Global Recombinant Plasma Protein Therapeutics Market
1. Preface
1.1. Report Scope and Market Segmentation
1.2. Research Highlights
1.3. Research Objectives
2. Assumptions and Research Methodology
2.1. Report Assumptions
2.2. Abbreviations
2.3. Research Methodology
2.3.1. Secondary Research
2.3.1.1. Secondary data
2.3.1.2. Secondary Sources
2.3.2. Primary Research
2.3.2.1. Data from Primary Sources
2.3.2.2. Breakdown of Primary Sources
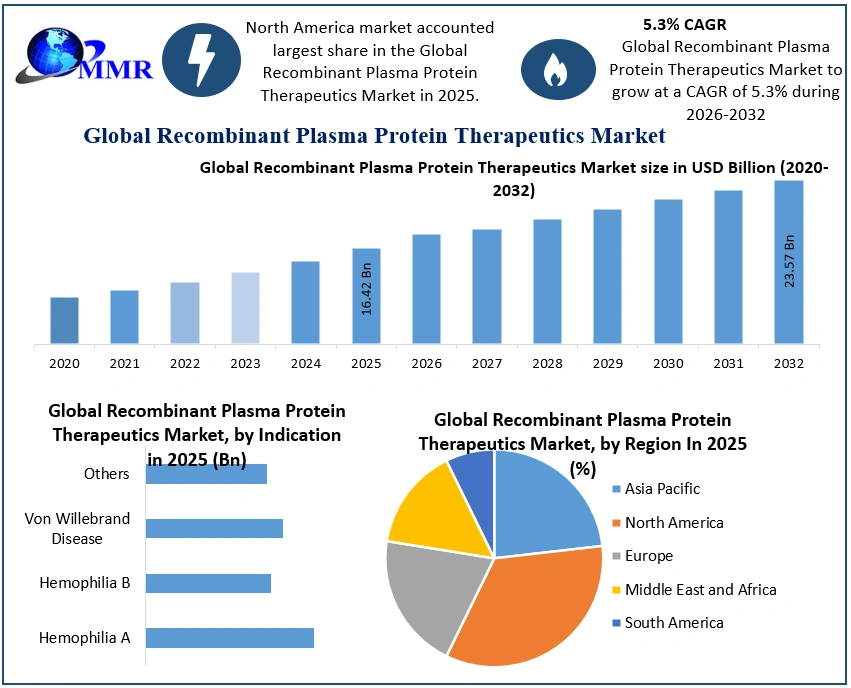
3. Executive Summary: Global Recombinant Plasma Protein Therapeutics Market Size, by Market Value (US$ Bn)
4. Market Overview
4.1. Introduction
4.2. Market Indicator
4.2.1. Drivers
4.2.2. Restraints
4.2.3. Opportunities
4.2.4. Challenges
4.3. Porter’s Analysis
4.4. Value Chain Analysis
4.5. Market Risk Analysis
4.6. SWOT Analysis
4.7. Industry Trends and Emerging Technologies
5. Supply Side and Demand Side Indicators
6. Global Recombinant Plasma Protein Therapeutics Market Analysis and Forecast
6.1. Global Recombinant Plasma Protein Therapeutics Market Size & Y-o-Y Growth Analysis
6.1.1. North America
6.1.2. Europe
6.1.3. Asia Pacific
6.1.4. Middle East & Africa
6.1.5. South America
7. Global Recombinant Plasma Protein Therapeutics Market Analysis and Forecast, by Drug Class
7.1. Introduction and Definition
7.2. Key Findings
7.3. Global Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Drug Class
7.4. Global Recombinant Plasma Protein Therapeutics Market Size (US$ Bn) Forecast, by Drug Class
7.5. Global Recombinant Plasma Protein Therapeutics Market Analysis, by Drug Class
7.6. Global Recombinant Plasma Protein Therapeutics Market Attractiveness Analysis, by Drug Class
8. Global Recombinant Plasma Protein Therapeutics Market Analysis and Forecast, by Cell Line
8.1. Introduction and Definition
8.2. Key Findings
8.3. Global Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Cell Line
8.4. Global Recombinant Plasma Protein Therapeutics Market Size (US$ Bn) Forecast, by Cell Line
8.5. Global Recombinant Plasma Protein Therapeutics Market Analysis, by Cell Line
8.6. Global Recombinant Plasma Protein Therapeutics Market Attractiveness Analysis, by Cell Line
9. Global Recombinant Plasma Protein Therapeutics Market Analysis and Forecast, by Indication
9.1. Introduction and Definition
9.2. Key Findings
9.3. Global Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Indication
9.4. Global Recombinant Plasma Protein Therapeutics Market Size (US$ Bn) Forecast, by Indication
9.5. Global Recombinant Plasma Protein Therapeutics Market Analysis, by Indication
9.6. Global Recombinant Plasma Protein Therapeutics Market Attractiveness Analysis, by Indication
10. Global Recombinant Plasma Protein Therapeutics Market Analysis, by Region
10.1. Global Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Region
10.2. Global Recombinant Plasma Protein Therapeutics Market Size (US$ Bn) Forecast, by Region
10.3. Global Recombinant Plasma Protein Therapeutics Market Attractiveness Analysis, by Region
11. North America Recombinant Plasma Protein Therapeutics Market Analysis
11.1. Key Findings
11.2. North America Recombinant Plasma Protein Therapeutics Market Overview
11.3. North America Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Drug Class
11.4. North America Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
11.4.1. Recombinant Coagulation Factors
11.4.1.1. Recombinant Coagulation Factor VIII
11.4.1.2. Recombinant Coagulation Factor IX
11.4.1.3. Recombinant Coagulation Factor VIIa
11.4.1.4. Others
11.4.2. Human C1 Esterase Inhibitor
11.5. North America Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Cell Line
