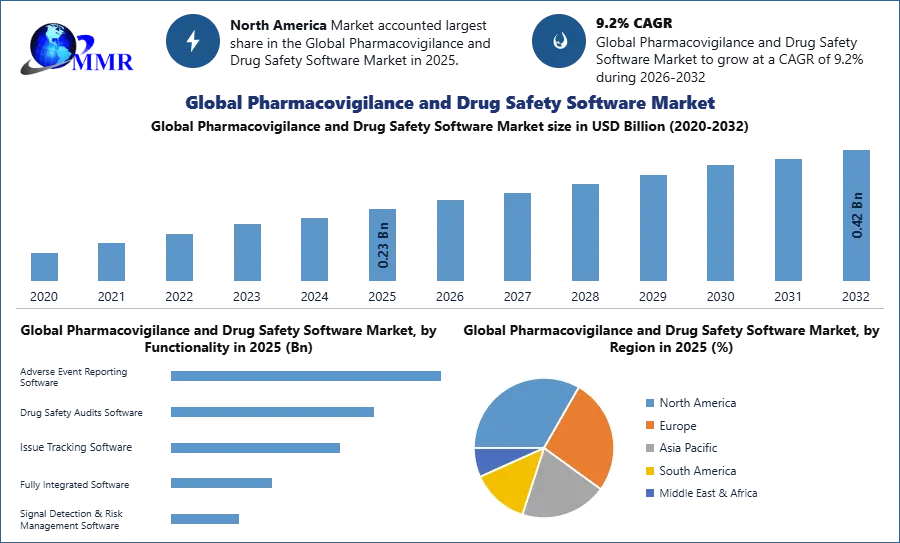
1. Global Pharmacovigilance and Drug Safety Software Market Introduction
1.1. Study Assumption and Market Definition
1.2. Scope of the Study
1.3. Executive Summary
2. Global Global Pharmacovigilance and Drug Safety Software Market: Competitive Landscape
2.1. MMR Competition Matrix
2.2. Competitive Landscape
2.3. Key Players Benchmarking
2.3.1. Company Name
2.3.2. Business Segment
2.3.3. End-user Segment
2.3.4. Revenue (2025)
2.3.5. Company Locations
2.4. Leading Global Pharmacovigilance and Drug Safety Software Market Companies, by market capitalization
2.5. Market Structure
2.5.1. Market Leaders
2.5.2. Market Followers
2.5.3. Emerging Players
2.6. Mergers and Acquisitions Details
3. Global Pharmacovigilance and Drug Safety Software Market: Dynamics
3.1. Global Pharmacovigilance and Drug Safety Software Market Trends by Region
3.1.1. North America Global Pharmacovigilance and Drug Safety Software Market Trends
3.1.2. Europe Global Pharmacovigilance and Drug Safety Software Market Trends
3.1.3. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Trends
3.1.4. Middle East and Africa Global Pharmacovigilance and Drug Safety Software Market Trends
3.1.5. South America Global Pharmacovigilance and Drug Safety Software Market Trends
3.2. Global Pharmacovigilance and Drug Safety Software Market Dynamics by Region
3.2.1. North America
3.2.1.1. North America Global Pharmacovigilance and Drug Safety Software Market Drivers
3.2.1.2. North America Global Pharmacovigilance and Drug Safety Software Market Restraints
3.2.1.3. North America Global Pharmacovigilance and Drug Safety Software Market Opportunities
3.2.1.4. North America Global Pharmacovigilance and Drug Safety Software Market Challenges
3.2.2. Europe
3.2.2.1. Europe Global Pharmacovigilance and Drug Safety Software Market Drivers
3.2.2.2. Europe Global Pharmacovigilance and Drug Safety Software Market Restraints
3.2.2.3. Europe Global Pharmacovigilance and Drug Safety Software Market Opportunities
3.2.2.4. Europe Global Pharmacovigilance and Drug Safety Software Market Challenges
3.2.3. Asia Pacific
3.2.3.1. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Drivers
3.2.3.2. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Restraints
3.2.3.3. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Opportunities
3.2.3.4. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Challenges
3.2.4. Middle East and Africa
3.2.4.1. Middle East and Africa Global Pharmacovigilance and Drug Safety Software Market Drivers
3.2.4.2. Middle East and Africa Global Pharmacovigilance and Drug Safety Software Market Restraints
3.2.4.3. Middle East and Africa Global Pharmacovigilance and Drug Safety Software Market Opportunities
3.2.4.4. Middle East and Africa Global Pharmacovigilance and Drug Safety Software Market Challenges
3.2.5. South America
3.2.5.1. South America Global Pharmacovigilance and Drug Safety Software Market Drivers
3.2.5.2. South America Global Pharmacovigilance and Drug Safety Software Market Restraints
3.2.5.3. South America Global Pharmacovigilance and Drug Safety Software Market Opportunities
3.2.5.4. South America Global Pharmacovigilance and Drug Safety Software Market Challenges
3.3. PORTER's Five Forces Analysis
3.4. PESTLE Analysis
3.5. Technology Roadmap
3.6. Regulatory Landscape by Region
3.6.1. North America
3.6.2. Europe
3.6.3. Asia Pacific
3.6.4. Middle East and Africa
3.6.5. South America
3.7. Key Opinion Leader Analysis For Global Pharmacovigilance and Drug Safety Software Industry
3.8. Analysis of Government Schemes and Initiatives For Global Pharmacovigilance and Drug Safety Software Industry
3.9. Global Pharmacovigilance and Drug Safety Software Market Trade Analysis
3.10. The Global Pandemic Impact on Global Pharmacovigilance and Drug Safety Software Market
4. Global Pharmacovigilance and Drug Safety Software Market: Global Market Size and Forecast by Segmentation (in USD Billion) 2025-2032
4.1. Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
4.1.1. Adverse Event Reporting Software
4.1.2. Drug Safety Audits Software
4.1.3. Issue Tracking Software
4.1.4. Fully Integrated Software
4.1.5. Signal Detection & Risk Management Software
4.2. Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
4.2.1. On-Premises
4.2.2. Cloud-based (SaaS)
4.3. Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
4.3.1. Software
4.3.2. Services (Implementation, Support & Maintenance)
4.4. Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
4.4.1. Pharmaceutical & Biotechnology Companies
4.4.2. Contract Research Organizations (CROs)
4.4.3. Business Process Outsourcing (BPO) Firms
4.4.4. Pharmacovigilance Service Providers
4.5. Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Region (2025-2032)
4.5.1. North America
4.5.2. Europe
