1. Vitro Diagnostics Market: Research Methodology
2. Vitro Diagnostics Market Introduction
2.1. Study Assumption and Market Definition
2.2. Scope of the Study
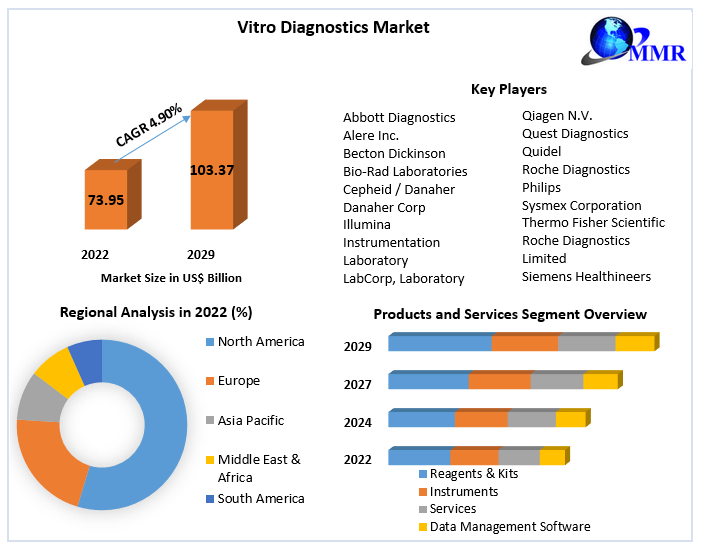
2.3. Executive Summary
3. Vitro Diagnostics Market: Qualitative Analysis
3.1. Techniques used in Diagnostic
3.2. Technological Advancements in diagnostic
4. Vitro Diagnostics Market: Dynamics
4.1. Vitro Diagnostics Market Trends by Region
4.1.1. North America Vitro Diagnostics Market Trends
4.1.2. Europe Vitro Diagnostics Market Trends
4.1.3. Asia Pacific Vitro Diagnostics Market Trends
4.1.4. Middle East and Africa Vitro Diagnostics Market Trends
4.1.5. South America Vitro Diagnostics Market Trends
4.2. Vitro Diagnostics Market Dynamics By Region
4.2.1. North America
4.2.1.1. North America Vitro Diagnostics Market Drivers
4.2.1.2. North America Vitro Diagnostics Market Restraints
4.2.1.3. North America Vitro Diagnostics Market Opportunities
4.2.1.4. North America Vitro Diagnostics Market Challenges
4.2.2. Europe
4.2.2.1. Europe Vitro Diagnostics Market Drivers
4.2.2.2. Europe Vitro Diagnostics Market Restraints
4.2.2.3. Europe Vitro Diagnostics Market Opportunities
4.2.2.4. Europe Vitro Diagnostics Market Challenges
4.2.3. Asia Pacific
4.2.3.1. Asia Pacific Vitro Diagnostics Market Drivers
4.2.3.2. Asia Pacific Vitro Diagnostics Market Restraints
4.2.3.3. Asia Pacific Vitro Diagnostics Market Opportunities
4.2.3.4. Asia Pacific Vitro Diagnostics Market Challenges
4.2.4. Middle East and Africa
4.2.4.1. Middle East and Africa Vitro Diagnostics Market Drivers
4.2.4.2. Middle East and Africa Vitro Diagnostics Market Restraints
4.2.4.3. Middle East and Africa Vitro Diagnostics Market Opportunities
4.2.4.4. Middle East and Africa Vitro Diagnostics Market Challenges
4.2.5. South America
4.2.5.1. South America Vitro Diagnostics Market Drivers
4.2.5.2. South America Vitro Diagnostics Market Restraints
4.2.5.3. South America Vitro Diagnostics Market Opportunities
4.2.5.4. South America Vitro Diagnostics Market Challenges
4.3. PORTER’s Five Forces Analysis
4.4. PESTLE Analysis
4.5. Regulatory Landscape By Region
4.5.1. North America
4.5.2. Europe
4.5.3. Asia Pacific
4.5.4. Middle East and Africa
4.5.5. South America
4.6. Key Opinion Leader Analysis For Industry
4.7. Analysis of Government Schemes and Initiatives Industry
4.8. The Global Pandemic Impact on Vitro Diagnostics Market
5. Vitro Diagnostics Market: Global Market Size and Forecast by Segmentation (by Value in USD Million) (2023-2030)
5.1. Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
5.1.1. Reagents & Kits
5.1.2. Instruments
5.1.3. Services
5.1.4. Data Management Software
5.2. Vitro Diagnostics Market Size and Forecast, by Technology (2023-2030)
5.2.1. Clinical Chemistry
5.2.2. Basic Metabolic Panel
5.2.3. Electrolyte Panel
5.2.4. Liver Panel
5.2.5. Lipid Profile
5.2.6. Renal Profile
5.3. Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
5.3.1. Diabetes
5.3.2. Infectious Diseases
5.3.3. Oncology/Cancer
5.3.4. Cardiology
5.3.5. Nephrology
5.3.6. Autoimmune Diseases
5.3.7. Drug Testing
5.4. Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
5.4.1. Hospitals
5.4.2. Laboratories
5.4.3. Academic Institutes
5.4.4. Point-Of-Care Testing
5.4.5. Patient Self-Testing
5.4.6. Other End Users
5.5. Vitro Diagnostics Market Size and Forecast, By Region (2023-2030)
5.5.1. North America
5.5.2. Europe
5.5.3. Asia Pacific
5.5.4. Middle East and Africa
5.5.5. South America
6. North America Vitro Diagnostics Market Size and Forecast by Segmentation (by Value in USD Million) (2023-2030)
6.1. North America Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
6.1.1. Reagents & Kits
6.1.2. Instruments
6.1.3. Services
6.1.4. Data Management Software
6.2. North America Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
6.2.1. Clinical Chemistry
6.2.2. Basic Metabolic Panel
6.2.3. Electrolyte Panel
6.2.4. Liver Panel
6.2.5. Lipid Profile
6.2.6. Renal Profile
6.3. North America Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
6.3.1. Diabetes
6.3.2. Infectious Diseases
6.3.3. Oncology/Cancer
6.3.4. Cardiology
6.3.5. Nephrology
6.3.6. Autoimmune Diseases
6.3.7. Drug Testing
6.4. North America Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
6.4.1. Hospitals
6.4.2. Laboratories
6.4.3. Academic Institutes
6.4.4. Point-Of-Care Testing
6.4.5. Patient Self-Testing
6.4.6. Other End Users
6.5. North America Vitro Diagnostics Market Size and Forecast, by Country (2023-2030)
6.5.1. United States
6.5.1.1. United States Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
