eClinical Solutions Market Size by Product, Mode of Delivery, Clinical Trial, End-User, Region, Industry-Wide Analysis, Competitive Landscape Assessment & Long-Term Forecast to 2032
Overview
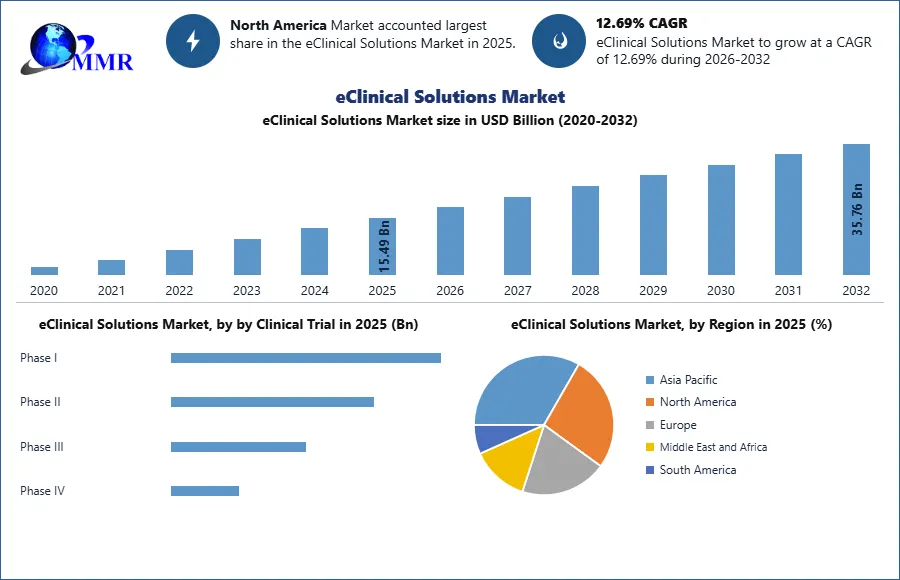
The eClinical Solutions Market was valued at USD15.49Bn in 2025, and the total revenue of the eClinical Solutions Market is expected to grow at a CAGR of 12.69% from 2026 to 2032, reaching nearly USD 35.76 Bn by 2032.
eClinical Solutions Market Overview
eClinical solutions are software and services designed to streamline clinical trials, including electronic data capture (EDC), clinical trial management systems (CTMS) and ePRO tools.
The eClinical solutions market growing due to the rising volume of clinical trials and stringent regulatory requirements, which necessitated efficient data management and compliance. Demand surged as pharmaceutical firms and CROs adopted digital solutions, while supply expanded with advancements in AI, cloud computing and decentralised trial models.
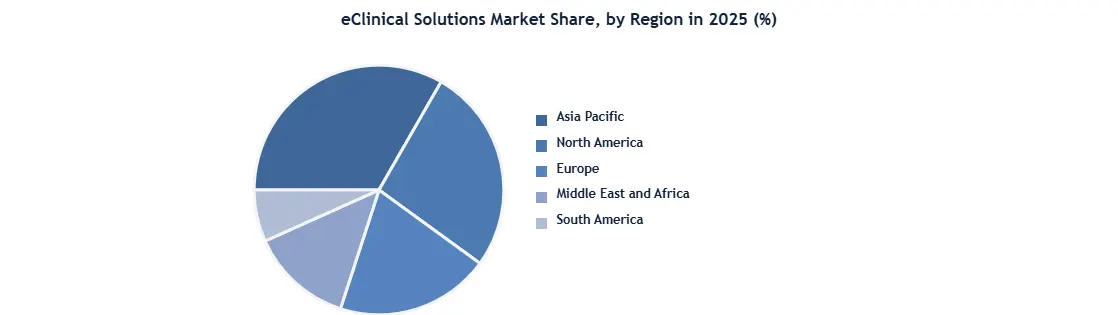
North America dominated, holding 45% revenue share in 2025, by robust R&D investments and high chronic disease prevalence. Also, Europe followed with strong regulatory frameworks and CRO partnerships. Key players like Oracle, Medidata and Veeva Systems led through technological innovation and strategic acquisitions. The market was further shaped by pharmaceutical companies as primary end-users, by data security concerns and high implementation costs posed challenges. Regulatory shifts, including FDA and EMA digital trial guidelines, also influenced adoption trends.