4.5.3. Asia Pacific
4.5.4. Middle East and Africa
4.5.5. South America
5. North America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast by Segmentation (in USD Billion) 2025-2032
5.1. North America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
5.1.1. Adverse Event Reporting Software
5.1.2. Drug Safety Audits Software
5.1.3. Issue Tracking Software
5.1.4. Fully Integrated Software
5.1.5. Signal Detection & Risk Management Software
5.2. North America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
5.2.1. On-Premises
5.2.2. Cloud-based (SaaS)
5.3. North America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
5.3.1. Software
5.3.2. Services (Implementation, Support & Maintenance)
5.4. North America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
5.4.1. Pharmaceutical & Biotechnology Companies
5.4.2. Contract Research Organizations (CROs)
5.4.3. Business Process Outsourcing (BPO) Firms
5.4.4. Pharmacovigilance Service Providers
5.5. North America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Country (2025-2032)
5.5.1. United States
5.5.1.1. United States Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
5.5.1.1.1. Adverse Event Reporting Software
5.5.1.1.2. Drug Safety Audits Software
5.5.1.1.3. Issue Tracking Software
5.5.1.1.4. Fully Integrated Software
5.5.1.1.5. Signal Detection & Risk Management Software
5.5.1.2. United States Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
5.5.1.2.1. On-Premises
5.5.1.2.2. Cloud-based (SaaS)
5.5.1.3. United States Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
5.5.1.3.1. Software
5.5.1.3.2. Services (Implementation, Support & Maintenance)
5.5.1.4. United States Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
5.5.1.4.1. Pharmaceutical & Biotechnology Companies
5.5.1.4.2. Contract Research Organizations (CROs)
5.5.1.4.3. Business Process Outsourcing (BPO) Firms
5.5.1.4.4. Pharmacovigilance Service Providers
5.5.2. Canada
5.5.2.1. Canada Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
5.5.2.1.1. Adverse Event Reporting Software
5.5.2.1.2. Drug Safety Audits Software
5.5.2.1.3. Issue Tracking Software
5.5.2.1.4. Fully Integrated Software
5.5.2.1.5. Signal Detection & Risk Management Software
5.5.2.2. Canada Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
5.5.2.2.1. On-Premises
5.5.2.2.2. Cloud-based (SaaS)
5.5.2.3. Canada Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
5.5.2.3.1. Software
5.5.2.3.2. Services (Implementation, Support & Maintenance)
5.5.2.4. Canada Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
5.5.2.4.1. Pharmaceutical & Biotechnology Companies
5.5.2.4.2. Contract Research Organizations (CROs)
5.5.2.4.3. Business Process Outsourcing (BPO) Firms
5.5.2.4.4. Pharmacovigilance Service Providers
5.5.3. Mexico
5.5.3.1. Mexico Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
5.5.3.1.1. Adverse Event Reporting Software
5.5.3.1.2. Drug Safety Audits Software
5.5.3.1.3. Issue Tracking Software
5.5.3.1.4. Fully Integrated Software
5.5.3.1.5. Signal Detection & Risk Management Software
5.5.3.2. Mexico Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
5.5.3.2.1. On-Premises
5.5.3.2.2. Cloud-based (SaaS)
5.5.3.3. Mexico Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
5.5.3.3.1. Software
5.5.3.3.2. Services (Implementation, Support & Maintenance)
5.5.3.4. Mexico Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
5.5.3.4.1. Pharmaceutical & Biotechnology Companies
5.5.3.4.2. Contract Research Organizations (CROs)
5.5.3.4.3. Business Process Outsourcing (BPO) Firms
5.5.3.4.4. Pharmacovigilance Service Providers
6. Europe Global Pharmacovigilance and Drug Safety Software Market Size and Forecast by Segmentation (in USD Billion) 2025-2032
6.1. Europe Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
6.2. Europe Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
6.3. Europe Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
6.4. Europe Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
6.5. Europe Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Country (2025-2032)
6.5.1. United Kingdom
6.5.1.1. United Kingdom Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
6.5.1.2. United Kingdom Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
6.5.1.3. United Kingdom Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
6.5.1.4. United Kingdom Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
6.5.2. France
6.5.2.1. France Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
6.5.2.2. France Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
6.5.2.3. France Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
6.5.2.4. France Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
6.5.3. Germany
6.5.3.1. Germany Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
