Allogeneic Cell Therapy Market by Type, Therapeutic Area, End User and Region - Global Market Size Estimation, Industry-Wide Analysis, Competitive Landscape Assessment & Long-Term Forecast to 2032
Overview
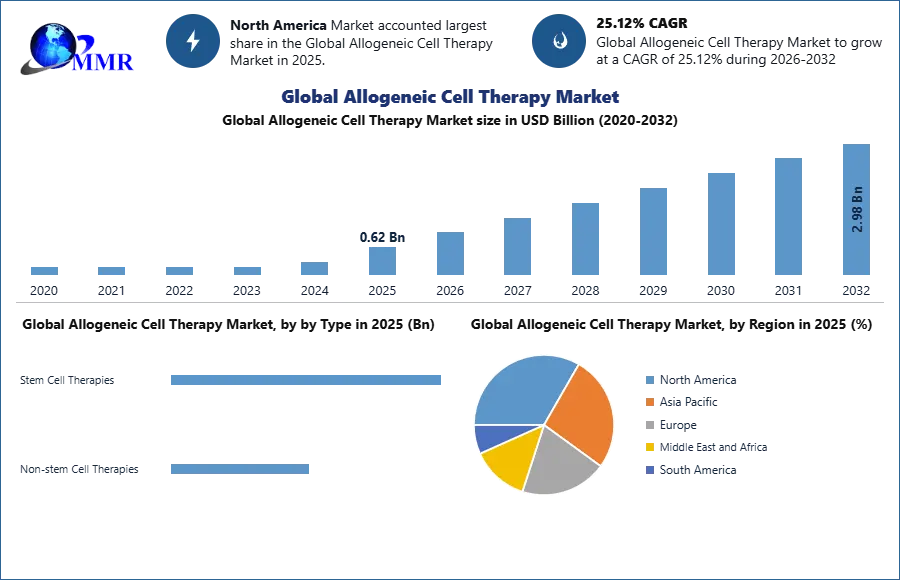
Global Allogeneic Cell Therapy Market size was valued at USD 0.62 Bn in 2025 and the total Allogeneic Cell Therapy Market revenue is expected to grow by 25.12% from 2025 to 2032, reaching nearly USD2.98 Bn by 2032.
Allogeneic Cell Therapy Market Overview
Allogeneic cell therapy is form of advanced regenerative medicine in which living cell, derived from healthy donor are processed, expanded and administered to recipient to treat or cure disease.
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
Global allogeneic cell therapy market has been experiencing robust growth driven by increasing demand for advanced cancer treatment, autoimmune therapies and regenerative medicine supported by favourable regulatory policies and rising investment in cell-based research. North America dominated allogeneic cell therapy market fuelled by cutting edge healthcare infrastructure, strong biotech R&D and high adoption in clinical application with innovation leader like Allogene Therapeutics, Mesoblast and Cellectis pioneering next generation therapies such as off shelf CAR-T cells, mesenchymal stem cell (MSC) therapies and gene edited allogeneic product. Market benefit from diverse application like oncology leverage allogeneic therapy for scalable cancer treatment, autoimmune disease utilizes them for immune modulation and regenerative medicine applies them for tissue repair. This dynamic landscape highlight how allogeneic cell therapies are evolving with breakthrough technologies, improved manufacturing scalability and enhanced safety profile meeting modern healthcare demand while aligning with precision medicine goal and costeffective therapeutic solution.
Allogeneic Cell Therapy Market Dynamics
Scalability & Accessibility to Boost Allogeneic Cell Therapy Market Growth
The key driving factor in Allogeneic Cell Therapy Market has been its use to wider audience. A wider range of patients can be treated with allogeneic cell treatments irrespective of patient's age, health status, or history of previous diseases and also the cells can be used in a larger population because they are not taken from their own body. By doing this, the pool of possible patients is widened and allogeneic cell therapy industry becomes more valuable.
Purity and Safety to Create Challenges in Allogeneic Cell Therapy Market
Purity of the sample is utmost challenging in Allogeneic Cell Therapy market, as any discrepancy in that can cause many harmful effects. Proper screening, testing, and quality control processes are necessary to minimize the risk of transmitting infectious diseases and genetic abnormalities associated with donor cells. Ethical concerns regarding use of donor cells, equitable access, and privacy of donor information all should be properly documented before use, to successfully implement the therapy.