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
eClinical Solutions Market Dynamics
Increasing Number of Clinical Trials and Regulatory Pressures to Drive the eClinical Solution Market Growth
The eClinical Solutions Market is witnessing notable growth, driven by the rising number of clinical trials and intensified regulatory requirements. The expanding pharmaceutical and biotechnology sectors emphasize the need for efficient data management and collaborative tools, making eClinical solutions integral to clinical trial operations. As trials grow more complex, driven by trends like precision medicine and real-world evidence, these solutions offer the capability to manage large, diverse data sets effectively. Technological advancements, including AI, machine learning, and cloud computing, are further transforming the industry by improving efficiency and enabling innovation. Regulatory pressures surrounding data integrity and compliance are also accelerating market adoption.
Additionally, the outsourcing of trials to Contract Research Organisations (CROs) enhances demand for solutions that support seamless collaboration. Despite these drivers, high implementation costs, including software, training, and infrastructure, remain a challenge, especially for smaller firms. Nonetheless, the market continues to offer significant opportunities for growth, efficiency, and competitive differentiation.
Data Security and Privacy Concerns with Limited Infrastructure to Create eClinical Solutions Market Challenge
Data security and privacy concerns constitute another significant restraint for the eClinical Solutions Market. The sensitivity of clinical trial data underscores the importance of addressing potential risks related to data breaches or unauthorised access. The lack of in-house IT expertise, especially in smaller research entities, poses a challenge to the market penetration of eClinical solutions. Integration challenges with existing healthcare systems and resistance to change from traditional methods are additional factors restraining the industry's widespread adoption.
In developing regions, the limited infrastructure and resources present challenges for implementing and sustaining eClinical solutions. These regions offer untapped opportunities for market penetration, but overcoming infrastructure limitations becomes crucial. The complex regulatory landscape, varying across regions, adds another layer of complexity. Adapting eClinical solutions to comply with diverse regulatory requirements requires industry players to navigate the challenges associated with a global market. The eClinical Solutions Market showcases promising growth opportunities fueled by industry drivers such as the increasing number of clinical trials and technological advancements. While innovation in industry practices and emerging trends contribute to the market's potential, challenges related to implementation costs, data security, and regulatory complexities must be addressed to fully capitalise on the opportunities presented by eClinical solutions in the clinical research ecosystem.
eClinical Solutions Market Segment Analysis
Based on End-User, Pharmaceutical and biopharmaceutical companies segment dominated the eClinical Solutions Market in 2025,driving the demand for comprehensive eClinical solutions. The eClinical Solutions industry's influence on shaping eClinical trends is significant, with a focus on innovation and efficiency in trial management. CROs play a pivotal role in the eClinical Solutions Market, often forming strategic partnerships with solution providers to enhance their service offerings. The dynamics of the CRO segment influence eClinical solution features, emphasising collaboration and data sharing. Hospitals are increasingly adopting eClinical solutions, particularly for late-phase trials and real-world evidence generation.
