Lung Cancer Diagnostics Market – Global Market Size, Strategic Growth Drivers, Risk Assessment Framework, Regulatory Landscape Review, Competitive Intensity Mapping & Long-Term Industry Outlook to 2032
Overview
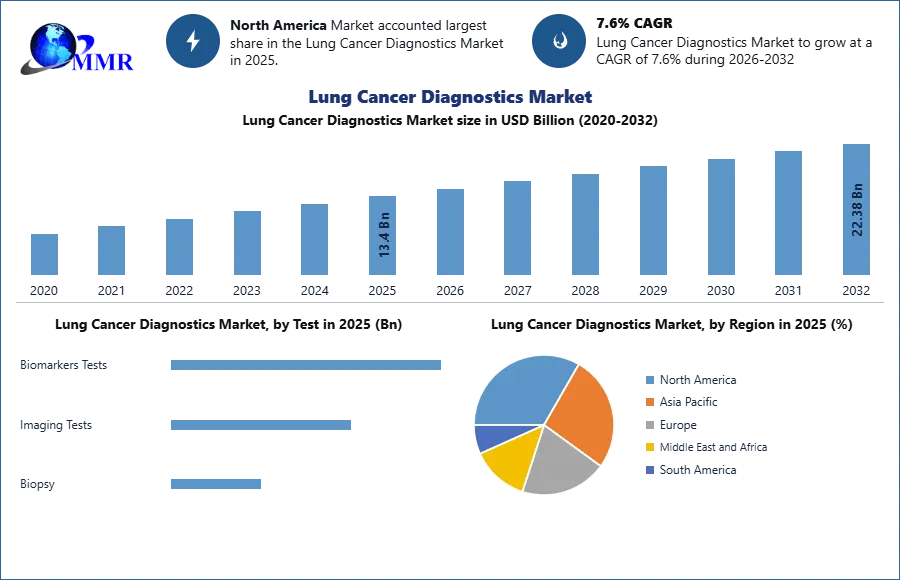
Lung Cancer Diagnostics Market is expected to reach USD 22.38 Bn. at a CAGR of 7.6% during the forecast period 2032.
The Lung Cancer Diagnostics market report is a comprehensive analysis of the industry, market, and key players. The report has covered the market by demand and supply-side by segments. The global Lung Cancer Diagnostics report also provides trends by market segments, technology, and investment with a competitive landscape.
Global Lung Cancer Diagnostics Market Overview:
The global lung cancer diagnostics market was valued at USD 13.4 billion in 2025 and is expected to reach USD 22.38 billion by 2032, at a CAGR of 7.6% during the forecasting period 2026-2032.
The lung cancer that begins in the lungs most often occurs in the people who smoke more. Some of the symptoms of lung cancer are caught with blood, chest pain, and weight loss. There are a lot of treatments available in hospitals. Still, the cost of that treatment is too high, so normal people do not afford that cost, so the government is decided to make government hospitals where anyone can give treatment free of cost.
To know about the Research Methodology:- Request Free Sample Report
Global Lung Cancer Diagnostics Market Dynamics:
Increasing the consumption of tobacco and rising cases of cervical cancer are increasing cancer cases, bolstering diagnostics prospects. Cervical cancer is the most common viral infection of the reproductive tract. Public and private associations, a joint effort to further develop a framework of analytic imaging communities, is driving a cellular breakdown in the lung of the demonstrative market. By the government and the health organization, they are trying to spread awareness about cancer globally. So it helps those people who show the same symptoms of cancer.
The high cost of a diagnostic system is restraining the growth of the market. Moreover due to insufficient experience people, it causes a challenge for the market.
Global Lung Cancer Diagnostics Market Segment Analysis:
The Global Lung Cancer Diagnostics Market is segmented into test, end-user, and indication.
On the basis of test, global lung cancer diagnostics market is sub-segmented biomarkers tests, imaging tests, and biopsy. In the Biomarkers test, it includes some test and they are EGFR Mutation Test, KRAS Mutation Test, ALK Test, HER2 Test, and others. In imaging test, also includes Computed Tomography (CT) Scan, Positron Emission Tomography (PET) Scan, Chest X-RAY, and others. The Biopsy segment also includes tests like Needle Biopsy, Bronchoscopy Biopsy, Open Biopsy, and others. Among all the tests Imaging Tests is a more important test for diagnosis of lung cancer because it is very easy to understand the position and the size of the tumor. Imaging Tests, one of the segments analyzed in the report, is projected to grow at 8% CAGR to reach US$2.2 Billion by the end of the analysis period.
Based on an end-user, the global lung cancer diagnostics market is sub-segmented into hospital associated labs, independent diagnostic laboratories, cancer research institutes, and others.
According to the MMR reports, the hospital segment held the largest shares in 2025. As it delivers well-organized, documented, retrieved, and logistical procedures. Hence, the hospital segment is expected to show fast growth due to rising the number of private hospitals, including many facilities in the hospital, quick treatment, and professional doctors. So many consumers are turning towards hospitals.
