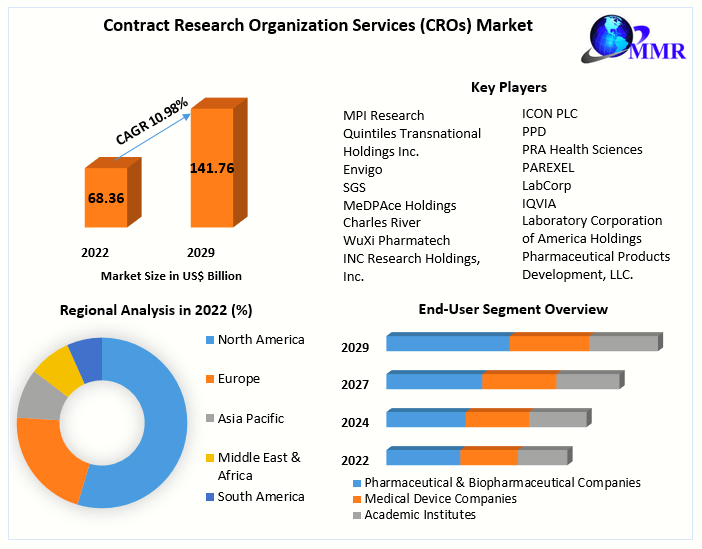
Global Contract Research Organization Services (CROs) Market
1. Preface
1.1. Report Scope and Market Segmentation
1.2. Research Highlights
1.3. Research Objectives
2. Assumptions and Research Methodology
2.1. Report Assumptions
2.2. Abbreviations
2.3. Research Methodology
2.3.1. Secondary Research
2.3.1.1. Secondary data
2.3.1.2. Secondary Sources
2.3.2. Primary Research
2.3.2.1. Data from Primary Sources
2.3.2.2. Breakdown of Primary Sources
3. Executive Summary: Global Contract Research Organization Services (CROs) Market Size, by Market Value (US$ Bn)
4. Market Overview
4.1. Introduction
4.2. Market Indicator
4.2.1. Drivers
4.2.2. Restraints
4.2.3. Opportunities
4.2.4. Challenges
4.3. Porter’s Analysis
4.4. Value Chain Analysis
4.5. Market Risk Analysis
4.6. SWOT Analysis
4.7. Industry Trends and Emerging Technologies
5. Supply Side and Demand Side Indicators
6. Global Contract Research Organization Services (CROs) Market Analysis and Forecast
6.1. Global Contract Research Organization Services (CROs) Market Size & Y-o-Y Growth Analysis
6.1.1. North America
6.1.2. Europe
6.1.3. Asia Pacific
6.1.4. Middle East & Africa
6.1.5. South America
7. Global Contract Research Organization Services (CROs) Market Analysis and Forecast, by Therapeutic Area
7.1. Introduction and Definition
7.2. Key Findings
7.3. Global Contract Research Organization Services (CROs) Market Value Share Analysis, by Therapeutic Area
7.4. Global Contract Research Organization Services (CROs) Market Size (US$ Bn) Forecast, by Therapeutic Area
7.5. Global Contract Research Organization Services (CROs) Market Analysis, by Therapeutic Area
7.6. Global Contract Research Organization Services (CROs) Market Attractiveness Analysis, by Therapeutic Area
8. Global Contract Research Organization Services (CROs) Market Analysis and Forecast, by End-user
8.1. Introduction and Definition
8.2. Key Findings
8.3. Global Contract Research Organization Services (CROs) Market Value Share Analysis, by End-user
8.4. Global Contract Research Organization Services (CROs) Market Size (US$ Bn) Forecast, by End-user
8.5. Global Contract Research Organization Services (CROs) Market Analysis, by End-user
8.6. Global Contract Research Organization Services (CROs) Market Attractiveness Analysis, by End-user
9. Global Contract Research Organization Services (CROs) Market Analysis and Forecast, by Type
9.1. Introduction and Definition
9.2. Key Findings
9.3. Global Contract Research Organization Services (CROs) Market Value Share Analysis, by Type
9.4. Global Contract Research Organization Services (CROs) Market Size (US$ Bn) Forecast, by Type
9.5. Global Contract Research Organization Services (CROs) Market Analysis, by Type
9.6. Global Contract Research Organization Services (CROs) Market Attractiveness Analysis, by Type
10. Global Contract Research Organization Services (CROs) Market Analysis, by Region
10.1. Global Contract Research Organization Services (CROs) Market Value Share Analysis, by Region
10.2. Global Contract Research Organization Services (CROs) Market Size (US$ Bn) Forecast, by Region
10.3. Global Contract Research Organization Services (CROs) Market Attractiveness Analysis, by Region
11. North America Contract Research Organization Services (CROs) Market Analysis
11.1. Key Findings
11.2. North America Contract Research Organization Services (CROs) Market Overview
11.3. North America Contract Research Organization Services (CROs) Market Value Share Analysis, by Therapeutic Area
11.4. North America Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
11.4.1. Oncology
11.4.2. Infectious Diseases
11.4.3. Central Nervous System (CNS) Disorders
11.4.4. Immunological Disorders
11.4.5. Cardiovascular Diseases
11.4.6. Respiratory Disorders
11.4.7. Diabetes
11.4.8. Other Therapeutic Areas
11.5. North America Contract Research Organization Services (CROs) Market Value Share Analysis, by End-user
11.6. North America Contract Research Organization Services (CROs) Market Forecast, by End-user
11.6.1. Pharmaceutical & Biopharmaceutical Companies
11.6.2. Medical Device Companies
11.6.3. Academic Institutes
11.7. North America Contract Research Organization Services (CROs) Market Value Share Analysis, by Type
11.8. North America Contract Research Organization Services (CROs) Market Forecast, by Type
11.8.1. Early Phase Development Services
11.8.1.1. Discovery Studies
11.8.1.2. Chemistry, Manufacturing & Control
11.8.1.3. Preclinical Services
11.8.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
11.8.3. Toxicology Testing
11.8.4. Clinic Research Services
11.8.4.1. Phase I
11.8.4.2. Phase II
11.8.4.3. Phase III
11.8.4.4. Phase IV
11.8.5. Laboratory Services
11.8.5.1. Bioanalytical Testing
11.8.5.2. Analytical Testing
11.8.6. Physical Characterization
11.8.7. Stability Testing
11.8.8. Batch Release Testing
11.8.9. Raw Material Testing
11.8.10. Other Analytical Testing
11.8.11. Consulting Services
