Newborn Screening Market Size by Product Type, Test Type, Technology, Test Type and Region – Segment-Level Market Assessment, Growth Opportunity Analysis, Competitive Mapping & Forecast to 2032
Overview
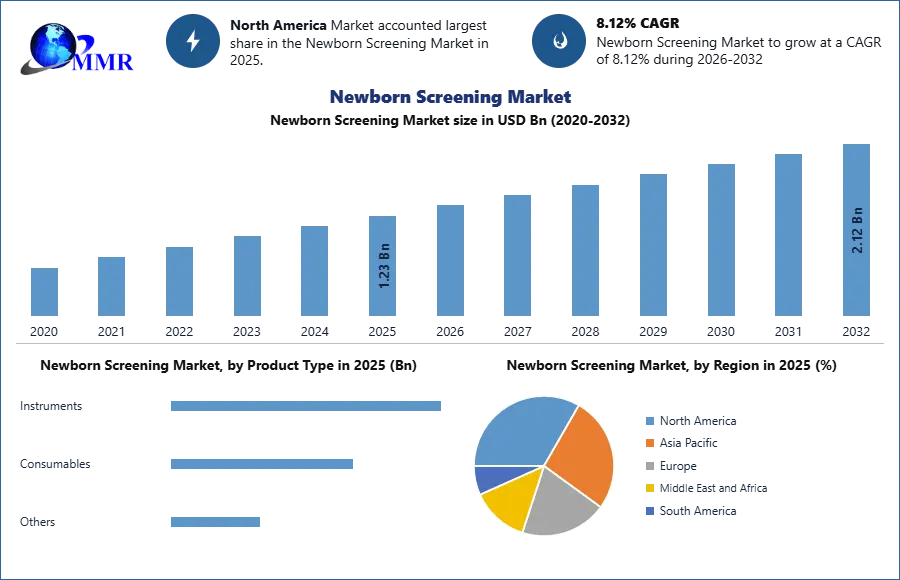
The Global Newborn Screening Market was valued at USD 1.23 Bn in the year 2025 and is expected to reach USD 2.12 Bn by 2032 with a growing CAGR of 8.12 % over the forecast period of 2026-2032.
Newborn Screening Market Overview:
The newborn screening program is a vital sector of healthcare, which focuses on early detection and diagnosis of genetic, metabolic, and congenital disorders in infants. Newborn screening programs typically involve collecting blood, or tissue samples from newborns shortly after birth and analyzing them for markers of various diseases. The screening process varies by region and healthcare system but often targets disorders such as phenylketonuria (PKU), cystic fibrosis, congenital hypothyroidism, and sickle cell disease. The factors driving the newborn screening market include increasing birth rates globally, rising awareness about the benefits of early diagnosis, and advancements in screening technologies with technological innovations, such as TMS, next-generation sequencing (NGS), and molecular diagnostics, which have significantly enhanced the accuracy and scope of newborn screening. These technologies allow for the detection of disorders with improved specificity which supports early detection and personalized treatment options in infants. Many countries have established national or regional newborn screening programs, mandating specific screening tests for all newborns to ensure uniformity and consistency in healthcare sector. Hearing screening test, detects hearing impairments early is a prominent segment in the market supporting detection and diagnosis of hearing disorders.
Some of the challenges in the newborn screening market are the high cost of advanced screening technologies, variability in screening protocols and guidelines across regions, and ethical considerations related to consent, and privacy of the patient information. The North America newborn screening market held the largest share in 2023 owing to standard healthcare infrastructure, research facilities and government bodies like CDC, AAP, HRSA, GARD and ACHDNC that mandate newborn screening test after the birth of infants. In US, 4 million babies are screened annually of which more than 12,500 are found to have severe disorders that are left untreated and 31 cases of serious disorders. The partnerships between healthcare providers, screening laboratories, and regulatory bodies are essential for driving innovation, expanding market reach, and enhancing the effectiveness of newborn screening programs.
Overall, the newborn screening market is evolving with a market potential of 1.75 Bn over the forecast period along with advancements in technology, healthcare infrastructure, and increasing global awareness. These factors collectively contribute to the growth and development of newborn screening platforms worldwide, with highlighting its critical role in early detection of diseases and improved health outcomes.