6.5.1.1.1. Reagents & Kits
6.5.1.1.2. Instruments
6.5.1.1.3. Services
6.5.1.1.4. Data Management Software
6.5.1.2. United States Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
6.5.1.2.1. Clinical Chemistry
6.5.1.2.2. Basic Metabolic Panel
6.5.1.2.3. Electrolyte Panel
6.5.1.2.4. Liver Panel
6.5.1.2.5. Lipid Profile
6.5.1.2.6. Renal Profile
6.5.1.3. United States Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
6.5.1.3.1. Diabetes
6.5.1.3.2. Infectious Diseases
6.5.1.3.3. Oncology/Cancer
6.5.1.3.4. Cardiology
6.5.1.3.5. Nephrology
6.5.1.3.6. Autoimmune Diseases
6.5.1.3.7. Drug Testing
6.5.1.4. United States Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
6.5.1.4.1. Hospitals
6.5.1.4.2. Laboratories
6.5.1.4.3. Academic Institutes
6.5.1.4.4. Point-Of-Care Testing
6.5.1.4.5. Patient Self-Testing
6.5.1.4.6. Other End Users
6.5.2. Canada
6.5.2.1. Canada Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
6.5.2.1.1. Reagents & Kits
6.5.2.1.2. Instruments
6.5.2.1.3. Services
6.5.2.1.4. Data Management Software
6.5.2.2. Canada Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
6.5.2.2.1. Clinical Chemistry
6.5.2.2.2. Basic Metabolic Panel
6.5.2.2.3. Electrolyte Panel
6.5.2.2.4. Liver Panel
6.5.2.2.5. Lipid Profile
6.5.2.2.6. Renal Profile
6.5.2.3. Canada Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
6.5.2.3.1. Diabetes
6.5.2.3.2. Infectious Diseases
6.5.2.3.3. Oncology/Cancer
6.5.2.3.4. Cardiology
6.5.2.3.5. Nephrology
6.5.2.3.6. Autoimmune Diseases
6.5.2.3.7. Drug Testing
6.5.2.4. Canada Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
6.5.2.4.1. Hospitals
6.5.2.4.2. Laboratories
6.5.2.4.3. Academic Institutes
6.5.2.4.4. Point-Of-Care Testing
6.5.2.4.5. Patient Self-Testing
6.5.2.4.6. Other End Users
6.5.3. Mexico
6.5.3.1. Mexico Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
6.5.3.1.1. Reagents & Kits
6.5.3.1.2. Instruments
6.5.3.1.3. Services
6.5.3.1.4. Data Management Software
6.5.3.2. Mexico Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
6.5.3.2.1. Clinical Chemistry
6.5.3.2.2. Basic Metabolic Panel
6.5.3.2.3. Electrolyte Panel
6.5.3.2.4. Liver Panel
6.5.3.2.5. Lipid Profile
6.5.3.2.6. Renal Profile
6.5.3.3. Mexico Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
6.5.3.3.1. Diabetes
6.5.3.3.2. Infectious Diseases
6.5.3.3.3. Oncology/Cancer
6.5.3.3.4. Cardiology
6.5.3.3.5. Nephrology
6.5.3.3.6. Autoimmune Diseases
6.5.3.3.7. Drug Testing
6.5.3.4. Mexico Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
6.5.3.4.1. Hospitals
6.5.3.4.2. Laboratories
6.5.3.4.3. Academic Institutes
6.5.3.4.4. Point-Of-Care Testing
6.5.3.4.5. Patient Self-Testing
6.5.3.4.6. Other End Users
7. Europe Vitro Diagnostics Market Size and Forecast by Segmentation (by Value in USD Million) (2023-2030)
7.1. Europe Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
7.2. Europe Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
7.3. Europe Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
7.4. Europe Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
7.5. Europe Vitro Diagnostics Market Size and Forecast, by Country (2023-2030)
7.5.1. United Kingdom
7.5.1.1. United Kingdom Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
7.5.1.2. United Kingdom Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
7.5.1.3. United Kingdom Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
7.5.1.4. United Kingdom Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
7.5.2. France
7.5.2.1. France Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
7.5.2.2. France Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
7.5.2.3. France Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
7.5.2.4. France Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
7.5.3. Germany
7.5.3.1. Germany Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
7.5.3.2. Germany Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
7.5.3.3. Germany Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
7.5.3.4. Germany Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
7.5.4. Italy
7.5.4.1. Italy Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
7.5.4.2. Italy Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
7.5.4.3. Italy Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
7.5.4.4. Italy Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
7.5.5. Spain
7.5.5.1. Spain Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
7.5.5.2. Spain Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
7.5.5.3. Spain Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
