Global Pharmacovigilance and Drug Safety Software Market
1. Preface
1.1. Report Scope and Market Segmentation
1.2. Research Highlights
1.3. Research Objectives
1.4. Key Questions Answered
2. Assumptions and Research Methodology
2.1. Report Assumptions
2.2. Abbreviations Used
2.3. Research Methodology
3. Executive Summary
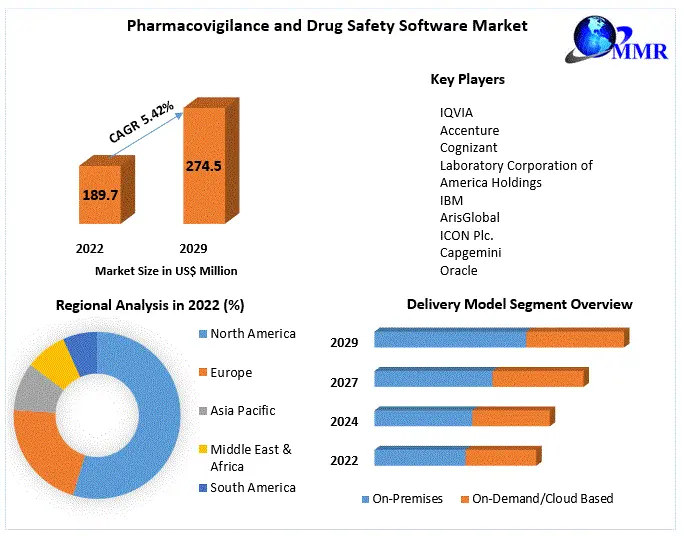
3.1. Global Pharmacovigilance and Drug Safety Software Market Size, by Market Value (US$ Mn) and Market, by Region
4. Market Overview
4.1. Introduction
4.2. Market Indicator
4.3. Drivers and Restraints Snapshot Analysis
4.3.1. Drivers
4.3.2. Restraints
4.3.3. Opportunities
4.3.4. Porter’s Analysis
4.3.5. Value Chain Analysis
4.3.6. SWOT Analysis
5. Global Pharmacovigilance and Drug Safety Software Market Analysis and Forecast
5.1. Global Pharmacovigilance and Drug Safety Software Market Analysis and Forecast
5.2. Global Pharmacovigilance and Drug Safety Software Market Size& Y-o-Y Growth Analysis
5.2.1. North America
5.2.2. Europe
5.2.3. Asia Pacific
5.2.4. Middle East & Africa
5.2.5. Latin America
6. Global Pharmacovigilance and Drug Safety Software Market Analysis and Forecast, by Functionality
6.1. Introduction and Definition
6.2. Key Findings
6.3. Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Functionality
6.4. Market Size (US$ Mn) Forecast, by Functionality
6.5. Global Pharmacovigilance and Drug Safety Software Market Analysis, by Functionality
6.6. Global Pharmacovigilance and Drug Safety Software Market Attractiveness Analysis, by Functionality
7. Global Pharmacovigilance and Drug Safety Software Market Analysis and Forecast, by Delivery Model
7.1. Introduction and Definition
7.2. Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Delivery Model
7.3. Market Size (US$ Mn) Forecast, by Delivery Model
7.4. Global Pharmacovigilance and Drug Safety Software Market Analysis, by Delivery Model
7.5. Global Pharmacovigilance and Drug Safety Software Market Attractiveness Analysis, by Delivery Model
8. Global Pharmacovigilance and Drug Safety Software Market Analysis and Forecast, by End-User
8.1. Introduction and Definition
8.2. Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by End-User
8.3. Market Size (US$ Mn) Forecast, by End-User
8.4. Global Pharmacovigilance and Drug Safety Software Market Analysis, by End-User
8.5. Global Pharmacovigilance and Drug Safety Software Market Attractiveness Analysis, by End-User
9. Global Pharmacovigilance and Drug Safety Software Market Analysis, by Region
9.1. Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Region
9.2. Market Size (US$ Mn) Forecast, by Region
9.3. Global Pharmacovigilance and Drug Safety Software Market Attractiveness Analysis, by Region
10. North America Global Pharmacovigilance and Drug Safety Software Market Analysis
10.1. Key Findings
10.2. North America Global Pharmacovigilance and Drug Safety Software Market Overview
10.3. North America Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Functionality
10.4. North America Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
10.4.1. Adverse Event Reporting Software
10.4.2. Drug Safety Audits Software
10.4.3. Issue Tracking Software
10.4.4. Fully Integrated Software
10.5. North America Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Delivery Model
10.6. North America Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
10.6.1. On-Premises
10.6.2. On-Demand/Cloud Based
10.7. North America Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by End-User
10.8. North America Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
10.8.1. Pharma and Biotech Companies
10.8.2. Contract Research Organizations (CROS)
10.8.3. Business Process Outsourcing (BPO) Firms
10.8.4. Other Pharmacovigilance Service Providers
10.9. North America Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Country
10.10. North America Global Pharmacovigilance and Drug Safety Software Market Forecast, by Country
10.10.1.1. U.S., 2018–2026
10.10.1.2. Canada, 2018–2026
10.11. North America Global Pharmacovigilance and Drug Safety Software Market Analysis, by Country
10.12. U.S. Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
10.12.1. Adverse Event Reporting Software
10.12.2. Drug Safety Audits Software
10.12.3. Issue Tracking Software
10.12.4. Fully Integrated Software
10.13. U.S. Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
10.13.1. On-Premises
10.13.2. On-Demand/Cloud Based
10.14. U.S. Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
10.14.1. Pharma and Biotech Companies
10.14.2. Contract Research Organizations (CROS)