11.8.12. Other
11.9. North America Contract Research Organization Services (CROs) Market Value Share Analysis, by Country
11.10. North America Contract Research Organization Services (CROs) Market Forecast, by Country
11.10.1. U.S.
11.10.2. Canada
11.11. North America Contract Research Organization Services (CROs) Market Analysis, by Country
11.12. U.S. Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
11.12.1. Oncology
11.12.2. Infectious Diseases
11.12.3. Central Nervous System (CNS) Disorders
11.12.4. Immunological Disorders
11.12.5. Cardiovascular Diseases
11.12.6. Respiratory Disorders
11.12.7. Diabetes
11.12.8. Other
11.13. U.S. Contract Research Organization Services (CROs) Market Forecast, by End-user
11.13.1. Pharmaceutical & Biopharmaceutical Companies
11.13.2. Medical Device Companies
11.13.3. Academic Institutes
11.14. U.S. Contract Research Organization Services (CROs) Market Forecast, by Type
11.14.1. Early Phase Development Services
11.14.1.1. Discovery Studies
11.14.1.2. Chemistry, Manufacturing & Control
11.14.1.3. Preclinical Services
11.14.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
11.14.3. Toxicology Testing
11.14.4. Clinic Research Services
11.14.4.1. Phase I
11.14.4.2. Phase II
11.14.4.3. Phase III
11.14.4.4. Phase IV
11.14.5. Laboratory Services
11.14.5.1. Bioanalytical Testing
11.14.5.2. Analytical Testing
11.14.6. Physical Characterization
11.14.7. Stability Testing
11.14.8. Batch Release Testing
11.14.9. Raw Material Testing
11.14.10. Other Analytical Testing
11.14.11. Consulting Services
11.14.12. Other
11.15. Canada Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
11.15.1. Oncology
11.15.2. Infectious Diseases
11.15.3. Central Nervous System (CNS) Disorders
11.15.4. Immunological Disorders
11.15.5. Cardiovascular Diseases
11.15.6. Respiratory Disorders
11.15.7. Diabetes
11.15.8. Other
11.16. Canada Contract Research Organization Services (CROs) Market Forecast, by End-user
11.16.1. Pharmaceutical & Biopharmaceutical Companies
11.16.2. Medical Device Companies
11.16.3. Academic Institutes
11.17. Canada Contract Research Organization Services (CROs) Market Forecast, by Type
11.17.1. Early Phase Development Services
11.17.1.1. Discovery Studies
11.17.1.2. Chemistry, Manufacturing & Control
11.17.1.3. Preclinical Services
11.17.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
11.17.3. Toxicology Testing
11.17.4. Clinic Research Services
11.17.4.1. Phase I
11.17.4.2. Phase II
11.17.4.3. Phase III
11.17.4.4. Phase IV
11.17.5. Laboratory Services
11.17.5.1. Bioanalytical Testing
11.17.5.2. Analytical Testing
11.17.6. Physical Characterization
11.17.7. Stability Testing
11.17.8. Batch Release Testing
11.17.9. Raw Material Testing
11.17.10. Other Analytical Testing
11.17.11. Consulting Services
11.17.12. Other
11.18. North America Contract Research Organization Services (CROs) Market Attractiveness Analysis
11.18.1. By Therapeutic Area
11.18.2. By End-user
11.18.3. By Type
11.19. PEST Analysis
11.20. Key Trends
11.21. Key Developments
12. Europe Contract Research Organization Services (CROs) Market Analysis
12.1. Key Findings
12.2. Europe Contract Research Organization Services (CROs) Market Overview
12.3. Europe Contract Research Organization Services (CROs) Market Value Share Analysis, by Therapeutic Area
12.4. Europe Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
12.4.1. Oncology
12.4.2. Infectious Diseases
12.4.3. Central Nervous System (CNS) Disorders
12.4.4. Immunological Disorders
12.4.5. Cardiovascular Diseases
12.4.6. Respiratory Disorders
12.4.7. Diabetes
12.4.8. Other
12.5. Europe Contract Research Organization Services (CROs) Market Value Share Analysis, by End-user
12.6. Europe Contract Research Organization Services (CROs) Market Forecast, by End-user
12.6.1. Pharmaceutical & Biopharmaceutical Companies
12.6.2. Medical Device Companies
12.6.3. Academic Institutes
12.7. Europe Contract Research Organization Services (CROs) Market Value Share Analysis, by Type
12.8. Europe Contract Research Organization Services (CROs) Market Forecast, by Type
12.8.1. Early Phase Development Services
12.8.1.1. Discovery Studies
12.8.1.2. Chemistry, Manufacturing & Control
12.8.1.3. Preclinical Services
12.8.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
12.8.3. Toxicology Testing
12.8.4. Clinic Research Services
12.8.4.1. Phase I
12.8.4.2. Phase II
12.8.4.3. Phase III
12.8.4.4. Phase IV
12.8.5. Laboratory Services
12.8.5.1. Bioanalytical Testing
12.8.5.2. Analytical Testing
12.8.6. Physical Characterization
12.8.7. Stability Testing
12.8.8. Batch Release Testing
12.8.9. Raw Material Testing
12.8.10. Other Analytical Testing
12.8.11. Consulting Services
12.8.12. Other
12.9. Europe Contract Research Organization Services (CROs) Market Value Share Analysis, by Country
12.10. Europe Contract Research Organization Services (CROs) Market Forecast, by Country
12.10.1. Germany
12.10.2. U.K.
12.10.3. France
12.10.4. Italy