7.5.5.4. Spain Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
7.5.6. Sweden
7.5.6.1. Sweden Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
7.5.6.2. Sweden Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
7.5.6.3. Sweden Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
7.5.6.4. Sweden Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
7.5.7. Austria
7.5.7.1. Austria Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
7.5.7.2. Austria Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
7.5.7.3. Austria Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
7.5.7.4. Austria Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
7.5.8. Rest of Europe
7.5.8.1. Rest of Europe Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
7.5.8.2. Rest of Europe Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
7.5.8.3. Rest of Europe Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
7.5.8.4. Rest of Europe Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
8. Asia Pacific Vitro Diagnostics Market Size and Forecast by Segmentation (by Value in USD Million) (2023-2030)
8.1. Asia Pacific Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
8.2. Asia Pacific Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
8.3. Asia Pacific Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
8.4. Asia Pacific Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
8.5. Asia Pacific Vitro Diagnostics Market Size and Forecast, by Country (2023-2030)
8.5.1. China
8.5.1.1. China Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
8.5.1.2. China Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
8.5.1.3. China Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
8.5.1.4. China Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
8.5.2. S Korea
8.5.2.1. S Korea Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
8.5.2.2. S Korea Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
8.5.2.3. S Korea Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
8.5.2.4. S Korea Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
8.5.3. Japan
8.5.3.1. Japan Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
8.5.3.2. Japan Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
8.5.3.3. Japan Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
8.5.3.4. Japan Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
8.5.4. India
8.5.4.1. India Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
8.5.4.2. India Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
8.5.4.3. India Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
8.5.4.4. India Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
8.5.5. Australia
8.5.5.1. Australia Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
8.5.5.2. Australia Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
8.5.5.3. Australia Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
8.5.5.4. Australia Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
8.5.6. Indonesia
8.5.6.1. Indonesia Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
8.5.6.2. Indonesia Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
8.5.6.3. Indonesia Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
8.5.6.4. Indonesia Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
8.5.7. Malaysia
8.5.7.1. Malaysia Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
8.5.7.2. Malaysia Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
8.5.7.3. Malaysia Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
8.5.7.4. Malaysia Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
8.5.8. Vietnam
8.5.8.1. Vietnam Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
8.5.8.2. Vietnam Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
8.5.8.3. Vietnam Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
8.5.8.4. Vietnam Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
8.5.9. Taiwan
8.5.9.1. Taiwan Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
8.5.9.2. Taiwan Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
8.5.9.3. Taiwan Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
8.5.9.4. Taiwan Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
8.5.10. Rest of Asia Pacific
8.5.10.1. Rest of Asia Pacific Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
8.5.10.2. Rest of Asia Pacific Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
8.5.10.3. Rest of Asia Pacific Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
8.5.10.4. Rest of Asia Pacific Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
9. Middle East and Africa Vitro Diagnostics Market Size and Forecast by Segmentation (by Value in USD Million) (2023-2030