10.14.3. Business Process Outsourcing (BPO) Firms
10.14.4. Other Pharmacovigilance Service Providers
10.15. Canada Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
10.15.1. Adverse Event Reporting Software
10.15.2. Drug Safety Audits Software
10.15.3. Issue Tracking Software
10.15.4. Fully Integrated Software
10.16. Canada Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
10.16.1. On-Premises
10.16.2. On-Demand/Cloud Based
10.17. Canada Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
10.17.1. Pharma and Biotech Companies
10.17.2. Contract Research Organizations (CROS)
10.17.3. Business Process Outsourcing (BPO) Firms
10.17.4. Other Pharmacovigilance Service Providers
10.18. North America Global Pharmacovigilance and Drug Safety Software Market Attractiveness Analysis
10.18.1. By Functionality
10.18.2. By Delivery Model
10.18.3. By End-User
10.19. PEST Analysis
11. Europe Global Pharmacovigilance and Drug Safety Software Market Analysis
11.1. Key Findings
11.2. Europe Global Pharmacovigilance and Drug Safety Software Market Overview
11.3. Europe Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Functionality
11.4. Europe Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
11.4.1. Adverse Event Reporting Software
11.4.2. Drug Safety Audits Software
11.4.3. Issue Tracking Software
11.4.4. Fully Integrated Software
11.5. Europe Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Delivery Model
11.6. Europe Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
11.6.1. On-Premises
11.6.2. On-Demand/Cloud Based
11.7. Europe Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by End-User
11.8. Europe Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
11.8.1. Pharma and Biotech Companies
11.8.2. Contract Research Organizations (CROS)
11.8.3. Business Process Outsourcing (BPO) Firms
11.8.4. Other Pharmacovigilance Service Providers
11.9. Europe Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Country
11.10. Europe Global Pharmacovigilance and Drug Safety Software Market Forecast, by Country
11.10.1.1. Germany
11.10.1.2. U.K.
11.10.1.3. France
11.10.1.4. Italy
11.10.1.5. Spain
11.10.1.6. Rest of Europe
11.11. Europe Global Pharmacovigilance and Drug Safety Software Market Analysis, by Country/ Sub-region
11.12. Germany Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
11.12.1. Adverse Event Reporting Software
11.12.2. Drug Safety Audits Software
11.12.3. Issue Tracking Software
11.12.4. Fully Integrated Software
11.13. Germany Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
11.13.1. On-Premises
11.13.2. On-Demand/Cloud Based
11.14. Germany Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
11.14.1. Pharma and Biotech Companies
11.14.2. Contract Research Organizations (CROS)
11.14.3. Business Process Outsourcing (BPO) Firms
11.14.4. Other Pharmacovigilance Service Providers
11.15. U.K. Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
11.15.1. Adverse Event Reporting Software
11.15.2. Drug Safety Audits Software
11.15.3. Issue Tracking Software
11.15.4. Fully Integrated Software
11.16. U.K. Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
11.16.1. On-Premises
11.16.2. On-Demand/Cloud Based
11.17. U.K. Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
11.17.1. Pharma and Biotech Companies
11.17.2. Contract Research Organizations (CROS)
11.17.3. Business Process Outsourcing (BPO) Firms
11.17.4. Other Pharmacovigilance Service Providers
11.18. France Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
11.18.1. Adverse Event Reporting Software
11.18.2. Drug Safety Audits Software
11.18.3. Issue Tracking Software
11.18.4. Fully Integrated Software
11.19. France Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
11.19.1. On-Premises
11.19.2. On-Demand/Cloud Based
11.20. France Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
11.20.1. Pharma and Biotech Companies
11.20.2. Contract Research Organizations (CROS)
11.20.3. Business Process Outsourcing (BPO) Firms
11.20.4. Other Pharmacovigilance Service Providers
11.21. Italy Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
11.21.1. Adverse Event Reporting Software
11.21.2. Drug Safety Audits Software
11.21.3. Issue Tracking Software
11.21.4. Fully Integrated Software
11.22. Italy Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
11.22.1. On-Premises
11.22.2. On-Demand/Cloud Based
11.23. Italy Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
11.23.1. Pharma and Biotech Companies
11.23.2. Contract Research Organizations (CROS)
11.23.3. Business Process Outsourcing (BPO) Firms