11.6. North America Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
11.6.1. Chinese Hamster Ovary (CHO) Cell Line
11.6.2. Baby Hamster Kidney (BHK) Cell Line
11.6.3. Human Embryonic Kidney (HEK) Cell Line
11.6.4. Others
11.7. North America Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Indication
11.8. North America Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
11.8.1. Hemophilia A
11.8.2. Hemophilia B
11.8.3. Von Willebrand Disease
11.8.4. Others
11.9. North America Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Country
11.10. North America Recombinant Plasma Protein Therapeutics Market Forecast, by Country
11.10.1. U.S.
11.10.2. Canada
11.11. North America Recombinant Plasma Protein Therapeutics Market Analysis, by Country
11.12. U.S. Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
11.12.1. Recombinant Coagulation Factors
11.12.1.1. Recombinant Coagulation Factor VIII
11.12.1.2. Recombinant Coagulation Factor IX
11.12.1.3. Recombinant Coagulation Factor VIIa
11.12.1.4. Others
11.12.2. Human C1 Esterase Inhibitor
11.13. U.S. Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
11.13.1. Chinese Hamster Ovary (CHO) Cell Line
11.13.2. Baby Hamster Kidney (BHK) Cell Line
11.13.3. Human Embryonic Kidney (HEK) Cell Line
11.13.4. Others
11.14. U.S. Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
11.14.1. Hemophilia A
11.14.2. Hemophilia B
11.14.3. Von Willebrand Disease
11.14.4. Others
11.15. Canada Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
11.15.1. Recombinant Coagulation Factors
11.15.1.1. Recombinant Coagulation Factor VIII
11.15.1.2. Recombinant Coagulation Factor IX
11.15.1.3. Recombinant Coagulation Factor VIIa
11.15.1.4. Others
11.15.2. Human C1 Esterase Inhibitor
11.16. Canada Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
11.16.1. Chinese Hamster Ovary (CHO) Cell Line
11.16.2. Baby Hamster Kidney (BHK) Cell Line
11.16.3. Human Embryonic Kidney (HEK) Cell Line
11.16.4. Others
11.17. Canada Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
11.17.1. Hemophilia A
11.17.2. Hemophilia B
11.17.3. Von Willebrand Disease
11.17.4. Others
11.18. North America Recombinant Plasma Protein Therapeutics Market Attractiveness Analysis
11.18.1. By Drug Class
11.18.2. By Cell Line
11.18.3. By Indication
11.19. PEST Analysis
11.20. Key Trends
11.21. Key Developments
12. Europe Recombinant Plasma Protein Therapeutics Market Analysis
12.1. Key Findings
12.2. Europe Recombinant Plasma Protein Therapeutics Market Overview
12.3. Europe Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Drug Class
12.4. Europe Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
12.4.1. Recombinant Coagulation Factors
12.4.1.1. Recombinant Coagulation Factor VIII
12.4.1.2. Recombinant Coagulation Factor IX
12.4.1.3. Recombinant Coagulation Factor VIIa
12.4.1.4. Others
12.4.2. Human C1 Esterase Inhibitor
12.5. Europe Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Cell Line
12.6. Europe Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
12.6.1. Chinese Hamster Ovary (CHO) Cell Line
12.6.2. Baby Hamster Kidney (BHK) Cell Line
12.6.3. Human Embryonic Kidney (HEK) Cell Line
12.6.4. Others
12.7. Europe Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Indication
12.8. Europe Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
12.8.1. Hemophilia A
12.8.2. Hemophilia B
12.8.3. Von Willebrand Disease
12.8.4. Others
12.9. Europe Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Country
12.10. Europe Recombinant Plasma Protein Therapeutics Market Forecast, by Country
12.10.1. Germany
12.10.2. U.K.
12.10.3. France
12.10.4. Italy
12.10.5. Spain
12.10.6. Rest of Europe
12.11. Europe Recombinant Plasma Protein Therapeutics Market Analysis, by Country
