Lansoprazole Market – Global Market Size, Strategic Growth Drivers, Risk Assessment Framework, Regulatory Landscape Review, Competitive Intensity Mapping & Long-Term Industry Outlook to 2032
Overview
Global Lansoprazole Market size was valued at USD 2.00 Bn. in 2024, and the total Lansoprazole Market revenue is expected to grow by 6% from 2025 to 2032, reaching nearly USD 3.19 Bn.
Lansoprazole Market Overview:
Lansoprazole is a common proton pump inhibitor (PPI) to suppress gastric acid secretion in the stomach that is used for many indications that include gastroesophageal reflux disease (GERD) disorder, gastric ulcer, duodenal ulcer, and Zollinger-Ellison syndrome (ZES). The lansoprazole market has evolved away from just branded prescriptions to antibiotic prescriptions and even over the counter; in capsule, delayed-release, orally disintegrating tablet (ODT), oral suspension, and injection dosage forms, to meet every patient's needs. Globally, the growth of lansoprazole is due to the prevalence of gastrointestinal disorders linked to urbanization lifestyles, stress, poor diets, and the increased awareness around diagnosing and treating. For example, in Asia-Pacific, in particular, India and China, the demand for lansoprazole is increasing as healthcare access improves and generic medication is available. In North America and Europe, the growth of the OTC lansoprazole market is growing fast because of the aging population, and individuals looking for long-term total acid suppression without a prescription.
The report covered a detailed analysis of key growth factors affecting the lansoprazole market. For example, a growing trend of self-medication, worldwide growth in the production of generic pharmaceuticals from manufacturers, and the innovative application of lansoprazole in combination therapies in treating H. pylori infection. Key companies in the global lansoprazole market, including Takeda Pharmaceutical, Teva Pharmaceuticals, Dr. Reddy's Laboratories, Pfizer Inc., and Sun Pharmaceutical Industries, have recently introduced new formulations and are growing their over-the-counter (OTC) product lines and distribution to other new markets. This study demonstrates the importance of understanding how aggressive pricing and patent expiries, and regulatory approvals have transformed the lansoprazole market. Emerging trends include: preferences for orally disintegrating tablets, growth in pediatric and geriatric formulations, and a growing reliance on hospital use for IV lansoprazole in acute care settings.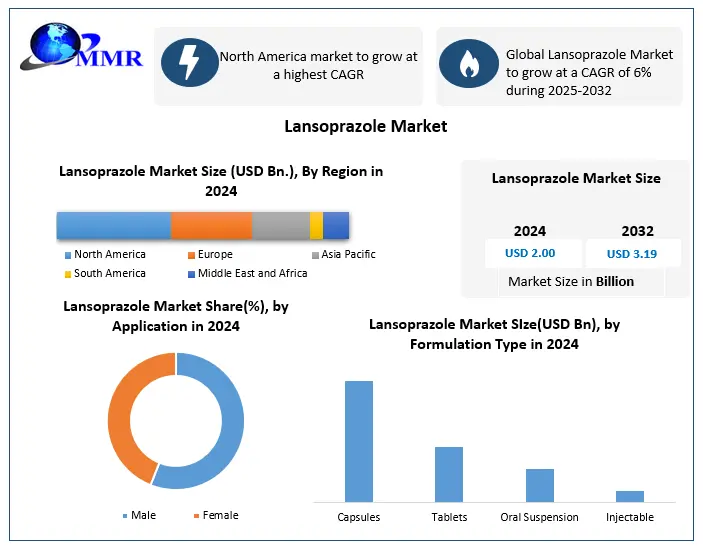 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
Global Lansoprazole Market Dynamics
Rising Incidence of Gastric Disorders to Drive Lansoprazole Market Growth
The demand for lansoprazole is transnationally driven by the increasing occurrence of gastrointestinal disorders (e.g., gastroesophageal reflux disorder (GERD), gastric ulcers, duodenal ulcers). Lansoprazole is a proton pump inhibitor (PPI) that is ideally suited for the long-term management of these disorders because it inhibits acid at the source rather than neutralizing stomach acid, like antacids. There is a rising incidence of acid-related disorders (due to lifestyle changes and the modern diet, including increased stress) that are creating sustained segmentation and not source-driven demand for long-term effective treatment options (including lansoprazole) on a global basis.