12.10.5. Spain
12.10.6. Rest of Europe
12.11. Europe Contract Research Organization Services (CROs) Market Analysis, by Country
12.12. Germany Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
12.12.1. Oncology
12.12.2. Infectious Diseases
12.12.3. Central Nervous System (CNS) Disorders
12.12.4. Immunological Disorders
12.12.5. Cardiovascular Diseases
12.12.6. Respiratory Disorders
12.12.7. Diabetes
12.12.8. Other
12.13. Germany Contract Research Organization Services (CROs) Market Forecast, by End-user
12.13.1. Pharmaceutical & Biopharmaceutical Companies
12.13.2. Medical Device Companies
12.13.3. Academic Institutes
12.14. Germany Contract Research Organization Services (CROs) Market Forecast, by Type
12.14.1. Early Phase Development Services
12.14.1.1. Discovery Studies
12.14.1.2. Chemistry, Manufacturing & Control
12.14.1.3. Preclinical Services
12.14.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
12.14.3. Toxicology Testing
12.14.4. Clinic Research Services
12.14.4.1. Phase I
12.14.4.2. Phase II
12.14.4.3. Phase III
12.14.4.4. Phase IV
12.14.5. Laboratory Services
12.14.5.1. Bioanalytical Testing
12.14.5.2. Analytical Testing
12.14.6. Physical Characterization
12.14.7. Stability Testing
12.14.8. Batch Release Testing
12.14.9. Raw Material Testing
12.14.10. Other Analytical Testing
12.14.11. Consulting Services
12.14.12. Other
12.15. U.K. Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
12.15.1. Oncology
12.15.2. Infectious Diseases
12.15.3. Central Nervous System (CNS) Disorders
12.15.4. Immunological Disorders
12.15.5. Cardiovascular Diseases
12.15.6. Respiratory Disorders
12.15.7. Diabetes
12.15.8. Other
12.16. U.K. Contract Research Organization Services (CROs) Market Forecast, by End-user
12.16.1. Pharmaceutical & Biopharmaceutical Companies
12.16.2. Medical Device Companies
12.16.3. Academic Institutes
12.17. U.K. Contract Research Organization Services (CROs) Market Forecast, by Type
12.17.1. Early Phase Development Services
12.17.1.1. Discovery Studies
12.17.1.2. Chemistry, Manufacturing & Control
12.17.1.3. Preclinical Services
12.17.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
12.17.3. Toxicology Testing
12.17.4. Clinic Research Services
12.17.4.1. Phase I
12.17.4.2. Phase II
12.17.4.3. Phase III
12.17.4.4. Phase IV
12.17.5. Laboratory Services
12.17.5.1. Bioanalytical Testing
12.17.5.2. Analytical Testing
12.17.6. Physical Characterization
12.17.7. Stability Testing
12.17.8. Batch Release Testing
12.17.9. Raw Material Testing
12.17.10. Other Analytical Testing
12.17.11. Consulting Services
12.17.12. Other
12.18. France Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
12.18.1. Oncology
12.18.2. Infectious Diseases
12.18.3. Central Nervous System (CNS) Disorders
12.18.4. Immunological Disorders
12.18.5. Cardiovascular Diseases
12.18.6. Respiratory Disorders
12.18.7. Diabetes
12.18.8. Other
12.19. France Contract Research Organization Services (CROs) Market Forecast, by End-user
12.19.1. Pharmaceutical & Biopharmaceutical Companies
12.19.2. Medical Device Companies
12.19.3. Academic Institutes
12.20. France Contract Research Organization Services (CROs) Market Forecast, by Type
12.20.1. Early Phase Development Services
12.20.1.1. Discovery Studies
12.20.1.2. Chemistry, Manufacturing & Control
12.20.1.3. Preclinical Services
12.20.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
12.20.3. Toxicology Testing
12.20.4. Clinic Research Services
12.20.4.1. Phase I
12.20.4.2. Phase II
12.20.4.3. Phase III
12.20.4.4. Phase IV
12.20.5. Laboratory Services
12.20.5.1. Bioanalytical Testing
12.20.5.2. Analytical Testing
12.20.6. Physical Characterization
12.20.7. Stability Testing
12.20.8. Batch Release Testing
12.20.9. Raw Material Testing
12.20.10. Other Analytical Testing
12.20.11. Consulting Services
12.20.12. Other
12.21. Italy Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
12.21.1. Oncology
12.21.2. Infectious Diseases
12.21.3. Central Nervous System (CNS) Disorders
12.21.4. Immunological Disorders
12.21.5. Cardiovascular Diseases
12.21.6. Respiratory Disorders
12.21.7. Diabetes
12.21.8. Other
12.22. Italy Contract Research Organization Services (CROs) Market Forecast, by End-user
12.22.1. Pharmaceutical & Biopharmaceutical Companies
12.22.2. Medical Device Companies
12.22.3. Academic Institutes
12.23. Italy Contract Research Organization Services (CROs) Market Forecast, by Type
12.23.1. Early Phase Development Services
12.23.1.1. Discovery Studies
12.23.1.2. Chemistry, Manufacturing & Control
12.23.1.3. Preclinical Services
12.23.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
12.23.3. Toxicology Testing
12.23.4. Clinic Research Services
12.23.4.1. Phase I
12.23.4.2. Phase II
12.23.4.3. Phase III
12.23.4.4. Phase IV
12.23.5. Laboratory Services
12.23.5.1. Bioanalytical Testing
12.23.5.2. Analytical Testing
12.23.6. Physical Characterization
12.23.7. Stability Testing
12.23.8. Batch Release Testing
12.23.9. Raw Material Testing