So, it becomes a crucial challenge for growth of the key players. Also, upon inserting the new and healthy cell, there are chances that the cells can damage the healthy tissues and not get familiar with the host environment. Thus, the damage can be irreparable and can cause huge problems. Cost is another huge concern especially when a new firm is entering the Allogeneic Cell Therapy demand. As this process is new, its cost is very expensive which act as a challenge for companies. Thus, curtailing this cost and simplifying the complexity of manufacturing and quality control can be a huge task for,market. High manufacturing cost of Allogeneic cell therapy and potential risk involved in its use can be two crucial factors which can hamper the future forecast and deprive the market of its right potential.
Allogeneic Cell Therapy Market Segment Analysis
Based on Type, Allogeneic Cell Therapy Market is segmented into Stem Cell and Non-Stem Cell Therapies. Stem cell and Non stem cell are the two types. In 2024, the stem cell therapies market had the greatest share. Diseases that are most frequently treated with allogeneic stem cell treatment include chronic illnesses such blood malignancies, leukaemia, and lymphoma as well as specific blood or autoimmune disorders. Non-Stem therapies are generally involving somatic cells that are isolated from human, administered to patients for cure and then for treatment of diseases
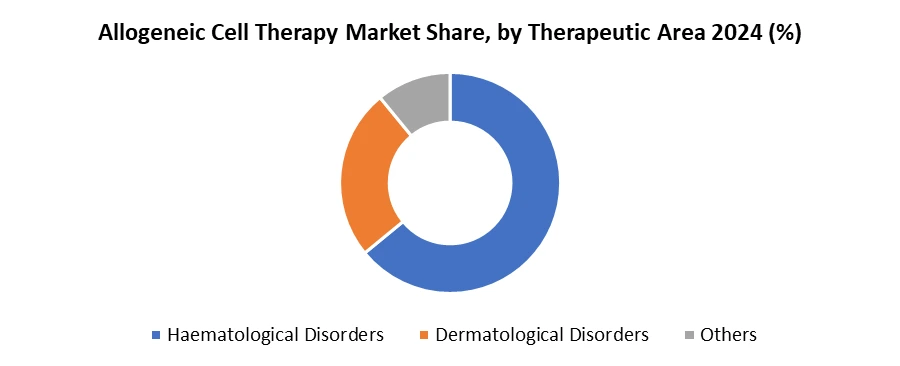
Based on Therapeutic Area, Allogeneic Cell Therapy Market is segmented into Haematological disorders, Dermatological Disorders, etc. Haematological disorders are mainly concerned with blood and blood forming organs and significant development of Allogeneic cell therapy is surrounded near immunology. Thus, haematological diseases have highest market percentage which is nearly 60% of the total segmentation. Many haematological cases have been cured because of using such therapies and thus contains a larger part of the segment. Dermatological disorders are not so common and do not have much of a market share as compared to haematological disorders.
Allogeneic Cell Therapy Market Regional Insights
North America Dominated Allogeneic Cell Therapy Market in 2025 and is expected to dominate during forecast period (2026-2032).
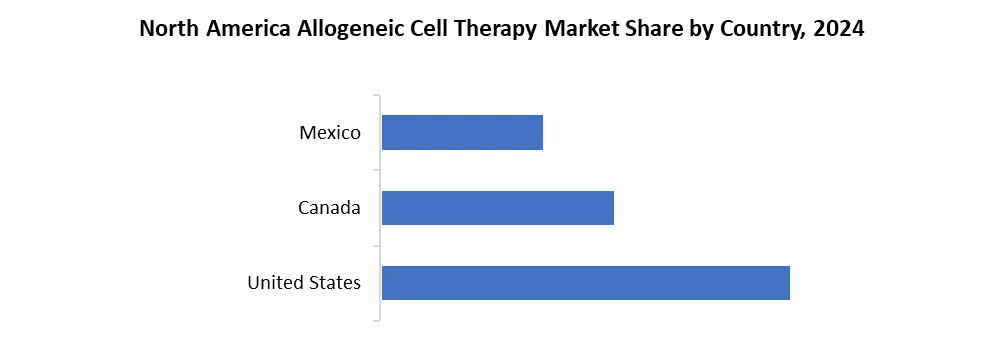
North America led the Allogeneic Cell Therapy market and occupies around nearly 78% of market share in the total market. US leads the way with highest market share amongst the other countries.US big players have contributed thoroughly in this market growth. Adaptimmune Therapeutics, Bluebird Bio, CARsgen Therapeutics are some of the companies with highest contributions. Also, high investments in research and analysis have already increased growth potential of the Allogeneic Cell Therapy industry.
Asia Pacific is currently at a steady market share and is forecasted to grow at a rapid rate which is because of increased population, higher demand and well-developed technology. The increased healthcare sector has directly improved the investments and also the internal structure of the pharma and healthcare market. Countries such as China, Japan, South Korea, India, and Singapore are key contributors to the single-use filtration assembly market. China leads the Allogeneic Cell Therapy market and has one of the largest markets in the world.