11.23.4. Other Pharmacovigilance Service Providers
11.24. Spain Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
11.24.1. Adverse Event Reporting Software
11.24.2. Drug Safety Audits Software
11.24.3. Issue Tracking Software
11.24.4. Fully Integrated Software
11.25. Spain Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
11.25.1. On-Premises
11.25.2. On-Demand/Cloud Based
11.26. Spain Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
11.26.1. Pharma and Biotech Companies
11.26.2. Contract Research Organizations (CROS)
11.26.3. Business Process Outsourcing (BPO) Firms
11.26.4. Other Pharmacovigilance Service Providers
11.27. Rest of Europe Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
11.27.1. Adverse Event Reporting Software
11.27.2. Drug Safety Audits Software
11.27.3. Issue Tracking Software
11.27.4. Fully Integrated Software
11.28. Rest of Europe Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
11.28.1. On-Premises
11.28.2. On-Demand/Cloud Based
11.29. Rest of Europe Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
11.29.1. Pharma and Biotech Companies
11.29.2. Contract Research Organizations (CROS)
11.29.3. Business Process Outsourcing (BPO) Firms
11.29.4. Other Pharmacovigilance Service Providers
11.30. Europe Global Pharmacovigilance and Drug Safety Software Market Attractiveness Analysis
11.30.1. By Functionality
11.30.2. By Delivery Model
11.30.3. By End-User
11.31. PEST Analysis
12. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Analysis
12.1. Key Findings
12.2. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Overview
12.3. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Functionality
12.4. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
12.4.1. Adverse Event Reporting Software
12.4.2. Drug Safety Audits Software
12.4.3. Issue Tracking Software
12.4.4. Fully Integrated Software
12.5. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Delivery Model
12.6. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
12.6.1. On-Premises
12.6.2. On-Demand/Cloud Based
12.7. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by End-User
12.8. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
12.8.1. Pharma and Biotech Companies
12.8.2. Contract Research Organizations (CROS)
12.8.3. Business Process Outsourcing (BPO) Firms
12.8.4. Other Pharmacovigilance Service Providers
12.9. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Country
12.10. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Forecast, by Country
12.10.1. China, 2018–2026
12.10.2. India, 2018–2026
12.10.3. Japan, 2018–2026
12.10.4. ASEAN, 2018–2026
12.10.5. Rest of Asia Pacific, 2018–2026
12.11. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Analysis, by Country/ Sub-region
12.12. China Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
12.12.1. Adverse Event Reporting Software
12.12.2. Drug Safety Audits Software
12.12.3. Issue Tracking Software
12.12.4. Fully Integrated Software
12.13. China Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
12.13.1. On-Premises
12.13.2. On-Demand/Cloud Based
12.14. China Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
12.14.1. Pharma and Biotech Companies
12.14.2. Contract Research Organizations (CROS)
12.14.3. Business Process Outsourcing (BPO) Firms
12.14.4. Other Pharmacovigilance Service Providers
12.15. India Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
12.15.1. Adverse Event Reporting Software
12.15.2. Drug Safety Audits Software
12.15.3. Issue Tracking Software
12.15.4. Fully Integrated Software
12.16. India Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
12.16.1. On-Premises
12.16.2. On-Demand/Cloud Based
12.17. India Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
12.17.1. Pharma and Biotech Companies
12.17.2. Contract Research Organizations (CROS)
12.17.3. Business Process Outsourcing (BPO) Firms
12.17.4. Other Pharmacovigilance Service Providers
12.18. Japan Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
12.18.1. Adverse Event Reporting Software
12.18.2. Drug Safety Audits Software
12.18.3. Issue Tracking Software
12.18.4. Fully Integrated Software
12.19. Japan Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
12.19.1. On-Premises
12.19.2. On-Demand/Cloud Based
12.20. Japan Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
12.20.1. Pharma and Biotech Companies
12.20.2. Contract Research Organizations (CROS)
12.20.3. Business Process Outsourcing (BPO) Firms
12.20.4. Other Pharmacovigilance Service Providers
12.21. ASEAN Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