12.23.10. Other Analytical Testing
12.23.11. Consulting Services
12.23.12. Other
12.24. Spain Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
12.24.1. Oncology
12.24.2. Infectious Diseases
12.24.3. Central Nervous System (CNS) Disorders
12.24.4. Immunological Disorders
12.24.5. Cardiovascular Diseases
12.24.6. Respiratory Disorders
12.24.7. Diabetes
12.24.8. Other
12.25. Spain Contract Research Organization Services (CROs) Market Forecast, by End-user
12.25.1. Pharmaceutical & Biopharmaceutical Companies
12.25.2. Medical Device Companies
12.25.3. Academic Institutes
12.26. Spain Contract Research Organization Services (CROs) Market Forecast, by Type
12.26.1. Early Phase Development Services
12.26.1.1. Discovery Studies
12.26.1.2. Chemistry, Manufacturing & Control
12.26.1.3. Preclinical Services
12.26.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
12.26.3. Toxicology Testing
12.26.4. Clinic Research Services
12.26.4.1. Phase I
12.26.4.2. Phase II
12.26.4.3. Phase III
12.26.4.4. Phase IV
12.26.5. Laboratory Services
12.26.5.1. Bioanalytical Testing
12.26.5.2. Analytical Testing
12.26.6. Physical Characterization
12.26.7. Stability Testing
12.26.8. Batch Release Testing
12.26.9. Raw Material Testing
12.26.10. Other Analytical Testing
12.26.11. Consulting Services
12.26.12. Other
12.27. Rest of Europe Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
12.27.1. Oncology
12.27.2. Infectious Diseases
12.27.3. Central Nervous System (CNS) Disorders
12.27.4. Immunological Disorders
12.27.5. Cardiovascular Diseases
12.27.6. Respiratory Disorders
12.27.7. Diabetes
12.27.8. Other
12.28. Rest of Europe Contract Research Organization Services (CROs) Market Forecast, by End-user
12.28.1. Pharmaceutical & Biopharmaceutical Companies
12.28.2. Medical Device Companies
12.28.3. Academic Institutes
12.29. Rest Of Europe Contract Research Organization Services (CROs) Market Forecast, by Type
12.29.1. Early Phase Development Services
12.29.1.1. Discovery Studies
12.29.1.2. Chemistry, Manufacturing & Control
12.29.1.3. Preclinical Services
12.29.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
12.29.3. Toxicology Testing
12.29.4. Clinic Research Services
12.29.4.1. Phase I
12.29.4.2. Phase II
12.29.4.3. Phase III
12.29.4.4. Phase IV
12.29.5. Laboratory Services
12.29.5.1. Bioanalytical Testing
12.29.5.2. Analytical Testing
12.29.6. Physical Characterization
12.29.7. Stability Testing
12.29.8. Batch Release Testing
12.29.9. Raw Material Testing
12.29.10. Other Analytical Testing
12.29.11. Consulting Services
12.29.12. Other
12.30. Europe Contract Research Organization Services (CROs) Market Attractiveness Analysis
12.30.1. By Therapeutic Area
12.30.2. By End-user
12.30.3. By Type
12.31. PEST Analysis
12.32. Key Trends
12.33. Key Developments
13. Asia Pacific Contract Research Organization Services (CROs) Market Analysis
13.1. Key Findings
13.2. Asia Pacific Contract Research Organization Services (CROs) Market Overview
13.3. Asia Pacific Contract Research Organization Services (CROs) Market Value Share Analysis, by Therapeutic Area
13.4. Asia Pacific Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
13.4.1. Oncology
13.4.2. Infectious Diseases
13.4.3. Central Nervous System (CNS) Disorders
13.4.4. Immunological Disorders
13.4.5. Cardiovascular Diseases
13.4.6. Respiratory Disorders
13.4.7. Diabetes
13.4.8. Other
13.5. Asia Pacific Contract Research Organization Services (CROs) Market Value Share Analysis, by End-user
13.6. Asia Pacific Contract Research Organization Services (CROs) Market Forecast, by End-user
13.6.1. Pharmaceutical & Biopharmaceutical Companies
13.6.2. Medical Device Companies
13.6.3. Academic Institutes
13.7. Asia Pacific Contract Research Organization Services (CROs) Market Value Share Analysis, by Type
13.8. Asia Pacific Contract Research Organization Services (CROs) Market Forecast, by Type
13.8.1. Early Phase Development Services
13.8.1.1. Discovery Studies
13.8.1.2. Chemistry, Manufacturing & Control
13.8.1.3. Preclinical Services
13.8.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
13.8.3. Toxicology Testing
13.8.4. Clinic Research Services
13.8.4.1. Phase I
13.8.4.2. Phase II
13.8.4.3. Phase III
13.8.4.4. Phase IV
13.8.5. Laboratory Services
13.8.5.1. Bioanalytical Testing
13.8.5.2. Analytical Testing
13.8.6. Physical Characterization
13.8.7. Stability Testing
13.8.8. Batch Release Testing
13.8.9. Raw Material Testing
13.8.10. Other Analytical Testing
13.8.11. Consulting Services
13.8.12. Other
13.9. Asia Pacific Contract Research Organization Services (CROs) Market Value Share Analysis, by Country
13.10. Asia Pacific Contract Research Organization Services (CROs) Market Forecast, by Country
13.10.1. China
13.10.2. India
13.10.3. Japan
13.10.4. ASEAN
13.10.5. Rest of Asia Pacific
13.11. Asia Pacific Contract Research Organization Services (CROs) Market Analysis, by Country
13.12. China Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
