In Vivo Toxicology Market – Global Market Size, Strategic Growth Drivers, Risk Assessment Framework, Regulatory Landscape Review, Competitive Intensity Mapping & Long-Term Industry Outlook to 2032
Overview
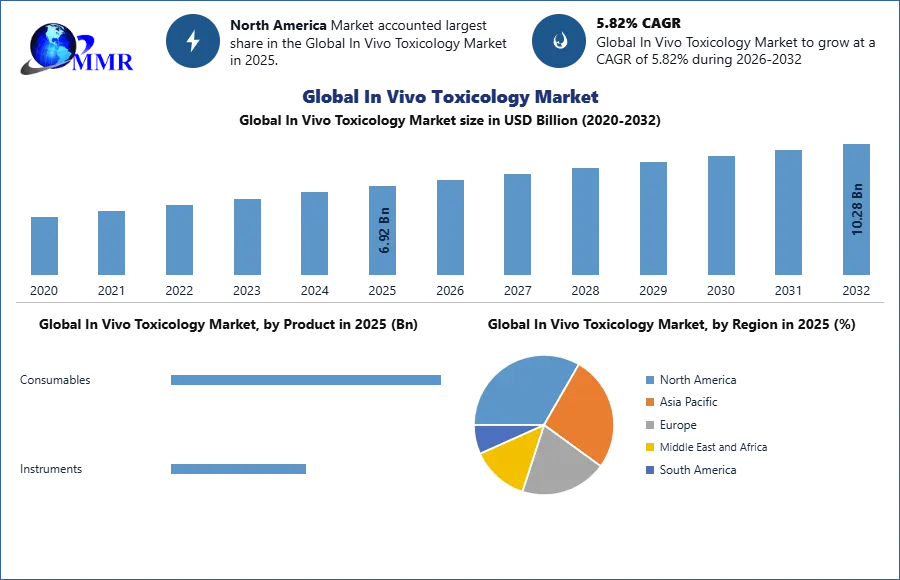
Global In Vivo Toxicology Market size was valued at USD 6.92 Bn. in 2025, and the is expected to grow at a CAGR of 5.82% from 2026 to 2032, reaching nearly USD 10.28 Bn. by 2032.
Global In Vivo Toxicology Market Overview:
The study of toxic effects of chemicals in non-human living organisms is known as in vivo toxicology. The use of living organisms to search for the existence of harmful or carcinogenic chemicals in new substances, as well as the impact of new substances on humans, is known as in-vivo toxicology research. These checks can also be used to ensure that the medication or product of choice does not contain any harmful chemicals. In-vivo testing is popular in pre-clinical studies. Primary in vivo toxicology tests aid in determining a drug's effects before first-in-human use.
In vivo toxicological experiments are carried out on guinea pigs, rats, rabbits, and other animals using a variety of exposure methods, including intravenous, topical, intramuscular, and others. Animal studies are carried out to see how industrial and environmental toxins affect the immune system.
A rise in R&D activities to find new drugs and vaccines has resulted from increasing demand for novel therapeutics. Testing of a drug in animals has been made obligatory by government regulatory bodies to prevent late-stage failures of a new drug and is, therefore, the market's main engine. However, animal models have been found to have some disadvantages as opposed to in vitro studies, which could affect market development.
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
Global In Vivo Toxicology Market Dynamics:
Drivers:
The growth of the global in-vivo toxicology market is fueled by factors such as rising research and development costs for detecting early-stage toxicity during the drug development phase, technical advances, and anti-animal testing sentiment. Furthermore, the development of novel in vivo toxicology studies are expected to boost the global demand for in vivo toxicology.
The increase in oncology research, the increased emphasis on the advancement of personalized medicine, and the demand for humanized animal models can all help the in vivo toxicology market grow.
Changing trends in the global healthcare markets have forced pharmaceutical and biotechnology companies to create products that provide tangible value rather than only incremental benefits. As a result, a growing number of pharmaceutical and medical device companies are focusing on innovation and improving their R&D performance. According to the Biotechnology Industry Organization (BIO) 2021, R&D for new therapeutics and innovative drugs accounted for approximately 82.7 percent of venture capital investment in the United States by emerging growth companies (EGCs) over the last decade. Similarly, designing biologic molecules/metabolites received 50% of venture capital investment in therapeutics. So, the Pharma and Biotechnology companies are also investing in research to advance novel molecules to serve to growing demands in the healthcare domain.