Based on an indication, lung cancer is sub-segmented into non-small-cell lung cancer and small-cell lung cancer. Small cell cancers vary, depending on the expression of specific genes and cells. Generally, small cell cancer is more aggressive than non-small cell lung cancer. Non-small cell lung cancer (NSCLC) was identified as the largest segment, owing to the availability of novel diagnostic testing, detecting large tumor growths, and the most protuberant form in terms of occurrence, and accounting for nearly 81% of the disease. The reason behind that is the cancer virus directly attacks cells and stops flowing the blood, so due to less blood supply, people die. Nowadays, new technologies and treatments are going on, but the cost of that treatment is high.
Global Lung Cancer Diagnostics Market Regional Insights:
North America held the largest share of the global lung cancer diagnostics market in 2025, and it can be attributed to the factors such as the increasing prevalence of cancer, rising recoveries in North America, and the introduction of favorable reimbursement policies, and there is some rule by the government in the US and Canada, due to all these North America lung cancer diagnostics market is growing. The data have provided by the National Cancer Institutes, which estimated the number of cases of cancer in the United Sated was 1.7 million in the year 2025, and it is expected to reach 20.3 million by 2032.
APAC has the second-largest market share in the year 2025. The Asia Pacific is expected to show a fastest growth rate in the global lung cancer diagnostics market in the last few years due to government policies and a rise in awareness of the manufactures about lung cancer diagnostics and their benefits.
Recent Industry Developments (2025–2026):
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 18 August 2025 | Daiichi Sankyo and Merck & Co. | The companies received U.S. FDA Breakthrough Therapy Designation for ifinatamab deruxtecan (I-DXd) for extensive-stage small cell lung cancer (SCLC). | This highlights the shift toward antibody-drug conjugates as critical diagnostic-driven therapy options for difficult-to-treat lung cancers. |
| 15 July 2025 | Thermo Fisher Scientific | The U.S. FDA cleared the Oncomine Dx Express Test, an NGS-based companion diagnostic for identifying EGFR exon 20 insertion mutations. | Enables rapid in-house genomic profiling with results in under 24 hours, significantly reducing diagnostic turnaround times. |
| 22 May 2025 | Roche Diagnostics | The VENTANA MET (SP44) RxDx Assay received FDA approval as a companion diagnostic to identify NSCLC patients for telisotuzumab vedotin-tllv therapy. | Strengthens Roche's leadership in personalized medicine by expanding the range of actionable protein overexpression biomarkers. |
| 14 April 2025 | Roche | The FDA granted Breakthrough Device Designation to the VENTANA TROP2 RxDx Device, an AI-powered computational pathology assay. | Accelerates the integration of artificial intelligence in evaluating complex biomarkers for non-small cell lung cancer (NSCLC). |
| 12 February 2025 | Imagene and Tempus | The companies announced a collaboration to develop an AI-driven multi-gene panel capable of predicting NSCLC biomarkers directly from biopsy images. | Streamlines clinical workflows by allowing near-instant biomarker profiling without the wait times typical of traditional sequencing. |
The objective of the report is to present a comprehensive analysis of the global Lung Cancer Diagnostics market to the stakeholders in the industry. The past and current status of the industry with the forecasted market size and trends are presented in the report with the analysis of complicated data in simple language. The report covers all the aspects of the industry with a dedicated study of key players that include market leaders, followers, and new entrants.
PORTER, PESTEL analysis with the potential impact of micro-economic factors of the market have been presented in the report. External as well as internal factors that are supposed to affect the business positively or negatively have been analysed, which will give a clear futuristic view of the industry to the decision-makers. The report also helps in understanding the global Lung Cancer Diagnostics market dynamics, structure by analyzing the market segments and project the global Lung Cancer Diagnostics market size. Clear representation of competitive analysis of key players by product, price, financial position, product portfolio, growth strategies, and regional presence in the global Lung Cancer Diagnostics market make the report investor’s guide.
Global Lung Cancer Diagnostics Market Scope: Inquire before buying
| Lung Cancer Diagnostics Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 13.4 USD Billion |
| Forecast Period 2026-2032 CAGR: | 7.6% | Market Size in 2032: | 22.38 USD Billion |
| Segments Covered: | by Test | Biomarkers Tests Imaging Tests Biopsy |
|
| by Indication | Non Small-cell Lung Cancer Small-cell Lung Cancer |
||
| by End User | Hospital Associated Labs Independent Diagnostic Laboratories Cancer Research Institutes Others |
||
Global Lung Cancer Diagnostics Market, by Region
North America (United States, Canada and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan and Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A)
South America (Brazil, Argentina Rest of South America)
Key Players / Competitors Profiles Covered in Brief in Global Lung Cancer Diagnostics Market Report in Strategic Perspective:
- F. Hoffmann-La Roche Ltd
- Thermo Fisher Scientific Inc.
- Illumina, Inc.
- Agilent Technologies, Inc.
- QIAGEN N.V.
- Abbott Laboratories Inc.
- Bio-Rad Laboratories, Inc.
- NeoGenomics Laboratories
- bioMérieux SA
- Myriad Genetics, Inc.
- AstraZeneca plc
- Sanofi S.A.
- Johnson & Johnson
- Siemens Healthineers AG
- Danaher Corporation
- Guardant Health, Inc.
- Veracyte, Inc.
- Natera, Inc.
- GE HealthCare
- MedGenome
- NanoString Technologies, Inc.
- Oncocyte Corporation
- Boditech Med, Inc.
- INOVIQ
- Biocartis