Innovation in Formulations to Create Lansoprazole Market Growth Opportunities
The lansoprazole market sees great opportunity with active clinical research and innovation surrounding the drug’s applicability and convenience for patients. Innovative formulations of lansoprazole in development aim to increase bioavailability, enhance-release formulations, and combination therapies to maximize treatment efficacy. Moreover, lansoprazole is available in both prescription and OTC formats, providing wider access to patients and consumers, and is especially relevant in emerging markets that have anywhere in the research from pharmaceutical research can improve patient healthcare and medical assistive devices. The innovations in the lansoprazole market will likely create new channels of continued growth and competition in the future.
Patient Shift to Alternative Therapies to Present Lansoprazole Market Challenges
Despite its proven efficacy, the lansoprazole market is facing strong pressures as more patients continue to choose alternative drug classes in addition to PPIs, such as H2 blockers and antacid services, including over-the-counter products. Although there are various PPIs in the market, these medicines are often misused, inappropriately used, and especially used too often for something like heartburn. Therefore, the appeal of immediate symptomatic relief, combined with increased awareness of and discomfort with the many potential long-term side effects associated with the long-term use of PPIs (e.g., nutrient malabsorption, dependency, etc.), has motivated the approach of many patients and medical providers to short-acting alternatives. Additionally, because many generic PPIs continue to be available as well as substitutes that are closely priced to PPIs, price pressure is estimated to increase, adversely affecting the performance of premium brands within the market.
Global Lansoprazole Market Segment Analysis
Based on the Route of Administration, the market is segmented into Oral and Intravenous. The oral route of administration segment is expected to hold the largest market share of xx% by 2032. This is due to patients with mild to moderate gastroesophageal reflux disease preferring capsule formulations, the low cost of oral capsules, and an increase in the number of manufacturers in the global market supplying lansoprazole in capsule form. These are the factors that are expected to drive the growth of the Oral route of administration segment in the Lansoprazole Market during the forecast period.
Based on the Formulation Type, the Lansoprazole market is segmented into tablets, capsules, oral suspension, and injectables. Capsule segment dominated the lansoprazole market in 2024 and is expected to hold the largest market share over the forecast period. The preference for Capsules is essentially due to capsule medications being the most commonly used and prescribed migraine formulation, because capsules are delayed-release dosage formulations that bypass the stomach and allow for the absorption of the medication in the intestine to produce more effective efficacy of therapeutic benefits. Capsules are easy to manufacture, provide improved drug stability, and are an effective route for treating GERD and peptic ulcers in long-term use.
Lansoprazole Market Regional Insights
The North American region is expected to dominate the Lansoprazole Market during the forecast period 2025-2032. The North American region is expected to hold the largest market share of xx% by 2032. This is due to an increase in the frequency of GERD in the United States and a larger demand in the region for sustained-release proton pump inhibitor medicines. According to the World Gastroenterology Organisation, the prevalence of GERD in Eastern Asia ranged from 2.5 percent to 6.6 percent in 2024, while in North America it ranged from 13.8 percent to 25.8%. These are the factors that are expected to drive the growth of the North American region in the Lansoprazole Market during the forecast period.
Lansoprazole Market Competitive Landscape
The Lansoprazole Market is highly competitive, as a combination of originator brands along with global generic pharmaceutical manufacturers. In the Lansoprazole Market, players such as Takeda Pharmaceutical and Teva Pharmaceuticals will lead with their innovation, distribution footprint, and regulatory approvals in large markets. Takeda Pharmaceutical (Japan) developed lansoprazole with the brand name Prevacid. Takeda Pharmaceutical's revenue had a very strong performance through a blockbuster drug, until it found its way off patent. In 2024, Prevacid is facing some generic erosion, but maintains a strong share of the premium and combination therapy market as it continues in Japan, the U.S., and portions of Europe. Takeda has shifted their focus to lifecycle extension strategies and accessibility of GI therapies in the Pacific and emerging markets. Teva Pharmaceuticals (Israel), one of the largest generic providers in the world, has a solid position in the lansoprazole space with FDA-approved delayed-release capsules and orally disintegrating tablets. In 2024, Teva’s generics portfolio, including lansoprazole, yielded a significant share of its 15 billion generics revenue. Teva continues to leverage global manufacturing scale and regulatory prowess to provide affordable proton pump inhibitors (PPIs) in North America, Europe, and Latin America. Both companies have similarly invested in GI treatment innovation and continue to compete through cost-efficient manufacturing, numerous formulations (ODT, combo-packs), and expanding access to over-the-counter markets to meet the increased global demand for acid suppressive therapy.
Lansoprazole Market Recent Development
• Takeda Pharmaceutical (Japan) announced in its FY2024 results in May 2025 that while the company has worked with loss of exclusivity for lansoprazole (Prevacid), it was managing this decline with high growth and launch product performance, as well as strong investment in R&D and focus on late-stage pipeline assets.