13.12.1. Oncology
13.12.2. Infectious Diseases
13.12.3. Central Nervous System (CNS) Disorders
13.12.4. Immunological Disorders
13.12.5. Cardiovascular Diseases
13.12.6. Respiratory Disorders
13.12.7. Diabetes
13.12.8. Other
13.13. China Contract Research Organization Services (CROs) Market Forecast, by End-user
13.13.1. Pharmaceutical & Biopharmaceutical Companies
13.13.2. Medical Device Companies
13.13.3. Academic Institutes
13.14. China Contract Research Organization Services (CROs) Market Forecast, by Type
13.14.1. Early Phase Development Services
13.14.1.1. Discovery Studies
13.14.1.2. Chemistry, Manufacturing & Control
13.14.1.3. Preclinical Services
13.14.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
13.14.3. Toxicology Testing
13.14.4. Clinic Research Services
13.14.4.1. Phase I
13.14.4.2. Phase II
13.14.4.3. Phase III
13.14.4.4. Phase IV
13.14.5. Laboratory Services
13.14.5.1. Bioanalytical Testing
13.14.5.2. Analytical Testing
13.14.6. Physical Characterization
13.14.7. Stability Testing
13.14.8. Batch Release Testing
13.14.9. Raw Material Testing
13.14.10. Other Analytical Testing
13.14.11. Consulting Services
13.14.12. Other
13.15. India Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
13.15.1. Oncology
13.15.2. Infectious Diseases
13.15.3. Central Nervous System (CNS) Disorders
13.15.4. Immunological Disorders
13.15.5. Cardiovascular Diseases
13.15.6. Respiratory Disorders
13.15.7. Diabetes
13.15.8. Other
13.16. India Contract Research Organization Services (CROs) Market Forecast, by End-user
13.16.1. Pharmaceutical & Biopharmaceutical Companies
13.16.2. Medical Device Companies
13.16.3. Academic Institutes
13.17. India Contract Research Organization Services (CROs) Market Forecast, by Type
13.17.1. Early Phase Development Services
13.17.1.1. Discovery Studies
13.17.1.2. Chemistry, Manufacturing & Control
13.17.1.3. Preclinical Services
13.17.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
13.17.3. Toxicology Testing
13.17.4. Clinic Research Services
13.17.4.1. Phase I
13.17.4.2. Phase II
13.17.4.3. Phase III
13.17.4.4. Phase IV
13.17.5. Laboratory Services
13.17.5.1. Bioanalytical Testing
13.17.5.2. Analytical Testing
13.17.6. Physical Characterization
13.17.7. Stability Testing
13.17.8. Batch Release Testing
13.17.9. Raw Material Testing
13.17.10. Other Analytical Testing
13.17.11. Consulting Services
13.17.12. Other
13.18. Japan Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
13.18.1. Oncology
13.18.2. Infectious Diseases
13.18.3. Central Nervous System (CNS) Disorders
13.18.4. Immunological Disorders
13.18.5. Cardiovascular Diseases
13.18.6. Respiratory Disorders
13.18.7. Diabetes
13.18.8. Other
13.19. Japan Contract Research Organization Services (CROs) Market Forecast, by End-user
13.19.1. Pharmaceutical & Biopharmaceutical Companies
13.19.2. Medical Device Companies
13.19.3. Academic Institutes
13.20. Japan Contract Research Organization Services (CROs) Market Forecast, by Type
13.20.1. Early Phase Development Services
13.20.1.1. Discovery Studies
13.20.1.2. Chemistry, Manufacturing & Control
13.20.1.3. Preclinical Services
13.20.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
13.20.3. Toxicology Testing
13.20.4. Clinic Research Services
13.20.4.1. Phase I
13.20.4.2. Phase II
13.20.4.3. Phase III
13.20.4.4. Phase IV
13.20.5. Laboratory Services
13.20.5.1. Bioanalytical Testing
13.20.5.2. Analytical Testing
13.20.6. Physical Characterization
13.20.7. Stability Testing
13.20.8. Batch Release Testing
13.20.9. Raw Material Testing
13.20.10. Other Analytical Testing
13.20.11. Consulting Services
13.20.12. Other
13.21. ASEAN Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
13.21.1. Oncology
13.21.2. Infectious Diseases
13.21.3. Central Nervous System (CNS) Disorders
13.21.4. Immunological Disorders
13.21.5. Cardiovascular Diseases
13.21.6. Respiratory Disorders
13.21.7. Diabetes
13.21.8. Other
13.22. ASEAN Contract Research Organization Services (CROs) Market Forecast, by End-user
13.22.1. Pharmaceutical & Biopharmaceutical Companies
13.22.2. Medical Device Companies
13.22.3. Academic Institutes
13.23. ASEAN Contract Research Organization Services (CROs) Market Forecast, by Type
13.23.1. Early Phase Development Services
13.23.1.1. Discovery Studies
13.23.1.2. Chemistry, Manufacturing & Control
13.23.1.3. Preclinical Services
13.23.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
13.23.3. Toxicology Testing
13.23.4. Clinic Research Services
13.23.4.1. Phase I
13.23.4.2. Phase II
13.23.4.3. Phase III
13.23.4.4. Phase IV
13.23.5. Laboratory Services
13.23.5.1. Bioanalytical Testing
13.23.5.2. Analytical Testing
13.23.6. Physical Characterization
13.23.7. Stability Testing
13.23.8. Batch Release Testing
13.23.9. Raw Material Testing
13.23.10. Other Analytical Testing
13.23.11. Consulting Services
13.23.12. Other
13.24. Rest of Asia Pacific Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
13.24.1. Oncology