12.12. Europe Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
12.12.1. Recombinant Coagulation Factors
12.12.1.1. Recombinant Coagulation Factor VIII
12.12.1.2. Recombinant Coagulation Factor IX
12.12.1.3. Recombinant Coagulation Factor VIIa
12.12.1.4. Others
12.12.2. Human C1 Esterase Inhibitor
12.13. Germany Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
12.13.1. Chinese Hamster Ovary (CHO) Cell Line
12.13.2. Baby Hamster Kidney (BHK) Cell Line
12.13.3. Human Embryonic Kidney (HEK) Cell Line
12.13.4. Others
12.14. Germany Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
12.14.1. Hemophilia A
12.14.2. Hemophilia B
12.14.3. Von Willebrand Disease
12.14.4. Others
12.15. U.K. Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
12.15.1. Recombinant Coagulation Factors
12.15.1.1. Recombinant Coagulation Factor VIII
12.15.1.2. Recombinant Coagulation Factor IX
12.15.1.3. Recombinant Coagulation Factor VIIa
12.15.1.4. Others
12.15.2. Human C1 Esterase Inhibitor
12.16. U.K. Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
12.16.1. Chinese Hamster Ovary (CHO) Cell Line
12.16.2. Baby Hamster Kidney (BHK) Cell Line
12.16.3. Human Embryonic Kidney (HEK) Cell Line
12.16.4. Others
12.17. U.K. Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
12.17.1. Hemophilia A
12.17.2. Hemophilia B
12.17.3. Von Willebrand Disease
12.17.4. Others
12.18. France Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
12.18.1. Recombinant Coagulation Factors
12.18.1.1. Recombinant Coagulation Factor VIII
12.18.1.2. Recombinant Coagulation Factor IX
12.18.1.3. Recombinant Coagulation Factor VIIa
12.18.1.4. Others
12.18.2. Human C1 Esterase Inhibitor
12.19. France Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
12.19.1. Chinese Hamster Ovary (CHO) Cell Line
12.19.2. Baby Hamster Kidney (BHK) Cell Line
12.19.3. Human Embryonic Kidney (HEK) Cell Line
12.19.4. Others
12.20. France Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
12.20.1. Hemophilia A
12.20.2. Hemophilia B
12.20.3. Von Willebrand Disease
12.20.4. Others
12.21. Italy Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
12.21.1. Recombinant Coagulation Factors
12.21.1.1. Recombinant Coagulation Factor VIII
12.21.1.2. Recombinant Coagulation Factor IX
12.21.1.3. Recombinant Coagulation Factor VIIa
12.21.1.4. Others
12.21.2. Human C1 Esterase Inhibitor
12.22. Italy Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
12.22.1. Chinese Hamster Ovary (CHO) Cell Line
12.22.2. Baby Hamster Kidney (BHK) Cell Line
12.22.3. Human Embryonic Kidney (HEK) Cell Line
12.22.4. Others
12.23. Italy Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
12.23.1. Hemophilia A
12.23.2. Hemophilia B
12.23.3. Von Willebrand Disease
12.23.4. Others
12.24. Spain Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
12.24.1. Recombinant Coagulation Factors
12.24.1.1. Recombinant Coagulation Factor VIII
12.24.1.2. Recombinant Coagulation Factor IX
12.24.1.3. Recombinant Coagulation Factor VIIa
12.24.1.4. Others
12.24.2. Human C1 Esterase Inhibitor
12.25. Spain Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
12.25.1. Chinese Hamster Ovary (CHO) Cell Line
12.25.2. Baby Hamster Kidney (BHK) Cell Line
12.25.3. Human Embryonic Kidney (HEK) Cell Line
12.25.4. Others
12.26. Spain Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
12.26.1. Hemophilia A
12.26.2. Hemophilia B
12.26.3. Von Willebrand Disease
12.26.4. Others
12.27. Rest of Europe Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
12.27.1. Recombinant Coagulation Factors
12.27.1.1. Recombinant Coagulation Factor VIII
12.27.1.2. Recombinant Coagulation Factor IX
