users. |
| 13 June 2025 | Innocan Pharma Corporation | Innocan Pharma published a breakthrough review in Cureus highlighting the potential of Liposomal Synthetic CBD (LPT-CBD) as a non-opioid analgesic. | The proprietary LPT platform provides steady plasma levels for up to four weeks, offering a promising alternative for patients failing opioid therapy due to refractory tolerance. |
| 31 January 2025 | Alvogen, Inc. | Alvogen issued a voluntary nationwide recall of one lot of Fentanyl Transdermal System patches due to multi-stacked delivery defects. | This recall underscores regulatory safety concerns regarding high-potency delivery systems, driving market demand for precision-dose abuse-deterrent formulations. |
| 21 January 2025 | Johnson & Johnson | The FDA expanded the label for Spravato (esketamine), which is increasingly utilized in clinics to manage opioid-induced hyperalgesia and chronic pain resistance. | This NMDA receptor antagonist provides a critical mechanism for resetting opioid sensitivity, directly improving outcomes for patients with advanced opioid tolerance. |
Drivers:
Due to increased opioid use for both medical and non-medical purposes as well as an increase in the number of opioid prescriptions written for therapeutic purposes, which can result in opioid tolerance if taken for an extended period of time, the market for opioid tolerance treatment is growing rapidly.
Additionally, ongoing clinical trials, numerous studies being conducted for opioid tolerant treatment by various research institutes & firms, rising drug misuse, and drug overdose occurrences are some of the primary factors causing the opioid tolerance treatment market to grow.
Drug misuse is becoming more prevalent in all nations and societies. The countries that are close to the Golden Crescent and Golden Triangle, areas in the Asia Pacific region known for illicit opium cultivation, are some of the territories that are facing a threat from opium use. Through research and development and the creation of better treatments, the government and medical community are attempting to combat this, creating the conditions for market expansion.
There is a substantial pipeline of treatments for tolerance on the market, and more ongoing trials are in progress. As a result, during the course of the projection period, the market for treating opioid tolerance will rise. One of the main drivers of growth will be the use in healthcare.
Due to increased opioid use for therapeutic and non-therapeutic uses, as well as increased opioid prescriptions, which can cause opioid tolerance if taken for an extended period of time, the market for opioid tolerance treatment is growing significantly.
Additional factors driving the growth of the opioid tolerance treatment market include ongoing clinical studies, various research projects being conducted for opioid tolerant therapy by different research institutes and companies, rising drug misuse, and an increase in drug overdose instances.
Restrain:
Digestive issues: When a patient begins taking opioids, they may have constipation, nausea, and vomiting. Grogginess, sluggish reflexes, or difficulty concentrating. Opioid addiction and tolerance: The patient may get addicted to opioids and require more of the medicine to relieve pain. Additionally, long-term opioid use can lead to dependency, and whenever the patient stops taking the prescription, they may experience withdrawal symptoms such anxiety, irritability, drug cravings, tremors, and others.
Additionally, market participants are concentrating on releasing non-opioid pain relief medicines, which is anticipated to slow market growth. For instance, the non-opioid post-operative pain medication Maxigesic IV was introduced to the European market by the speciality biopharmaceutical business Hyloris Pharmaceuticals SA in July 2022.
Opportunities:
The rising incidence of chronic pain, the rising number of surgical procedures, and the rising number of product approvals are the key drivers propelling the expansion of the global market for opioid tolerance treatment. But some of the issues limiting the growth of this market are the high cost of these therapies and the lack of awareness of these treatments.
| Opioid Tolerance Treatment Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 1.88 USD Bn |
| Forecast Period 2026-2032 CAGR: | 3.51% | Market Size in 2032: | 2.4 USD Bn |
| Segments Covered: | by Drug | NMDA Antagonists Ketamine Dextromethorphan Others (including Guaifenisin) Alpha2-adrenoceptor Agonists Clonidine Tizanidne Others (including Lofexidine) Others (including Endothelin Receptor Antagonists, NSAIDs, Opioid Combinations, and Multimodal Pain Management Drugs) |
|
| by Opioid Tolerance | Innate Tolerance Acquired Tolerance |
||
| by Route of Administration | Enteral Parenteral Others (including Transdermal Route, Patches, and Cartons of Patches) |
||
| by End-User | Hospitals & ASCs Rehabilitation Centers Others (including Home Care Settings and Research Institutes) |
||
Global Opioid Tolerance Treatment Market Segment Analysis:
In 2025, the NMDA Antagonists segment shows the highest demand in the opioid tolerance treatment market. Drugs such as ketamine and dextromethorphan are widely used due to their ability to modulate glutamate receptors and reduce opioid tolerance while improving analgesic effectiveness. These agents are increasingly incorporated into multimodal pain management strategies in hospitals and specialized pain clinics. Alpha2-adrenoceptor Agonists, including clonidine and tizanidine, are also commonly used to manage withdrawal symptoms and support opioid tolerance management, but their adoption is slightly lower compared to NMDA antagonists. Other treatment approaches, including endothelin receptor antagonists, NSAIDs, and opioid combination therapies, are used as supportive options in complex pain management cases.
Based on opioid tolerance type, Acquired Tolerance represents the most significant and widely treated segment in 2025. This form of tolerance develops after prolonged opioid exposure in patients undergoing long-term pain management, particularly in chronic pain and cancer treatment settings. Healthcare providers focus on adjusting therapeutic strategies and integrating alternative medications to maintain pain control while reducing opioid dependence. Innate Tolerance, which occurs naturally in some individuals due to genetic or physiological factors, represents a smaller share of the market but still requires targeted clinical approaches in certain patient populations.
Opioid Tolerance Treatment Market Regional Insights:
PORTER, PESTEL analysis with the potential impact of micro-economic factors of the market have been presented in the report. External as well as internal factors that are supposed to affect the business positively or negatively have been analyzed, which will give a clear futuristic view of the industry to the decision-makers.
The reports also help in understanding the Global Opioid Tolerance Treatment Market dynamic, structure by analyzing the market segments and projecting the Global Opioid Tolerance Treatment Market size. Clear representation of competitive analysis of key players by Vehicle type, price, financial position, product portfolio, growth strategies, and regional presence in the Global Opioid Tolerance Treatment Market make the report investor’s guide.
Global Opioid Tolerance Treatment Market Scope: Inquire before buying
Opioid Tolerance Treatment Market by Region
North America (United States, Canada and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan and Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A)
South America (Brazil, Argentina Rest of South America)
Key Players /Competitor Profiles Covered in the Opioid Tolerance Treatment Market Report from a Strategic Perspective.
1. Boehringer Ingelheim International GmbH
2. GlaxoSmithKline plc.
3. Teva Pharmaceutical Industries Ltd.
4. Mylan N.V.
5. Otsuka Pharmaceutical Co. Ltd.
6. Johnson & Johnson Services
7. Aurobindo Pharma
8. Pfizer Inc.
9. Acorda Therapeutics, Inc.
10.Zydus Cadila
11.Janssen Pharmaceuticals, Inc.,
12.Actavis Plc.,
13.Sanofi,
14.Sun Pharmaceutical Industries Ltd.,
15.Mallinckrodt Pharmaceuticals,
16.Endo Pharmaceuticals Inc.
17.Zyla Life Sciences.