As hospitals become more integral in clinical research, the eClinical Solutions Market has the opportunity to increase its reach and impact. Medical device manufacturers have unique requirements, leading to the development of specialised eClinical solutions catering to their specific needs. Ongoing innovation focuses on addressing the distinct challenges faced by medical device manufacturers in the clinical trial process. Consultancy service companies often play an advisory role in guiding organisations through the selection and implementation of eClinical solutions.
eClinical Solutions Market Regional Insights
North America led the worldwide market and accounted for the biggest revenue share of 45% in 2025, owing to the rising incidence of cardiovascular and respiratory disorders, technical developments in ECMO technology, and increased use of ECMO systems. According to the Centers for Disease Control and Prevention (CDC), 659,000 individuals in the United States would die from heart disease, accounting for one out of every four fatalities. In addition, about 805,000 people in the United States experience heart attacks. The region's requirement for Extracorporeal Membrane Oxygenation (ECMO) is also growing owing to the increased adoption of new technological goods and changes in lifestyle choices that result in chronic diseases such as cardiac and respiratory ailments.
An increase in the number of improvements and sophisticated technology in these Extracorporeal Membrane Oxygenation (ECMO) devices, as well as an increase in public awareness of their many applications, has resulted in a significant quantity of output for hospitals. Additionally, in the United States, high blood pressure, obesity, type 2 diabetes, and poor eating habits have all increased the number of instances of cardiovascular disease. ECMO (extracorporeal membrane oxygenation) has the potential to reduce the number of in-hospital mortalities.
eClinical Solutions Market Competitive Landscape
The eClinical solutions market is highly competitive, with key players such as Oracle Corporation, Medidata Solutions, Veeva Systems, ICON plc and Parexel International Corporation dominating the industry. Companies are led by their advanced technological innovations, strategic acquisitions and strong global presence.
Oracle Corporation focuses on an integrated cloud-based eClinical suite, leveraging AI and machine learning for real-time data analytics. Veeva Systems focuses on end-to-end clinical trial management, with a strong emphasis on regulatory compliance and Vault CDMS. ICON plc leads in data-driven clinical research, integrating AI for faster trial execution, while Parexel emphasises patient-centric trials with adaptive designs and real-world evidence (RWE) integration.
Medidata, a Dassault Systèmes company and a leading eClinical Solutions market share holder, has prolonged its partnership with Catalyst Clinical Research to support the global oncology brand, Catalyst Oncology. This partnership renewal includes the use of Medidata Rave EDC, Rave CTMS, and Rave eTMF, along with additional offerings like Medidata Grants Manager and Medidata AI Intelligent Trials. The collaboration enables Catalyst to run complex clinical trials efficiently, streamline workflows, automate document management, and enhance visibility into critical trial metrics. Catalyst, a prominent manufacturer in the eClinical Solutions market, has managed over 150 oncology studies and currently oversees more than 80 next-generation cancer clinical trials across Phases I-III.
eClinical Solutions Market Key Trends
| Trend | Description | Impact on Market |
| Decentralised Clinical Trials (DCTs) | Remote monitoring, telehealth, and digital patient engagement tools reduce site visits. | Increases trial efficiency, patient recruitment, and retention. |
| AI & Machine Learning Integration | AI-driven analytics for patient matching, risk prediction, and real-time data insights. | Speeds up trial timelines, reduces costs, and improves accuracy. |
| Cloud-Based eClinical Platforms | Shift from on-premise to SaaS solutions for scalability and remote access. | Enhances collaboration, data security, and regulatory compliance. |
eClinical Solutions Market Recent Development
• On March 19, 2025, Medidata Solutions (United States) announced that its parent company, Dassault Systèmes, made a strategic investment in Click Therapeutics to advance prescription digital therapeutics, aiming to enhance the patient experience across clinical and real-world care.
• On January 29, 2025, Veeva Systems (United States) reported that over 200 companies, including 17 of the top 20 biopharma firms, adopted its Veeva CTMS platform to improve trial efficiency.
• On March 11, 2025, Veeva Systems (United States) launched the “One Medicine Platform” with Boehringer Ingelheim, using Veeva Development Cloud to unify clinical, regulatory, and quality data.