12.27.1.3. Recombinant Coagulation Factor VIIa
12.27.1.4. Others
12.27.2. Human C1 Esterase Inhibitor
12.28. Rest of Europe Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
12.28.1. Chinese Hamster Ovary (CHO) Cell Line
12.28.2. Baby Hamster Kidney (BHK) Cell Line
12.28.3. Human Embryonic Kidney (HEK) Cell Line
12.28.4. Others
12.29. Rest Of Europe Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
12.29.1. Hemophilia A
12.29.2. Hemophilia B
12.29.3. Von Willebrand Disease
12.29.4. Others
12.30. Europe Recombinant Plasma Protein Therapeutics Market Attractiveness Analysis
12.30.1. By Drug Class
12.30.2. By Cell Line
12.30.3. By Indication
12.31. PEST Analysis
12.32. Key Trends
12.33. Key Developments
13. Asia Pacific Recombinant Plasma Protein Therapeutics Market Analysis
13.1. Key Findings
13.2. Asia Pacific Recombinant Plasma Protein Therapeutics Market Overview
13.3. Asia Pacific Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Drug Class
13.4. Asia Pacific Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
13.4.1. Recombinant Coagulation Factors
13.4.1.1. Recombinant Coagulation Factor VIII
13.4.1.2. Recombinant Coagulation Factor IX
13.4.1.3. Recombinant Coagulation Factor VIIa
13.4.1.4. Others
13.4.2. Human C1 Esterase Inhibitor
13.5. Asia Pacific Recombinant Plasma Protein Therapeutics Market Value Share Analysis, By Cell Line
13.6. Asia Pacific Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
13.6.1. Chinese Hamster Ovary (CHO) Cell Line
13.6.2. Baby Hamster Kidney (BHK) Cell Line
13.6.3. Human Embryonic Kidney (HEK) Cell Line
13.6.4. Others
13.7. Asia Pacific Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Indication
13.8. Asia Pacific Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
13.8.1. Hemophilia A
13.8.2. Hemophilia B
13.8.3. Von Willebrand Disease
13.8.4. Others
13.9. Asia Pacific Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Country
13.10. Asia Pacific Recombinant Plasma Protein Therapeutics Market Forecast, by Country
13.10.1. China
13.10.2. India
13.10.3. Japan
13.10.4. ASEAN
13.10.5. Rest of Asia Pacific
13.11. Asia Pacific Recombinant Plasma Protein Therapeutics Market Analysis, by Country
13.12. China Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
13.12.1. Recombinant Coagulation Factors
13.12.1.1. Recombinant Coagulation Factor VIII
13.12.1.2. Recombinant Coagulation Factor IX
13.12.1.3. Recombinant Coagulation Factor VIIa
13.12.1.4. Others
13.12.2. Human C1 Esterase Inhibitor
13.13. China Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
13.13.1. Chinese Hamster Ovary (CHO) Cell Line
13.13.2. Baby Hamster Kidney (BHK) Cell Line
13.13.3. Human Embryonic Kidney (HEK) Cell Line
13.13.4. Others
13.14. China Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
13.14.1. Hemophilia A
13.14.2. Hemophilia B
13.14.3. Von Willebrand Disease
13.14.4. Others
13.15. India Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
13.15.1. Recombinant Coagulation Factors
13.15.1.1. Recombinant Coagulation Factor VIII
13.15.1.2. Recombinant Coagulation Factor IX
13.15.1.3. Recombinant Coagulation Factor VIIa
13.15.1.4. Others
13.15.2. Human C1 Esterase Inhibitor
13.16. India Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
13.16.1. Chinese Hamster Ovary (CHO) Cell Line
13.16.2. Baby Hamster Kidney (BHK) Cell Line
13.16.3. Human Embryonic Kidney (HEK) Cell Line
13.16.4. Others
13.17. India Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
13.17.1. Hemophilia A
13.17.2. Hemophilia B
13.17.3. Von Willebrand Disease
13.17.4. Others
13.18. Japan Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
13.18.1. Recombinant Coagulation Factors