• Teva Pharmaceutical (Israel) announced the launch of its FDA-approved generic, delayed-release orally disintegrating tablets of lansoprazole (the therapeutic equivalent of Prevacid SoluTab) in early 2025, which provided patients with a cost savings and ease of administration.
• In May 2025, Dr. Reddy's Laboratories (India/USA) launched lansoprazole delayed-release capsules (15 mg) into the U.S. OTC market to increase options for self-care; it also continues to manufacture delayed-release orally disintegrating tablets in the U.S. as a generic equivalent to Prevacid SoluTab.
• Sun Pharmaceutical Industries Ltd. (India) reaffirmed its continued strong position in generics and lansoprazole—in Q4 FY2025 in India, with support from its 6-8% annual investment in R&D, and manufacturing facility capabilities from APIs to specialty generics.
• Pfizer Inc. (USA) continues to support its position in the lansoprazole market with its Zoton FasTab™ delayed-release orally disintegrating tablets in the UK, supported by its wider pipeline of pharmaceutical products and ongoing opportunities in global marketing.
Lansoprazole Market Recent Trends
| Category | Key Trend | Example Product | Market Impact |
| Formulation Innovation | Shift toward orally disintegrating tablets (ODTs) for ease of administration. | Dr. Reddy’s Lansoprazole ODT (Generic Prevacid SoluTab) | Increased patient compliance and market share in pediatric and geriatric populations |
| Combination Therapies | Integration of lansoprazole in H. pylori treatment kits with antibiotics | Takeda’s Lansoprazole + Amoxicillin + Clarithromycin Pack | Strengthened clinical utility in ulcer management; boosts hospital and specialist segment demand |
| Generic Expansion | Surge in FDA/EMA-approved generic lansoprazole by global manufacturers | Teva Lansoprazole Delayed-Release Tablets | Intensified competition and affordability; generics dominate >85% of global lansoprazole supply |
Lansoprazole Market Scope: Inquiry Before Buying
| Lansoprazole Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2024 | Forecast Period: | 2025-2032 |
| Historical Data: | 2019 to 2024 | Market Size in 2024: | USD 2.00 Bn. |
| Forecast Period 2025 to 2032 CAGR: | 6% | Market Size in 2032: | USD 3.19 Bn. |
| Segments Covered: | by Route of Administration | Oral Intravenous |
|
| by Application | Male Female |
||
| by Formulation Type | Capsule Tablet Oral suspension Injectable |
||
Lansoprazole Market by Region
North America (United States, Canada, and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria, and the Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, ASEAN, and the Rest of APAC)
Middle East & Africa (South Africa, GCC, Nigeria, Rest of ME&A)
South America (Brazil, Argentina, Rest of South America)
Lansoprazole Market Key Players
North America
1. Pfizer Inc. (USA)
2. Perrigo Company plc (USA)
Europe
1. Teva Pharmaceuticals (Israel)
2. Sandoz (Novartis division) (Switzerland)
3. GSK (GlaxoSmithKline) (United Kingdom)
Asia Pacific
1. Takeda Pharmaceutical (Japan)
2. Dr. Reddy's Laboratories (India)
3. Taj Pharmaceuticals (India)
4. Sun Pharmaceutical Industries Ltd. (India)
5. Mylan (India)
6. Cisen Pharmaceutical (China)
7. Lepu Pharmaceuticals (China)
8. Nang Kuang Pharmaceutical (Taiwan)
9. Luoxin Biotechnology (China)
10. Beijing Honglin Pharma (China)
11. Suzhou YUSHI Pharmaceutical Co. (China)
12. Changzhou Siyao Pharmaceuticals (China)
Frequently Asked Questions:
1. Which region has the largest share in the Global Lansoprazole Market?
Ans: The North America region held the highest share in 2024.
2. What is the growth rate of the Global Lansoprazole Market?
Ans: The Global Market is expected to grow at a CAGR of 6% during the forecast period 2025-2032.
3. What is the scope of the Global Lansoprazole Market report?
Ans: The Global Lansoprazole Market report helps with the PESTEL, Porter's, Recommendations for Investors and leaders, and market estimation for the forecast period.
4. Who are the key players in the Global Lansoprazole Market?
Ans: The important key players in the Global Lansoprazole Market are- Taj Pharmaceuticals (India), Sun Pharmaceutical Industries Ltd. (India), Mylan (India), Cisen Pharmaceutical (China), Lepu Pharmaceuticals (China), Nang Kuang Pharmaceutical (Taiwan), etc.
5. What is the study period of this market?
Ans: The Global Lansoprazole Market is studied from 2024 to 2032.