12.21.1. Adverse Event Reporting Software
12.21.2. Drug Safety Audits Software
12.21.3. Issue Tracking Software
12.21.4. Fully Integrated Software
12.22. ASEAN Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
12.22.1. On-Premises
12.22.2. On-Demand/Cloud Based
12.23. ASEAN Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
12.23.1. Pharma and Biotech Companies
12.23.2. Contract Research Organizations (CROS)
12.23.3. Business Process Outsourcing (BPO) Firms
12.23.4. Other Pharmacovigilance Service Providers
12.24. Rest of Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
12.24.1. Adverse Event Reporting Software
12.24.2. Drug Safety Audits Software
12.24.3. Issue Tracking Software
12.24.4. Fully Integrated Software
12.25. Rest of Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
12.25.1. On-Premises
12.25.2. On-Demand/Cloud Based
12.26. Rest of Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
12.26.1. Pharma and Biotech Companies
12.26.2. Contract Research Organizations (CROS)
12.26.3. Business Process Outsourcing (BPO) Firms
12.26.4. Other Pharmacovigilance Service Providers
12.27. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Attractiveness Analysis
12.27.1. By Functionality
12.27.2. By Delivery Model
12.27.3. By End-User
12.28. PEST Analysis
13. Middle East & Africa Global Pharmacovigilance and Drug Safety Software Market Analysis
13.1. Key Findings
13.2. Middle East & Africa Global Pharmacovigilance and Drug Safety Software Market Overview
13.3. Middle East & Africa Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Functionality
13.4. Middle East & Africa Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
13.4.1. Adverse Event Reporting Software
13.4.2. Drug Safety Audits Software
13.4.3. Issue Tracking Software
13.4.4. Fully Integrated Software
13.5. Middle East & Africa Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Delivery Model
13.6. Middle East & Africa Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
13.6.1. On-Premises
13.6.2. On-Demand/Cloud Based
13.7. Middle East & Africa Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by End-User
13.8. Middle East & Africa Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
13.8.1. Pharma and Biotech Companies
13.8.2. Contract Research Organizations (CROS)
13.8.3. Business Process Outsourcing (BPO) Firms
13.8.4. Other Pharmacovigilance Service Providers
13.9. Middle East & Africa Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Country
13.10. Middle East & Africa Global Pharmacovigilance and Drug Safety Software Market Forecast, by Country
13.10.1. GCC, 2018–2026
13.10.2. South Africa, 2018–2026
13.10.3. Rest of Middle East & Africa, 2018–2026
13.11. Middle East & Africa Global Pharmacovigilance and Drug Safety Software Market Analysis, by Country/ Sub-region
13.12. GCC Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
13.12.1. Adverse Event Reporting Software
13.12.2. Drug Safety Audits Software
13.12.3. Issue Tracking Software
13.12.4. Fully Integrated Software
13.13. GCC Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
13.13.1. On-Premises
13.13.2. On-Demand/Cloud Based
13.14. GCC Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
13.14.1. Pharma and Biotech Companies
13.14.2. Contract Research Organizations (CROS)
13.14.3. Business Process Outsourcing (BPO) Firms
13.14.4. Other Pharmacovigilance Service Providers
13.15. South Africa Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
13.15.1. Adverse Event Reporting Software
13.15.2. Drug Safety Audits Software
13.15.3. Issue Tracking Software
13.15.4. Fully Integrated Software
13.16. South Africa Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
13.16.1. On-Premises
13.16.2. On-Demand/Cloud Based
13.17. South Africa Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
13.17.1. Pharma and Biotech Companies
13.17.2. Contract Research Organizations (CROS)
13.17.3. Business Process Outsourcing (BPO) Firms
13.17.4. Other Pharmacovigilance Service Providers
13.18. Rest of Middle East & Africa Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
13.18.1. Adverse Event Reporting Software
13.18.2. Drug Safety Audits Software
13.18.3. Issue Tracking Software
13.18.4. Fully Integrated Software
13.19. Rest of Middle East & Africa Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
13.19.1. On-Premises
13.19.2. On-Demand/Cloud Based
13.20. Rest of Middle East & Africa Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
13.20.1. Pharma and Biotech Companies
13.20.2. Contract Research Organizations (CROS)