13.18.1.1. Recombinant Coagulation Factor VIII
13.18.1.2. Recombinant Coagulation Factor IX
13.18.1.3. Recombinant Coagulation Factor VIIa
13.18.1.4. Others
13.18.2. Human C1 Esterase Inhibitor
13.19. Japan Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
13.19.1. Chinese Hamster Ovary (CHO) Cell Line
13.19.2. Baby Hamster Kidney (BHK) Cell Line
13.19.3. Human Embryonic Kidney (HEK) Cell Line
13.19.4. Others
13.20. Japan Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
13.20.1. Hemophilia A
13.20.2. Hemophilia B
13.20.3. Von Willebrand Disease
13.20.4. Others
13.21. ASEAN Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
13.21.1. Recombinant Coagulation Factors
13.21.1.1. Recombinant Coagulation Factor VIII
13.21.1.2. Recombinant Coagulation Factor IX
13.21.1.3. Recombinant Coagulation Factor VIIa
13.21.1.4. Others
13.21.2. Human C1 Esterase Inhibitor
13.22. ASEAN Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
13.22.1. Chinese Hamster Ovary (CHO) Cell Line
13.22.2. Baby Hamster Kidney (BHK) Cell Line
13.22.3. Human Embryonic Kidney (HEK) Cell Line
13.22.4. Others
13.23. ASEAN Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
13.23.1. Hemophilia A
13.23.2. Hemophilia B
13.23.3. Von Willebrand Disease
13.23.4. Others
13.24. Rest of Asia Pacific Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
13.24.1. Recombinant Coagulation Factors
13.24.1.1. Recombinant Coagulation Factor VIII
13.24.1.2. Recombinant Coagulation Factor IX
13.24.1.3. Recombinant Coagulation Factor VIIa
13.24.1.4. Others
13.24.2. Human C1 Esterase Inhibitor
13.25. Rest of Asia Pacific Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
13.25.1. Chinese Hamster Ovary (CHO) Cell Line
13.25.2. Baby Hamster Kidney (BHK) Cell Line
13.25.3. Human Embryonic Kidney (HEK) Cell Line
13.25.4. Others
13.26. Rest of Asia Pacific Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
13.26.1. Hemophilia A
13.26.2. Hemophilia B
13.26.3. Von Willebrand Disease
13.26.4. Others
13.27. Asia Pacific Recombinant Plasma Protein Therapeutics Market Attractiveness Analysis
13.27.1. By Drug Class
13.27.2. By Cell Line
13.27.3. By Indication
13.28. PEST Analysis
13.29. Key Trends
13.30. Key Developments
14. Middle East & Africa Recombinant Plasma Protein Therapeutics Market Analysis
14.1. Key Findings
14.2. Middle East & Africa Recombinant Plasma Protein Therapeutics Market Overview
14.3. Middle East & Africa Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Drug Class
14.4. Middle East & Africa Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
14.4.1. Recombinant Coagulation Factors
14.4.1.1. Recombinant Coagulation Factor VIII
14.4.1.2. Recombinant Coagulation Factor IX
14.4.1.3. Recombinant Coagulation Factor VIIa
14.4.1.4. Others
14.4.2. Human C1 Esterase Inhibitor
14.5. Middle East & Africa Recombinant Plasma Protein Therapeutics Market Value Share Analysis, By Cell Line
14.6. Middle East & Africa Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
14.6.1. Chinese Hamster Ovary (CHO) Cell Line
14.6.2. Baby Hamster Kidney (BHK) Cell Line
14.6.3. Human Embryonic Kidney (HEK) Cell Line
14.6.4. Others
14.7. Middle East & Africa Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Indication
14.8. Middle East & Africa Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
14.8.1. Hemophilia A
14.8.2. Hemophilia B
14.8.3. Von Willebrand Disease
14.8.4. Others
14.9. Middle East & Africa Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Country
14.10. Middle East & Africa Recombinant Plasma Protein Therapeutics Market Forecast, by Country
14.10.1. GCC
14.10.2. South Africa
14.10.3. Rest of Middle East & Africa