 To know about the Research Methodology:-Request Free Sample Report
To know about the Research Methodology:-Request Free Sample Report
Newborn Screening Market Dynamics:
Increasing birth rate and population growth are driving the newborn screening market:
The increasing number of births worldwide directly affects the demand for newborn screening tests as government of many countries have mandated newborn screening programs, which boost the market growth by increasing the number of tests performed. The innovations in screening technologies, such as tandem mass spectrometry (MS/MS) and next-generation sequencing (NGS), enhance the accuracy of newborn screening, thereby driving the newborn market growth. The growing awareness about the benefits of early detection of congenital disorders and genetic diseases drives the adoption of newborn screening tests. Developments in genetic research and in the field of genomics contribute to the expanding range of conditions that are screened in newborns at early stage. The improvements in healthcare infrastructure have lead to increased access to newborn screening services. The increasing number of genetic disorders and chronic diseases among newborns has necessitated the early detection in newborn, thereby driving the demand for newborn screening market. The integration of newborn screening technologies with other diagnostic systems facilitates the processes and enhances market growth. According to Indian Pediatric Academy, occurrence of congenital heart disease in children is high and every year more than 200000 children are born with congenital heart disease.
The cost of screening and awareness among the population are the newborn screening market restraints:
The high cost associated with advanced screening technologies, such as next-generation sequencing (NGS) and tandem mass spectrometry limits the accessibility, particularly in remote regions with constrained healthcare budgets. The lack of awareness among healthcare providers, parents, and caregivers about the importance of newborn screening and the availability of screening programs results in low number of screening rates, delaying diagnosis and intervention for infants with genetic disorders. Certain ethical concerns related to consent for screening, storage of genetic information, and the implications of false results impose challenges to the adoption and acceptance of newborn screening programs. The inadequate healthcare infrastructure and limited availability of trained personnel, especially in rural areas, affects the implementation and expansion of screening programs.
Opportunities in the newborn screening market:
The expansion of test panels is creating an opportunity to expand the range of conditions screened in newborns apart from the traditional metabolic disorders. The advances in genetic testing and genomics allow for the inclusion of more rare genetic conditions and diseases, that provide broad screening panels. The continuous advancements in newborn screening technologies, such as next-generation sequencing (NGS), microarray analysis, and digital PCR, hold opportunities to improve the accuracy and speed of newborn screening tests. These technologies enable more precise diagnosis and early intervention of any disease occurring in a newborn. The development of point-of-care testing (POCT) devices for newborn screening offers opportunities to simplify the screening process, reduce turnaround times, and improve the accessibility of screening tests in resource-limited and remote areas. According to UN Children Fund, India records 21 million births every year and this high birth rate demands for newborn screening tools and tests availability across public and private hospitals and childcare centres.
Newborn Screening Market Segmentation:
Based on test, the dry blood spot test type of segment has the largest market share of newborn screening market over the forecast period as there are product launches for the screening of dry blood spot tests. The dry blood spot test kits are used for more than 40+ conditions associated with newborn screening. The initiatives taken by government authorities to promote blood tests are boosting the growth of the segment. The hearing segment also holds a significant market share as hearing screening test is a critical component of newborn screening programs worldwide healthcare guidelines in many countries. Early detection of hearing loss does timely intervention and diagnosis, which improves the outcomes in infants. The prominence of the hearing screening segment highlights its essential role in newborn care and the overall newborn screening market.
Based on end user, the hospital segment is expected to dominate the newborn screening market over the forecast period. These healthcare facilities are at the primary places of newborn screening programs, where screening tests are routinely conducted after birth. Hospitals play a vital role in implementing screening protocols, collecting samples, conducting tests, interpreting results, and initiating the diagnostic interventions as required. They serve as the primary point of care for newborns and their families, ensuring that screening results are communicated effectively and appropriate follow-up care is provided. Healthcare providers in hospitals and clinics collaborate with laboratories and screening centres to ensure the integration of newborn screening into routine healthcare practices, thereby contributing significantly to the adoption and expansion of newborn screening programs.
Newborn Screening Market, Regional Insights:
The North America newborn screening market held the largest market share in 2025 which is attributed to factors such as well-established healthcare infrastructure, awareness about the benefits of newborn screening, government initiatives mandating screening programs, and the presence of advanced healthcare technologies and research facilities. The United States is the largest market for newborn screening, which accounts for a significant portion of the region's market share. The U.S. has a robust healthcare system with broad newborn screening programs that cover a range of genetic disorders and other conditions. There is also a strong emphasis on early detection and diagnosis, which is supported by funding agencies for newborn screening initiatives. According to NIH, 4 million infants are born each year in US, and 25-30% of them undergo newborn screening process and other states report show participation upto 95% in newborn screening test. Other regions, such as Europe and Asia-Pacific, contribute significantly to the global newborn screening market. The European countries like Germany, France, and the UK have well-established screening programs, although the market scenario varies by country due to differences in healthcare policies. In Asia-Pacific, countries such as China, Japan, and India are expanding their newborn screening centres, which is driven by increasing healthcare expenditure, rising awareness, and improving the existing healthcare infrastructure. India and China being the two most populated countries within Asia-Pacific are driving the growth of the market in the region.