Eclinical Solutions Industry Ecosystem:
eClinical Solutions Market Scope: Inquire before buying
| eClinical Solutions Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 15.49 USD Billion |
| Forecast Period 2026-2032 CAGR: | 12.69% | Market Size in 2032: | 35.76 USD Billion |
| Segments Covered: | by Product | Electronic Data Capture (EDC) Clinical Data Management Systems (CDMS) Clinical Trial Management Systems (CTMS) Randomization and Trial Supply Management (RTSM) Electronic Patient Reported Outcomes (ePRO) Electronic Clinical Outcome Assessment (eCOA) Clinical Analytics Platforms Clinical Data Integration Platforms eConsent Safety Solutions (pharmacovigilance platforms) Regulatory Information Management (RIM) Risk-Based Monitoring (RBM) Electronic Trial Master File (eTMF) Wearable/Remote Monitoring Devices Integration Artificial Intelligence/Machine Learning in eClinical Others |
|
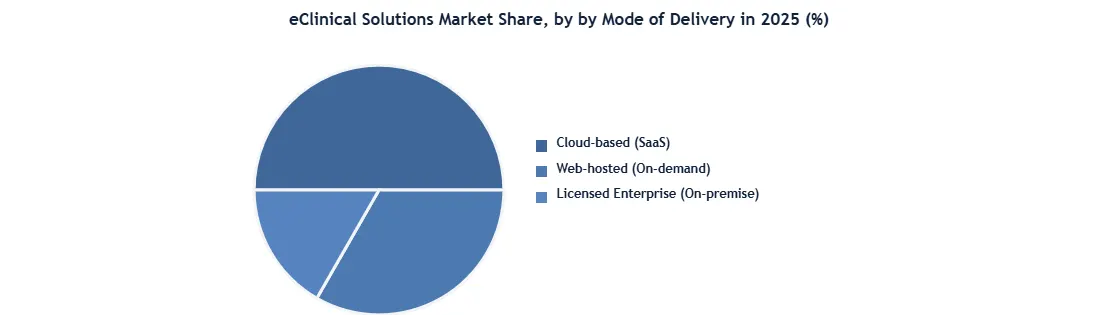
| by Mode of Delivery | Web-hosted (On-demand) Licensed Enterprise (On-premise) Cloud-based (SaaS) |
||
| by Clinical Trial | Phase I Phase II Phase III Phase IV |
||
| by Application | Data Collection Document Management & Storage Supply Management Data Analytics Clinical Trial Operations Regulatory Information Management Other Applications |
||
| by End-User | Pharmaceuticals & Biopharmaceutical Companies Contract Research Organizers Hospitals Medical Device Manufacturer Consultancy Service Companies Academic & Research Institutes |
||
eClinical Solutions Market, by Region
North America (United States, Canada, and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria, and the Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, ASEAN, Indonesia, Philippines, Malaysia, Vietnam, Thailand, ASEAN, Rest of Asia Pacific)
Middle East & Africa (South Africa, GCC, Nigeria, Rest of ME&A)
South America (Brazil, Argentina, Rest of South America)
eClinical Solutions Market, Key Players
- Medidata Solutions.
- Oracle Corporation
- Veeva Systems
- Parexel International Corporation
- PRA Health Sciences
- Medrio, Inc.
- eClinicalWorks
- IBM Corporation
- Bio‑Optronics, Inc.
- Datatrak International, Inc.
- Bioclinica (Clario)
- CRF Health (Signant Health)
- IQVIA Inc.
- Anju Software, Inc.
- OpenClinica, LLC
- MasterControl, Inc.
- ClinCapture, Inc.
- Medable, Inc.
- Castor EDC
- Saama Technologies, Inc.
- OmniComm Systems, Inc.
- Forte Research Systems, Inc.
- TransPerfect Life Sciences
- Perceptive (formerly Calyx)
- Genedata
- Clinipace
- ERT Clinical (Clario)
- Datamed
- Tigermed
- CMIC Holdings Co., Ltd.
Others