13.24.2. Infectious Diseases
13.24.3. Central Nervous System (CNS) Disorders
13.24.4. Immunological Disorders
13.24.5. Cardiovascular Diseases
13.24.6. Respiratory Disorders
13.24.7. Diabetes
13.24.8. Other
13.25. Rest of Asia Pacific Contract Research Organization Services (CROs) Market Forecast, by End-user
13.25.1. Pharmaceutical & Biopharmaceutical Companies
13.25.2. Medical Device Companies
13.25.3. Academic Institutes
13.26. Rest of Asia Pacific Contract Research Organization Services (CROs) Market Forecast, by Type
13.26.1. Early Phase Development Services
13.26.1.1. Discovery Studies
13.26.1.2. Chemistry, Manufacturing & Control
13.26.1.3. Preclinical Services
13.26.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
13.26.3. Toxicology Testing
13.26.4. Clinic Research Services
13.26.4.1. Phase I
13.26.4.2. Phase II
13.26.4.3. Phase III
13.26.4.4. Phase IV
13.26.5. Laboratory Services
13.26.5.1. Bioanalytical Testing
13.26.5.2. Analytical Testing
13.26.6. Physical Characterization
13.26.7. Stability Testing
13.26.8. Batch Release Testing
13.26.9. Raw Material Testing
13.26.10. Other Analytical Testing
13.26.11. Consulting Services
13.26.12. Other
13.27. Asia Pacific Contract Research Organization Services (CROs) Market Attractiveness Analysis
13.27.1. By Therapeutic Area
13.27.2. By End-user
13.27.3. By Type
13.28. PEST Analysis
13.29. Key Trends
13.30. Key Developments
14. Middle East & Africa Contract Research Organization Services (CROs) Market Analysis
14.1. Key Findings
14.2. Middle East & Africa Contract Research Organization Services (CROs) Market Overview
14.3. Middle East & Africa Contract Research Organization Services (CROs) Market Value Share Analysis, by Therapeutic Area
14.4. Middle East & Africa Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
14.4.1. Oncology
14.4.2. Infectious Diseases
14.4.3. Central Nervous System (CNS) Disorders
14.4.4. Immunological Disorders
14.4.5. Cardiovascular Diseases
14.4.6. Respiratory Disorders
14.4.7. Diabetes
14.4.8. Other
14.5. Middle East & Africa Contract Research Organization Services (CROs) Market Value Share Analysis, by End-user
14.6. Middle East & Africa Contract Research Organization Services (CROs) Market Forecast, by End-user
14.6.1. Pharmaceutical & Biopharmaceutical Companies
14.6.2. Medical Device Companies
14.6.3. Academic Institutes
14.7. Middle East & Africa Contract Research Organization Services (CROs) Market Value Share Analysis, by Type
14.8. Middle East & Africa Contract Research Organization Services (CROs) Market Forecast, by Type
14.8.1. Early Phase Development Services
14.8.1.1. Discovery Studies
14.8.1.2. Chemistry, Manufacturing & Control
14.8.1.3. Preclinical Services
14.8.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
14.8.3. Toxicology Testing
14.8.4. Clinic Research Services
14.8.4.1. Phase I
14.8.4.2. Phase II
14.8.4.3. Phase III
14.8.4.4. Phase IV
14.8.5. Laboratory Services
14.8.5.1. Bioanalytical Testing
14.8.5.2. Analytical Testing
14.8.6. Physical Characterization
14.8.7. Stability Testing
14.8.8. Batch Release Testing
14.8.9. Raw Material Testing
14.8.10. Other Analytical Testing
14.8.11. Consulting Services
14.8.12. Other
14.9. Middle East & Africa Contract Research Organization Services (CROs) Market Value Share Analysis, by Country
14.10. Middle East & Africa Contract Research Organization Services (CROs) Market Forecast, by Country
14.10.1. GCC
14.10.2. South Africa
14.10.3. Rest of Middle East & Africa
14.11. Middle East & Africa Contract Research Organization Services (CROs) Market Analysis, by Country
14.12. GCC Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
14.12.1. Oncology
14.12.2. Infectious Diseases
14.12.3. Central Nervous System (CNS) Disorders
14.12.4. Immunological Disorders
14.12.5. Cardiovascular Diseases
14.12.6. Respiratory Disorders
14.12.7. Diabetes
14.12.8. Other
14.13. GCC Contract Research Organization Services (CROs) Market Forecast, by End-user
14.13.1. Pharmaceutical & Biopharmaceutical Companies
14.13.2. Medical Device Companies
14.13.3. Academic Institutes
14.14. GCC Contract Research Organization Services (CROs) Market Forecast, by Type
14.14.1. Early Phase Development Services
14.14.1.1. Discovery Studies
14.14.1.2. Chemistry, Manufacturing & Control
14.14.1.3. Preclinical Services
14.14.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
14.14.3. Toxicology Testing
14.14.4. Clinic Research Services
14.14.4.1. Phase I
14.14.4.2. Phase II
14.14.4.3. Phase III
14.14.4.4. Phase IV
14.14.5. Laboratory Services
14.14.5.1. Bioanalytical Testing
14.14.5.2. Analytical Testing
14.14.6. Physical Characterization
14.14.7. Stability Testing
14.14.8. Batch Release Testing
14.14.9. Raw Material Testing
14.14.10. Other Analytical Testing
14.14.11. Consulting Services
14.14.12. Other
14.15. South Africa Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
14.15.1. Oncology
14.15.2. Infectious Diseases
14.15.3. Central Nervous System (CNS) Disorders