Restraints:
However, the disadvantages of animal testing and the growing demand to develop and implement alternative animal testing methods can limit the global in vivo toxicology market's development. Animal use in in vivo toxicology testing is being replaced, reduced, or refined by researchers and toxicology experts.
The Organization for Economic Co-operation and Development (OECD), the European Centre for the Validation of Alternative Methods (ECVAM), and the Interagency Coordinating Committee on the Validation of Alternative Methods have developed validation criteria for tests (ICCVAM).
Alternative tests include:
o In vitro test: These experiments are carried out in laboratories using different lab equipment in a controlled environment outside of a living organism.
o Ex vivo test: These experiments are carried out in or on tissues from animals in the wild, with only minor changes to the natural environment.
o In silico test: These experiments are performed using computer simulation.
In the coming years, the growing acceptance of alternative methods for toxicology research is expected to reduce the demand for animal models, which will affect the in vivo toxicology industry.
Global In Vivo Toxicology Market Segment Analysis:
Based on the Product, the global in vivo toxicology market is fragmented into consumables and instruments. The consumables segment is further fragmented into animal models, reagents & kits. Animal models are divided into three categories: rabbits, rats, and other species. In 2025, the consumables segment of the in vivo toxicology market had the largest share. The widespread use of reagents and kits in research activities, as well as an increase in funding for research projects and their widespread use in in vivo toxicology studies, are driving this segment's development.
Based on the Test Type, The global in vivo toxicology market is segmented into acute, sub-acute, chronic, and sub-chronic test types. The chronic test type segment category dominated the market in 2021, followed by the sub-chronic test type. The chronic test form market is growing due to increased research on drugs used for longer-duration treatment, such as anti-cancer, anti-convulsive, anti-arthritis, and anti-hypertensives.
Based on the Testing Facility, the global in vivo toxicology market is fragmented into outsourced testing facilities and in-house testing facilities. The outsourced testing facility segment accounted for the largest share in the market, in 2025. The large share of this segment can be attributed to pharmaceutical, biopharmaceutical, and medical device companies' increased R&D expenditures and cost-cutting strategies, resulting in increased outsourcing of services to CROs.
Based on the Toxicity End Point, the global in vivo toxicology market is fragmented into ADME (Absorption, Distribution, metabolism, and Excretion), immunotoxicity, systemic toxicity, carcinogenicity, genotoxicity, and developmental & reproductive toxicity. The immunotoxicity segment dominated the global in vivo toxicology market in 2025, led by the systemic toxicity segment. The immunotoxicity segment is expanding due to increased demand for biologics and biosimilars.
Based on the End Users, the global in vivo toxicology market is categorized into academic and research institutes, pharmaceutical and biotechnology companies, contract research organizations, food industry, and chemical industry. In 2025, the segment of academic and research institutes held the largest share of the in vivo toxicology market. The increase in the number of research activities in the field of in vivo toxicology, as well as funding to academic and research institutes to perform in vivo toxicology research.
Global In Vivo Toxicology Market Regional Insights:
In 2025, North America, which includes the US and Canada, has the largest share of the global in vivo toxicology market, followed by Europe. The area benefits from the presence of major players operating in the US, growing biomedical research in the US, rising preclinical activities by CROs and pharmaceutical companies in the region, significant government support, mandatory toxicology testing rules, and simplified toxicology guidelines, resulting in a higher number of studies.
North America, especially the United States, had the highest demand for in vivo toxicology products. North America has the highest revenue share, accounting for over 40% of total revenue, followed by Europe, which accounts for 30% of total revenue. In any given sector, over 99 percent of consumption is seen in ADME, safety pharmacology, and genotoxicity are the three primary fields.
During the forecast era, Asia Pacific is projected to be the highest augmentation in the Vivo toxicology market. Several global pharmaceutical companies have joined the APAC market to take advantage of the substantial growth opportunities in emerging Asian countries while also lowering their production costs by relocating their drug development R&D and manufacturing operations to the area. Some of the major factors driving this development are a large number of trained researchers and low-cost operations in APAC countries such as India and China. Furthermore, this demand is expected to expand moderately in the Middle East and Africa, as well as South America, in the coming years.