14.11. Middle East & Africa Recombinant Plasma Protein Therapeutics Market Analysis, by Country
14.12. GCC Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
14.12.1. Recombinant Coagulation Factors
14.12.1.1. Recombinant Coagulation Factor VIII
14.12.1.2. Recombinant Coagulation Factor IX
14.12.1.3. Recombinant Coagulation Factor VIIa
14.12.1.4. Others
14.12.2. Human C1 Esterase Inhibitor
14.13. GCC Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
14.13.1. Chinese Hamster Ovary (CHO) Cell Line
14.13.2. Baby Hamster Kidney (BHK) Cell Line
14.13.3. Human Embryonic Kidney (HEK) Cell Line
14.13.4. Others
14.14. GCC Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
14.14.1. Hemophilia A
14.14.2. Hemophilia B
14.14.3. Von Willebrand Disease
14.14.4. Others
14.15. South Africa Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
14.15.1. Recombinant Coagulation Factors
14.15.1.1. Recombinant Coagulation Factor VIII
14.15.1.2. Recombinant Coagulation Factor IX
14.15.1.3. Recombinant Coagulation Factor VIIa
14.15.1.4. Others
14.15.2. Human C1 Esterase Inhibitor
14.16. South Africa Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
14.16.1. Chinese Hamster Ovary (CHO) Cell Line
14.16.2. Baby Hamster Kidney (BHK) Cell Line
14.16.3. Human Embryonic Kidney (HEK) Cell Line
14.16.4. Others
14.17. South Africa Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
14.17.1. Hemophilia A
14.17.2. Hemophilia B
14.17.3. Von Willebrand Disease
14.17.4. Others
14.18. Rest of Middle East & Africa Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
14.18.1. Recombinant Coagulation Factors
14.18.1.1. Recombinant Coagulation Factor VIII
14.18.1.2. Recombinant Coagulation Factor IX
14.18.1.3. Recombinant Coagulation Factor VIIa
14.18.1.4. Others
14.18.2. Human C1 Esterase Inhibitor
14.19. Rest of Middle East & Africa Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
14.19.1. Chinese Hamster Ovary (CHO) Cell Line
14.19.2. Baby Hamster Kidney (BHK) Cell Line
14.19.3. Human Embryonic Kidney (HEK) Cell Line
14.19.4. Others
14.20. Rest of Middle East & Africa Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
14.20.1. Hemophilia A
14.20.2. Hemophilia B
14.20.3. Von Willebrand Disease
14.20.4. Others
14.21. Middle East & Africa Recombinant Plasma Protein Therapeutics Market Attractiveness Analysis
14.21.1.1. By Drug Class
14.21.1.2. By Cell Line
14.21.1.3. By Indication
14.21.1.4. PEST Analysis
14.21.1.5. Key Trends
14.21.1.6. Key Developments
15. South America Recombinant Plasma Protein Therapeutics Market Analysis
15.1. Key Findings
15.2. South America Recombinant Plasma Protein Therapeutics Market Overview
15.3. South America Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Drug Class
15.4. South America Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
15.4.1. Recombinant Coagulation Factors
15.4.1.1. Recombinant Coagulation Factor VIII
15.4.1.2. Recombinant Coagulation Factor IX
15.4.1.3. Recombinant Coagulation Factor VIIa
15.4.1.4. Others
15.4.2. Human C1 Esterase Inhibitor
15.5. South America Recombinant Plasma Protein Therapeutics Market Value Share Analysis, By Cell Line
15.6. South America Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
15.6.1. Chinese Hamster Ovary (CHO) Cell Line
15.6.2. Baby Hamster Kidney (BHK) Cell Line
15.6.3. Human Embryonic Kidney (HEK) Cell Line
15.6.4. Others
15.7. South America Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Indication
15.8. South America Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
15.8.1. Hemophilia A
15.8.2. Hemophilia B
15.8.3. Von Willebrand Disease
15.8.4. Others
15.9. South America Recombinant Plasma Protein Therapeutics Market Value Share Analysis, by Country