9.1. Middle East and Africa Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
9.2. Middle East and Africa Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
9.3. Middle East and Africa Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
9.4. Middle East and Africa Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
9.5. Middle East and Africa Vitro Diagnostics Market Size and Forecast, by Country (2023-2030)
9.5.1. South Africa
9.5.1.1. South Africa Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
9.5.1.2. South Africa Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
9.5.1.3. South Africa Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
9.5.1.4. South Africa Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
9.5.2. GCC
9.5.2.1. GCC Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
9.5.2.2. GCC Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
9.5.2.3. GCC Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
9.5.2.4. GCC Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
9.5.3. Nigeria
9.5.3.1. Nigeria Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
9.5.3.2. Nigeria Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
9.5.3.3. Nigeria Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
9.5.3.4. Nigeria Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
9.5.4. Rest of ME&A
9.5.4.1. Rest of ME&A Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
9.5.4.2. Rest of ME&A Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
9.5.4.3. Rest of ME&A Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
9.5.4.4. Rest of ME&A Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
10. South America Vitro Diagnostics Market Size and Forecast by Segmentation (by Value in USD Million) (2023-2030
10.1. South America Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
10.2. South America Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
10.3. South America Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
10.4. South America Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
10.5. South America Vitro Diagnostics Market Size and Forecast, by Country (2023-2030)
10.5.1. Brazil
10.5.1.1. Brazil Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
10.5.1.2. Brazil Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
10.5.1.3. Brazil Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
10.5.1.4. Brazil Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
10.5.2. Argentina
10.5.2.1. Argentina Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
10.5.2.2. Argentina Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
10.5.2.3. Argentina Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
10.5.2.4. Argentina Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
10.5.3. Rest Of South America
10.5.3.1. Rest Of South America Vitro Diagnostics Market Size and Forecast, By Products and Services (2023-2030)
10.5.3.2. Rest Of South America Vitro Diagnostics Market Size and Forecast, By Technology (2023-2030)
10.5.3.3. Rest Of South America Vitro Diagnostics Market Size and Forecast, By Application (2023-2030)
10.5.3.4. Rest Of South America Vitro Diagnostics Market Size and Forecast, By End-User (2023-2030)
11. Global Vitro Diagnostics Market: Competitive Landscape
11.1. MMR Competition Matrix
11.2. Competitive Landscape
11.3. Key Players Benchmarking
11.3.1. Company Name
11.3.2. Service Segment
11.3.3. End-user Segment
11.3.4. Revenue (2023)
11.3.5. Company Locations
11.4. Leading Vitro Diagnostics Market Companies, by Market Capitalization
11.5. Market Structure
11.5.1. Market Leaders
11.5.2. Market Followers
11.5.3. Emerging Players
11.6. Mergers and Acquisitions Details
12. Company Profile: Key Players
12.1. Abbott Diagnostics
12.1.1. Company Overview
12.1.2. Business Portfolio
12.1.3. Financial Overview
12.1.4. SWOT Analysis
12.1.5. Strategic Analysis
12.1.6. Recent Developments
12.2. Bio-Rad Laboratories
12.3. Danaher Corp
12.4. Illumina
12.5. Instrumentation Laboratory
12.6. LabCorp, Laboratory Corporation of America
12.7. Luminex
12.8. Ortho Clinical Diagnostics
12.9. PerkinElmer
12.10. Qiagen N.V.
12.11. Quest Diagnostics
12.12. Quidel
12.13. Roche Diagnostics
12.14. Philips
12.15. Sysmex Corporation
12.16. Thermo Fisher Scientific
12.17. Roche Diagnostics Limited
12.18. Siemens Healthineers
12.19. Abbott Laboratories
12.20. Johnson & Johnson
12.21. Becton, Dickinson and Company
13. Key Findings
14. Industry Recommendations
 To know about the Research Methodology:-Request Free Sample Report
To know about the Research Methodology:-Request Free Sample Report