14.15.4. Immunological Disorders
14.15.5. Cardiovascular Diseases
14.15.6. Respiratory Disorders
14.15.7. Diabetes
14.15.8. Other
14.16. South Africa Contract Research Organization Services (CROs) Market Forecast, by End-user
14.16.1. Pharmaceutical & Biopharmaceutical Companies
14.16.2. Medical Device Companies
14.16.3. Academic Institutes
14.17. South Africa Contract Research Organization Services (CROs) Market Forecast, by Type
14.17.1. Early Phase Development Services
14.17.1.1. Discovery Studies
14.17.1.2. Chemistry, Manufacturing & Control
14.17.1.3. Preclinical Services
14.17.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
14.17.3. Toxicology Testing
14.17.4. Clinic Research Services
14.17.4.1. Phase I
14.17.4.2. Phase II
14.17.4.3. Phase III
14.17.4.4. Phase IV
14.17.5. Laboratory Services
14.17.5.1. Bioanalytical Testing
14.17.5.2. Analytical Testing
14.17.6. Physical Characterization
14.17.7. Stability Testing
14.17.8. Batch Release Testing
14.17.9. Raw Material Testing
14.17.10. Other Analytical Testing
14.17.11. Consulting Services
14.17.12. Other
14.18. Rest of Middle East & Africa Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
14.18.1. Oncology
14.18.2. Infectious Diseases
14.18.3. Central Nervous System (CNS) Disorders
14.18.4. Immunological Disorders
14.18.5. Cardiovascular Diseases
14.18.6. Respiratory Disorders
14.18.7. Diabetes
14.18.8. Other
14.19. Rest of Middle East & Africa Contract Research Organization Services (CROs) Market Forecast, by End-user
14.19.1. Contract Research Organizations
14.19.2. Diagnostic Laboratories
14.19.3. Hospitals
14.19.4. Academic Institute
14.20. Rest of Middle East & Africa Contract Research Organization Services (CROs) Market Forecast, by Type
14.20.1. Early Phase Development Services
14.20.1.1. Discovery Studies
14.20.1.2. Chemistry, Manufacturing & Control
14.20.1.3. Preclinical Services
14.20.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
14.20.3. Toxicology Testing
14.20.4. Clinic Research Services
14.20.4.1. Phase I
14.20.4.2. Phase II
14.20.4.3. Phase III
14.20.4.4. Phase IV
14.20.5. Laboratory Services
14.20.5.1. Bioanalytical Testing
14.20.5.2. Analytical Testing
14.20.6. Physical Characterization
14.20.7. Stability Testing
14.20.8. Batch Release Testing
14.20.9. Raw Material Testing
14.20.10. Other Analytical Testing
14.20.11. Consulting Services
14.20.12. Other
14.21. Middle East & Africa Contract Research Organization Services (CROs) Market Attractiveness Analysis
14.21.1. By Therapeutic Area
14.21.2. By End-user
14.21.3. By Type
14.22. PEST Analysis
14.23. Key Trends
14.24. Key Developments
15. South America Contract Research Organization Services (CROs) Market Analysis
15.1. Key Findings
15.2. South America Contract Research Organization Services (CROs) Market Overview
15.3. South America Contract Research Organization Services (CROs) Market Value Share Analysis, by Therapeutic Area
15.4. South America Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
15.4.1. Oncology
15.4.2. Infectious Diseases
15.4.3. Central Nervous System (CNS) Disorders
15.4.4. Immunological Disorders
15.4.5. Cardiovascular Diseases
15.4.6. Respiratory Disorders
15.4.7. Diabetes
15.4.8. Other
15.5. South America Contract Research Organization Services (CROs) Market Value Share Analysis, by End-user
15.6. South America Contract Research Organization Services (CROs) Market Forecast, by End-user
15.6.1. Pharmaceutical & Biopharmaceutical Companies
15.6.2. Medical Device Companies
15.6.3. Academic Institutes
15.7. South America Contract Research Organization Services (CROs) Market Value Share Analysis, by Type
15.8. South America Contract Research Organization Services (CROs) Market Forecast, by Type
15.8.1. Early Phase Development Services
15.8.1.1. Discovery Studies
15.8.1.2. Chemistry, Manufacturing & Control
15.8.1.3. Preclinical Services
15.8.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
15.8.3. Toxicology Testing
15.8.4. Clinic Research Services
15.8.4.1. Phase I
15.8.4.2. Phase II
15.8.4.3. Phase III
15.8.4.4. Phase IV
15.8.5. Laboratory Services
15.8.5.1. Bioanalytical Testing
15.8.5.2. Analytical Testing
15.8.6. Physical Characterization
15.8.7. Stability Testing
15.8.8. Batch Release Testing
15.8.9. Raw Material Testing
15.8.10. Other Analytical Testing
15.8.11. Consulting Services
15.8.12. Other
15.9. South America Contract Research Organization Services (CROs) Market Value Share Analysis, by Country
15.10. South America Contract Research Organization Services (CROs) Market Forecast, by Country
15.10.1. Brazil
15.10.2. Mexico
15.10.3. Rest of South America
15.11. South America Contract Research Organization Services (CROs) Market Analysis, by Country
15.12. Brazil Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
15.12.1. Oncology
15.12.2. Infectious Diseases
15.12.3. Central Nervous System (CNS) Disorders
15.12.4. Immunological Disorders
15.12.5. Cardiovascular Diseases