15.10. South America Recombinant Plasma Protein Therapeutics Market Forecast, by Country
15.10.1. Brazil
15.10.2. Mexico
15.10.3. Rest of South America
15.11. South America Recombinant Plasma Protein Therapeutics Market Analysis, by Country
15.12. Brazil Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
15.12.1. Recombinant Coagulation Factors
15.12.1.1. Recombinant Coagulation Factor VIII
15.12.1.2. Recombinant Coagulation Factor IX
15.12.1.3. Recombinant Coagulation Factor VIIa
15.12.1.4. Others
15.12.2. Human C1 Esterase Inhibitor
15.13. Brazil Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
15.13.1. Chinese Hamster Ovary (CHO) Cell Line
15.13.2. Baby Hamster Kidney (BHK) Cell Line
15.13.3. Human Embryonic Kidney (HEK) Cell Line
15.13.4. Others
15.14. Brazil Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
15.14.1. Hemophilia A
15.14.2. Hemophilia B
15.14.3. Von Willebrand Disease
15.14.4. Others
15.15. Mexico Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
15.15.1. Recombinant Coagulation Factors
15.15.1.1. Recombinant Coagulation Factor VIII
15.15.1.2. Recombinant Coagulation Factor IX
15.15.1.3. Recombinant Coagulation Factor VIIa
15.15.1.4. Others
15.15.2. Human C1 Esterase Inhibitor
15.16. Mexico Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
15.16.1. Chinese Hamster Ovary (CHO) Cell Line
15.16.2. Baby Hamster Kidney (BHK) Cell Line
15.16.3. Human Embryonic Kidney (HEK) Cell Line
15.16.4. Others
15.17. Mexico Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
15.17.1. Hemophilia A
15.17.2. Hemophilia B
15.17.3. Von Willebrand Disease
15.17.4. Others
15.18. Rest of South America Recombinant Plasma Protein Therapeutics Market Forecast, by Drug Class
15.18.1. Recombinant Coagulation Factors
15.18.1.1. Recombinant Coagulation Factor VIII
15.18.1.2. Recombinant Coagulation Factor IX
15.18.1.3. Recombinant Coagulation Factor VIIa
15.18.1.4. Others
15.18.2. Human C1 Esterase Inhibitor
15.19. Rest of South America Recombinant Plasma Protein Therapeutics Market Forecast, by Cell Line
15.19.1. Chinese Hamster Ovary (CHO) Cell Line
15.19.2. Baby Hamster Kidney (BHK) Cell Line
15.19.3. Human Embryonic Kidney (HEK) Cell Line
15.19.4. Others
15.20. Rest of South America Recombinant Plasma Protein Therapeutics Market Forecast, by Indication
15.20.1. Hemophilia A
15.20.2. Hemophilia B
15.20.3. Von Willebrand Disease
15.20.4. Others
15.21. South America Recombinant Plasma Protein Therapeutics Market Attractiveness Analysis
15.21.1. By Drug Class
15.21.2. By Cell Line
15.21.3. By Indication
15.22. PEST Analysis
15.23. Key Trends
15.24. Key Developments
16. Company Profiles
16.1. Market Share Analysis, by Company
16.2. Competition Matrix
16.2.1. Competitive Benchmarking of key players by price, presence, market share, Applications and R&D investment
16.2.2. New Product Launches and Product Enhancements
16.2.3. Market Consolidation
16.2.3.1. M&A by Regions, Investment and Applications
16.2.3.2. M&A Key Players, Forward Integration and Backward Integration
16.3. Company Profiles: Key Players
16.3.1. CSL Limited
16.3.1.1. Company Overview
16.3.1.2. Financial Overview
16.3.1.3. Product Portfolio
16.3.1.4. Business Strategy
16.3.1.5. Recent Developments
16.3.1.6. Development Footprint
16.3.2. Shire Takeda Pharmaceutical Company Limited
16.3.3. Octapharma
16.3.4. Novo Nordisk A/S
16.3.5. Bayer AG
16.3.6. Bioverativ Therapeutics
16.3.7. Aptevo Therapeutics
16.3.8. Pharming Group NV
16.3.9. Pfizer Inc.
16.3.10. Ferring B.V.
16.3.11. Baxter
16.3.12. HEMA Biologics, LLC
16.3.13. OPKO Health, Inc
16.3.14. Sinocelltech Ltd
17. Primary Key Insights
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report