6.5.3.2. Germany Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
6.5.3.3. Germany Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
6.5.3.4. Germany Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
6.5.4. Italy
6.5.4.1. Italy Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
6.5.4.2. Italy Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
6.5.4.3. Italy Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
6.5.4.4. Italy Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
6.5.5. Spain
6.5.5.1. Spain Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
6.5.5.2. Spain Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
6.5.5.3. Spain Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
6.5.5.4. Spain Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
6.5.6. Sweden
6.5.6.1. Sweden Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
6.5.6.2. Sweden Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
6.5.6.3. Sweden Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
6.5.6.4. Sweden Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
6.5.7. Austria
6.5.7.1. Austria Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
6.5.7.2. Austria Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
6.5.7.3. Austria Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
6.5.7.4. Austria Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
6.5.8. Rest of Europe
6.5.8.1. Rest of Europe Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
6.5.8.2. Rest of Europe Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
6.5.8.3. Rest of Europe Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
6.5.8.4. Rest of Europe Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
7. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Size and Forecast by Segmentation (in USD Billion) 2025-2032
7.1. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
7.2. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
7.3. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
7.4. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
7.5. Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Country (2025-2032)
7.5.1. China
7.5.1.1. China Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
7.5.1.2. China Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
7.5.1.3. China Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
7.5.1.4. China Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
7.5.2. S Korea
7.5.2.1. S Korea Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
7.5.2.2. S Korea Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
7.5.2.3. S Korea Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
7.5.2.4. S Korea Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
7.5.3. Japan
7.5.3.1. Japan Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
7.5.3.2. Japan Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
7.5.3.3. Japan Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
7.5.3.4. Japan Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
7.5.4. India
7.5.4.1. India Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
7.5.4.2. India Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
7.5.4.3. India Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
7.5.4.4. India Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
7.5.5. Australia
7.5.5.1. Australia Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
7.5.5.2. Australia Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
7.5.5.3. Australia Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
7.5.5.4. Australia Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
7.5.6. Indonesia
7.5.6.1. Indonesia Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
7.5.6.2. Indonesia Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
7.5.6.3. Indonesia Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
7.5.6.4. Indonesia Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
7.5.7. Malaysia
7.5.7.1. Malaysia Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
7.5.7.2. Malaysia Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
7.5.7.3. Malaysia Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
7.5.7.4. Malaysia Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
7.5.8. Vietnam
7.5.8.1. Vietnam Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
7.5.8.2. Vietnam Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
7.5.8.3. Vietnam Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
7.5.8.4. Vietnam Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
7.5.9. Taiwan
7.5.9.1. Taiwan Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
7.5.9.2. Taiwan Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
7.5.9.3. Taiwan Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
7.5.9.4. Taiwan Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
7.5.10. Rest of Asia Pacific