15.12.6. Respiratory Disorders
15.12.7. Diabetes
15.12.8. Other
15.13. Brazil Contract Research Organization Services (CROs) Market Forecast, by End-user
15.13.1. Pharmaceutical & Biopharmaceutical Companies
15.13.2. Medical Device Companies
15.13.3. Academic Institutes
15.14. Brazil Contract Research Organization Services (CROs) Market Forecast, by Type
15.14.1. Early Phase Development Services
15.14.1.1. Discovery Studies
15.14.1.2. Chemistry, Manufacturing & Control
15.14.1.3. Preclinical Services
15.14.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
15.14.3. Toxicology Testing
15.14.4. Clinic Research Services
15.14.4.1. Phase I
15.14.4.2. Phase II
15.14.4.3. Phase III
15.14.4.4. Phase IV
15.14.5. Laboratory Services
15.14.5.1. Bioanalytical Testing
15.14.5.2. Analytical Testing
15.14.6. Physical Characterization
15.14.7. Stability Testing
15.14.8. Batch Release Testing
15.14.9. Raw Material Testing
15.14.10. Other Analytical Testing
15.14.11. Consulting Services
15.14.12. Other
15.15. Mexico Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
15.15.1. Oncology
15.15.2. Infectious Diseases
15.15.3. Central Nervous System (CNS) Disorders
15.15.4. Immunological Disorders
15.15.5. Cardiovascular Diseases
15.15.6. Respiratory Disorders
15.15.7. Diabetes
15.15.8. Other
15.16. Mexico Contract Research Organization Services (CROs) Market Forecast, by End-user
15.16.1. Pharmaceutical & Biopharmaceutical Companies
15.16.2. Medical Device Companies
15.16.3. Academic Institutes
15.17. Mexico Contract Research Organization Services (CROs) Market Forecast, by Type
15.17.1. Early Phase Development Services
15.17.1.1. Discovery Studies
15.17.1.2. Chemistry, Manufacturing & Control
15.17.1.3. Preclinical Services
15.17.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
15.17.3. Toxicology Testing
15.17.4. Clinic Research Services
15.17.4.1. Phase I
15.17.4.2. Phase II
15.17.4.3. Phase III
15.17.4.4. Phase IV
15.17.5. Laboratory Services
15.17.5.1. Bioanalytical Testing
15.17.5.2. Analytical Testing
15.17.6. Physical Characterization
15.17.7. Stability Testing
15.17.8. Batch Release Testing
15.17.9. Raw Material Testing
15.17.10. Other Analytical Testing
15.17.11. Consulting Services
15.17.12. Other
15.18. Rest of South America Contract Research Organization Services (CROs) Market Forecast, by Therapeutic Area
15.18.1. Oncology
15.18.2. Infectious Diseases
15.18.3. Central Nervous System (CNS) Disorders
15.18.4. Immunological Disorders
15.18.5. Cardiovascular Diseases
15.18.6. Respiratory Disorders
15.18.7. Diabetes
15.18.8. Other
15.19. Rest of South America Contract Research Organization Services (CROs) Market Forecast, by End-user
15.19.1. Pharmaceutical & Biopharmaceutical Companies
15.19.2. Medical Device Companies
15.19.3. Academic Institutes
15.20. Rest of South America Contract Research Organization Services (CROs) Market Forecast, by Type
15.20.1. Early Phase Development Services
15.20.1.1. Discovery Studies
15.20.1.2. Chemistry, Manufacturing & Control
15.20.1.3. Preclinical Services
15.20.2. Pharmacokinetics/Pharmacodynamics (PK/PD)
15.20.3. Toxicology Testing
15.20.4. Clinic Research Services
15.20.4.1. Phase I
15.20.4.2. Phase II
15.20.4.3. Phase III
15.20.4.4. Phase IV
15.20.5. Laboratory Services
15.20.5.1. Bioanalytical Testing
15.20.5.2. Analytical Testing
15.20.6. Physical Characterization
15.20.7. Stability Testing
15.20.8. Batch Release Testing
15.20.9. Raw Material Testing
15.20.10. Other Analytical Testing
15.20.11. Consulting Services
15.20.12. Other
15.21. South America Contract Research Organization Services (CROs) Market Attractiveness Analysis
15.21.1. By Therapeutic Area
15.21.2. By End-user
15.21.3. By Type
15.22. PEST Analysis
15.23. Key Trends
15.24. Key Developments
16. Company Profiles
16.1. Market Share Analysis, by Company
16.2. Competition Matrix
16.2.1. Competitive Benchmarking of key players by price, presence, market share, Applications and R&D investment
16.2.2. New Product Launches and Product Enhancements
16.2.3. Market Consolidation
16.2.3.1. M&A by Regions, Investment and Applications
16.2.3.2. M&A Key Players, Forward Integration and Backward
Integration
16.3. Company Profiles: Key Players
16.4.1. MPI Research
16.4.1.1. Company Overview
16.4.1.2. Financial Overview
16.4.1.3. Product Portfolio
16.4.1.4. Business Strategy
16.4.1.5. Recent Developments
16.4.1.6. Manufacturing Footprint
16.4.2. Quintiles Transnational Holdings Inc.
16.4.3. Envigo
16.4.4. SGS
16.4.5. MeDPAce Holdings
16.4.6. Charles River
16.4.7. WuXi Pharmatech
16.4.8. INC Research Holdings, Inc.
16.4.9. Syneos Health
16.4.10. ICON PLC
16.4.11. PPD
16.4.12. PRA Health Sciences
16.4.13. PAREXEL
16.4.14. LabCorp
16.4.15. IQVIA
16.4.16. Laboratory Corporation of America Holdings
16.4.17. Pharmaceutical Products Development, LLC.
16.4.18. Clintec
16.4.19. KCR S.A.
16.4.20. Covance Inc.
17. Primary Key Insights
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report