13.20.3. Business Process Outsourcing (BPO) Firms
13.20.4. Other Pharmacovigilance Service Providers
13.21. Middle East & Africa Global Pharmacovigilance and Drug Safety Software Market Attractiveness Analysis
13.21.1. By Functionality
13.21.2. By Delivery Model
13.21.3. By End-User
13.22. PEST Analysis
14. Latin America Global Pharmacovigilance and Drug Safety Software Market Analysis
14.1. Key Findings
14.2. Latin America Global Pharmacovigilance and Drug Safety Software Market Overview
14.3. Latin America Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Functionality
14.4. Latin America Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
14.4.1. Adverse Event Reporting Software
14.4.2. Drug Safety Audits Software
14.4.3. Issue Tracking Software
14.4.4. Fully Integrated Software
14.5. Latin America Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Delivery Model
14.6. Latin America Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
14.6.1. On-Premises
14.6.2. On-Demand/Cloud Based
14.7. Latin America Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by End-User
14.8. Latin America Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
14.8.1. Pharma and Biotech Companies
14.8.2. Contract Research Organizations (CROS)
14.8.3. Business Process Outsourcing (BPO) Firms
14.8.4. Other Pharmacovigilance Service Providers
14.9. Latin America Global Pharmacovigilance and Drug Safety Software Market Value Share Analysis, by Country
14.10. Latin America Global Pharmacovigilance and Drug Safety Software Market Forecast, by Country
14.10.1.1. Brazil, 2018–2026
14.10.1.2. Mexico, 2018–2026
14.10.1.3. Rest of Latin America, 2018–2026
14.11. Latin America Global Pharmacovigilance and Drug Safety Software Market Analysis, by Country/ Sub-region
14.12. Brazil Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
14.12.1. Adverse Event Reporting Software
14.12.2. Drug Safety Audits Software
14.12.3. Issue Tracking Software
14.12.4. Fully Integrated Software
14.13. Brazil Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
14.13.1. On-Premises
14.13.2. On-Demand/Cloud Based
14.14. Brazil Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
14.14.1. Pharma and Biotech Companies
14.14.2. Contract Research Organizations (CROS)
14.14.3. Business Process Outsourcing (BPO) Firms
14.14.4. Other Pharmacovigilance Service Providers
14.15. Mexico Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
14.15.1. Adverse Event Reporting Software
14.15.2. Drug Safety Audits Software
14.15.3. Issue Tracking Software
14.15.4. Fully Integrated Software
14.16. Mexico Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
14.16.1. On-Premises
14.16.2. On-Demand/Cloud Based
14.17. Mexico Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
14.17.1. Pharma and Biotech Companies
14.17.2. Contract Research Organizations (CROS)
14.17.3. Business Process Outsourcing (BPO) Firms
14.17.4. Other Pharmacovigilance Service Providers
14.18. Rest of Latin America Global Pharmacovigilance and Drug Safety Software Market Forecast, by Functionality
14.18.1. Adverse Event Reporting Software
14.18.2. Drug Safety Audits Software
14.18.3. Issue Tracking Software
14.18.4. Fully Integrated Software
14.19. Rest of Latin America Global Pharmacovigilance and Drug Safety Software Market Forecast, by Delivery Model
14.19.1. On-Premises
14.19.2. On-Demand/Cloud Based
14.20. Rest of Latin America Global Pharmacovigilance and Drug Safety Software Market Forecast, by End-User
14.20.1. Pharma and Biotech Companies
14.20.2. Contract Research Organizations (CROS)
14.20.3. Business Process Outsourcing (BPO) Firms
14.20.4. Other Pharmacovigilance Service Providers
14.21. Latin America Global Pharmacovigilance and Drug Safety Software Market Attractiveness Analysis
14.21.1. By Functionality
14.21.2. By Delivery Model
14.21.3. By End-User
14.22. PEST Analysis
15. Company Profiles
15.1. Market Share Analysis, by Company
15.2. Competition Matrix
15.3. Company Profiles: Key Players
15.3.1. ArisGlobal
15.3.1.1. Company Overview
15.3.1.2. Financial Overview
15.3.1.3. Business Strategy
15.3.1.4. Recent Developments
15.3.1.5. Manufacturing Footprint
15.3.2. EXTEDO GmbH
15.3.3. Online Business Applications
15.3.4. Oracle Corporation
15.3.5. Sarjen Systems Pvt. Ltd
15.3.6. Sparta Systems, Inc.
15.3.7. United BioSource Corporation
15.3.8. United BioSource Corporation
15.3.9. Sparta Systems, Inc.
15.3.10. Oracle Corporation
15.3.11. Max Application
15.3.12. AB Cube
15.3.13. Relsys
15.3.14. UMBRA Global LLC
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report