7.5.10.1. Rest of Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
7.5.10.2. Rest of Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
7.5.10.3. Rest of Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
7.5.10.4. Rest of Asia Pacific Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
8. Middle East and Africa Global Pharmacovigilance and Drug Safety Software Market Size and Forecast by Segmentation (in USD Billion) 2025-2032
8.1. Middle East and Africa Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
8.2. Middle East and Africa Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
8.3. Middle East and Africa Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
8.4. Middle East and Africa Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
8.5. Middle East and Africa Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Country (2025-2032)
8.5.1. South Africa
8.5.1.1. South Africa Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
8.5.1.2. South Africa Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
8.5.1.3. South Africa Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
8.5.1.4. South Africa Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
8.5.2. GCC
8.5.2.1. GCC Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
8.5.2.2. GCC Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
8.5.2.3. GCC Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
8.5.2.4. GCC Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
8.5.3. Nigeria
8.5.3.1. Nigeria Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
8.5.3.2. Nigeria Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
8.5.3.3. Nigeria Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
8.5.3.4. Nigeria Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
8.5.4. Rest of ME&A
8.5.4.1. Rest of ME&A Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
8.5.4.2. Rest of ME&A Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
8.5.4.3. Rest of ME&A Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
8.5.4.4. Rest of ME&A Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
9. South America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast by Segmentation (in USD Billion) 2025-2032
9.1. South America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
9.2. South America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
9.3. South America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
9.4. South America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
9.5. South America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Country (2025-2032)
9.5.1. Brazil
9.5.1.1. Brazil Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
9.5.1.2. Brazil Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
9.5.1.3. Brazil Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
9.5.1.4. Brazil Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
9.5.2. Argentina
9.5.2.1. Argentina Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
9.5.2.2. Argentina Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
9.5.2.3. Argentina Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
9.5.2.4. Argentina Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
9.5.3. Rest Of South America
9.5.3.1. Rest Of South America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Funtionality (2025-2032)
9.5.3.2. Rest Of South America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Delivery Mode (2025-2032)
9.5.3.3. Rest Of South America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by Component (2025-2032)
9.5.3.4. Rest Of South America Global Pharmacovigilance and Drug Safety Software Market Size and Forecast, by End User (2025-2032)
10. Company Profile: Key Players
10.1. Oracle Corporation
10.1.1. Company Overview
10.1.2. Business Portfolio
10.1.3. Financial Overview
10.1.4. SWOT Analysis
10.1.5. Strategic Analysis
10.1.6. Scale of Operation (small, medium, and large)
10.1.7. Details on Partnership
10.1.8. Regulatory Accreditations and Certifications Received by Them
10.1.9. Awards Received by the Firm
10.1.10. Recent Developments
10.2. IQVIA Holdings Inc.
10.3. ArisGlobal LLC
10.4. Veeva Systems Inc.
10.5. SAS Institute Inc.
10.6. Accenture PLC
10.7. Cognizant Technology Solutions Corporation
10.8. Wipro Limited
10.9. Clarivate PLC
10.10. Ennov SAS
10.11. AB Cube SARL
10.12. Sarjen Systems Pvt. Ltd.
10.13. EXTEDO GmbH
10.14. Pegasystems Inc.
10.15. Honeywell International Inc.
10.16. Indegene Pvt. Ltd.
10.17. United BioSource LLC
10.18. TriNetX LLC
10.19. BaseCon AS
10.20. Max Application Srl
10.21. Genpact Limited
10.22. ICON plc
10.23. Parexel International Corporation
10.24. Ergomed Group
10.25. Quanticate International Ltd.
11. Key Findings
12. Industry Recommendations
13. Global Pharmacovigilance and Drug Safety Software Market: Research Methodology
14. Terms and Glossary