The objective of the report is to present a comprehensive analysis of the global in vivo toxicology market to the stakeholders in the industry. The past and current status of the industry with the forecasted market size and trends are presented in the report with the analysis of complicated data in simple language. The report covers all the aspects of the industry with a dedicated study of key players that include market leaders, followers, and new entrants.
PORTER, SVOR, PESTEL analysis with the potential impact of micro-economic factors of the market have been presented in the report. External as well as internal factors that are supposed to affect the business positively or negatively have been analyzed, which will give a clear futuristic view of the industry to the decision-makers.
The report also helps in understanding the global in vivo toxicology market dynamics, structure by analyzing the market segments and project the global in vivo toxicology market size. Clear representation of competitive analysis of key players by product, price, financial position, product portfolio, growth strategies, and regional presence in the global in vivo toxicology market make the report investor’s guide.
Global In Vivo Toxicology Market Key Developments:
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 11 December 2025 | Taconic Biosciences, Inc. | The company launched the FcResolv® NOG-EXL mouse model, the industry’s first platform featuring FcgR knockout technology. | This breakthrough eliminates murine Fc receptor interference, allowing for more precise and expedited preclinical outcomes in antibody-based drug studies. |
| 08 December 2025 | The Jackson Laboratory (JAX) | The Jackson Laboratory secured a $30 million contract from ARPA-H to lead the CARDIOVERSE project for AI-powered drug safety testing. | This initiative utilizes humanized mouse models and AI to create "virtual hearts," aiming to predict cardiotoxicity and reduce reliance on large-animal studies. |
| 20 October 2025 | The Jackson Laboratory (JAX) | JAX completed the acquisition of the New York Stem Cell Foundation (NYSCF) to unify genetics research with automated stem cell technology. | The merger creates a powerful platform for predictive toxicology, helping researchers identify disease mechanisms and safety risks much earlier in the R&D cycle. |
| 17 June 2025 | genOway | genOway announced the formal validation of its double humanized genO-hCD47/hSIRPα mouse model for toxicology assessments. | The model provides a critical tool for assessing next-generation checkpoint inhibitors, offering higher translational accuracy for immune-related safety profiles. |
| 21 January 2025 | Charles River Laboratories | Charles River Laboratories announced a manufacturing and research collaboration with Gazi University to support preclinical development programs. | The partnership strengthens the global network for discovery and safety assessment services, expanding access to GLP-compliant in vivo testing in the EMEA region. |
Global In Vivo Toxicology Market Scope: Inquire before buying
| Global In Vivo Toxicology Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 6.92 USD Billion |
| Forecast Period 2026-2032 CAGR: | 5.82% | Market Size in 2032: | 10.28 USD Billion |
| Segments Covered: | by Product | Consumables Instruments |
|
| by Test Type | Acute Toxicity Sub-Acute Toxicity Sub-Chronic Toxicity Chronic Toxicity |
||
| by Testing Facility | Outsourced Testing Facility In-house Testing Facility |
||
| by Toxicity End Point | Immunotoxicity Systemic Toxicity Carcinogenicity Genotoxicity ADME (Absorption, Distribution, Metabolism, and Excretion) Reproductive & Developmental Toxicity |
||
| by End Users | Academic and Research Institutes Pharmaceutical and Biotechnology Companies Contract Research Organizations Food Industry Chemical Industry |
||
Global In Vivo Toxicology Market, by Region:
• North America
• Europe
• Asia Pacific
• South America
• The Middle East and Africa
Global In Vivo Toxicology Market Key Players:
- PerkinElmer Inc.
- Eurofins Scientific
- Charles River Laboratories
- The Jackson Laboratory
- Envigo
- Taconic Biosciences, Inc.
- Janvier Labs
- Labcorp (Laboratory Corporation of America Holdings)
- Thermo Fisher Scientific Inc.
- Danaher Corporation
- Waters Corporation
- Agilent Technologies, Inc.
- Shimadzu Corporation
- Bruker Corporation
- Merck KGaA
- GE HealthCare Technologies Inc.
- Bio-Rad Laboratories, Inc.
- genOway
- Cyagen Biosciences
- GVK BIO
- PolyGene AG
- Crown Bioscience Inc.
- TransCure bioServices
- Ozgene Pty Ltd.
- Harbour BioMed
Others