Contributions of regulatory bodies for newborn screening test:
1. The Texas Department of Health and Human Services began its Newborn Screening Program (NBS) in 2021 for detection of Spinal Muscular Atrophy (SMA). SMA is a rare neuromuscular disorder that results in the loss of motor neurons and progressive muscle wasting and is among the leading genetic causes of death in infants and toddlers.
2. The American Academy of Pediatrics (AAP) has strongly recommended newborn screening as the best way to detect a health problem at an early stage that can cause serious lifelong disabilities and, in some cases, end a child's life. More than 50 states of US have adopted the screening programs under the guideline from AAP.
Competitive Landscape of Newborn Screening Market:
The competitive landscape of the newborn screening market is characterized by a presence of market players including healthcare providers, diagnostic laboratories, and regulatory bodies. Key participants in this market include major diagnostic companies like PerkinElmer, Bio-Rad Laboratories, and GE Healthcare, which offer a range of screening tests and technologies. These companies focus on enhancing the sensitivity, and throughput of screening assays, often leveraging advanced technologies such as TMS, next-generation sequencing (NGS), and molecular diagnostics. There is significant competition among technology providers in developing innovative point-of-care testing (POCT) solutions that aim at improving accessibility of screening results. Regulatory bodies play a role in modifying the competitive landscape by setting guidelines and standards for newborn screening protocols and approving new screening tests. Collaborations and partnerships between industries and healthcare providers are common strategies to drive innovation and expand market reach in the evolving healthcare sector and increasing demand for comprehensive newborn screening solutions globally.
Recent Industry Developments (2025–2026)
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 17 February 2026 | Danaher Corporation | The company entered into a definitive agreement to acquire Masimo Corporation, integrating leading pulse oximetry technology into its diagnostics portfolio. | This acquisition consolidates market leadership in critical congenital heart disease (CCHD) screening for newborns. |
| 12 November 2025 | Waters Corporation | Waters announced a co-marketing agreement with Enfanos to connect enzyme activity assays with the Xevo TQ-S micro IVD mass spectrometry platform. | The partnership optimizes workflows for detecting lysosomal storage disorders (LSDs) and inborn errors of metabolism. |
| 06 October 2025 | Quantabio | The company launched the sparQ Lysis Kit, a specialized solution for extracting high-quality DNA from dried blood spots for next-generation sequencing. | This development significantly reduces the time-to-result for genomic-based newborn screening panels. |
| 18 August 2025 | Precedence Research | Market records confirmed the commercial rollout of AI-enhanced eye imaging systems specifically designed for neonatal retinopathy screening. | The introduction of automated detection tools addresses the shortage of pediatric ophthalmologists in emerging markets. |
| 12 March 2025 | Bio-Techne | The company launched the AmplideX Nanopore Carrier Plus Kit in collaboration with Oxford Nanopore, targeting heritable genetic disorders. | This provides a scalable sequencing alternative to traditional biochemical assays for comprehensive newborn panels. |
| 15 January 2025 | QIAGEN N.V. | QIAGEN partnered with Genomics England to provide curated genomic content for a project sequencing the genomes of 100,000 newborns. | This initiative establishes a new clinical standard for rapid variant interpretation in public health screening programs. |
Newborn Screening Market Scope: Inquire before buying
| Newborn Screening Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 1.23 USD Bn |
| Forecast Period 2026-2032 CAGR: | 8.12% | Market Size in 2032: | 2.12 USD Bn |
| Segments Covered: | by Product Type | Instruments Consumables Others |
|
| by Test Type | Dry blood spot test Hearing screening test Heart screening test Others |
||
| by Technology | Tandem Mass Spectroscopy (TMS) Immunoassays Hearing screening technology Others |
||
| by Test Type | Hospitals Clinical Laboratories |
||
Newborn Screening Market, by Region:
North America (United States, Canada and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan and Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A)
South America (Brazil, Argentina Rest of South America)
Key players/ competitors profile covered in brief in Newborn Screening Market report in strategic perspective
- PerkinElmer, Inc.
- Natus Medical Incorporated
- Trivitron Healthcare
- Waters Corporation
- Bio-Rad Laboratories, Inc.
- Agilent Technologies, Inc.
- GE HealthCare Technologies Inc.
- Masimo Corporation
- Danaher Corporation
- Thermo Fisher Scientific Inc.
- Medtronic plc
- Revvity
- Baebies, Inc.
- Zentech S.A.
- Demant A/S
- AB Sciex LLC
- Hill-Rom Holdings, Inc.
- Luminex Corporation
- MP Biomedicals
- Zivak Technologies
- Enfanos
- BioMedomics, Inc.
- Synergy Medical Systems LLP
- Quantabio
- QIAGEN N.V.