Europe has a significant market share in Allogeneic Cell Therapy industry and falls just behind North America. Guidelines and restrictions are provided by the European Medicines Agency (EMA) to guarantee the efficacy, effectiveness, and safety of these treatments. The research and commercialization of allogeneic cell treatments in Europe have been helped by the formation of the Advanced Therapy Medicinal Products (ATMP) regulatory framework. Thus Europe has a huge Allogeneic Cell Therapy market which has the potential to become the leading market player of Allogeneic Cell Industry.
The Middle East is an emerging sector with many opportunities and risk. Saudi Arabia, UAE, Qatar have made significant investments to build a sustainable place for biotechnology and research. In Saudi Arabia, the government's Vision 2030 plan aims to diversify the economy and develop the healthcare sector. Other regions of UAE like Dubai, Qatar have also started making healthcare and biotechnology hub, attracting international biotechnology and pharmaceutical companies and research organizations.
South America expresses a steady market share and is mainly constricted to bigger countries like Brazil, Argentina. Argentina has focused on innovation, research and development, and quality manufacturing process and ensured that they expand their Allogeneic Cell Therapy market share. Columbia has been investing in healthcare from past decade, which gives it an opportunity to have similar market share as of that of Brazil and Argentina. Other countries like Peru, Chile are also slowly accepting the changes of single use technologies
Allogeneic Cell Therapy Market Competitive Analysis
Allogene Therapeutics is pioneer in off the shelf allogeneic CAR-T therapies competing with leader like Cellectis (France), Precision BioSciences (US) and CRISPR Therapeutics (Switzerland) in gene edited allogeneic space. The company’s lead candidate, ALLO-501A (for lymphoma) positions it against autologous CAR-T giants (e.g., Gilead/Kite, Novartis) and emerging allogeneic rivals like Atara Biotherapeutics (EBV-targeted therapies). Allogene’s edge lies in its FasT CAR™ platform enabling faster manufacturing and scalability but it face challenges from CRISPR based allogeneic therapies (e.g., CRISPR/Vertex’s CTX110) and regulatory hurdles tied to graft rejection risks. While partnerships (e.g., with Servier) bolster its pipeline, competition intensifies as Big Pharma (Pfizer, Bayer) invests in allogeneic cell therapy startups, threatening Allogene’s first-mover advantage in the race for off the shelf solutions.
Allogeneic Cell Therapy Market Recent Development:
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 06 March 2026 | Sumitomo Pharma | The company obtained conditional marketing authorization in Japan for AMCHEPRY®, an allogeneic iPS cell-derived dopaminergic neural progenitor cell product for Parkinson's disease. | This approval represents a major milestone for iPS cell-based therapies, establishing a new therapeutic path expected to generate significant long-term sales. |
| 10 March 2026 | Legend Biotech | The company reported first-in-human clinical results for LUCAR-G39D, an allogeneic CAR-T therapy, while reaching a milestone of over 10,000 patients treated with its cell therapy portfolio. | These results validate the expansion into in vivo and allogeneic programs, broadening the company’s reach beyond autologous treatments. |
| 06 March 2025 | Neurotech Pharmaceuticals | The FDA approved Encelto (revakinagene taroretcel-lwey), the first allogeneic, encapsulated cell therapy implant for macular telangiectasia type 2. | This novel sustained-delivery platform provides a critical treatment option for a previously untreatable retinal condition. |
| 21 January 2025 | Grafapex | The FDA approved Grafapex (treosulfan) for use in combination with fludarabine as a preparative regimen for allogeneic hematopoietic stem cell transplantation. | This provides a standardized, approved conditioning protocol to improve outcomes for patients undergoing life-saving allogeneic transplants. |
Allogeneic Cell Therapy Market Key Trends
• AI-Driven Optimization of Donor Matching & Manufacturing Workflows: AI-driven optimization of donor matching and manufacturing workflows is an emerging trend enabling more precise allogeneic cell therapy production by predicting immune compatibility, reducing batch failures, and accelerating time-to-market through intelligent automation.
• Non-Viral Gene Editing for Allogeneic Cell Engineering: Non-viral gene editing for allogeneic cell engineering is a rising trend that leverages safer, faster, and more scalable methods—like mRNA or nanoparticle delivery—to modify cells without the complexity and risks of viral vectors.
• Rise of Universal iPSC-Derived Immune Cells Beyond CAR-T/NK: The rise of universal iPSC-derived immune cells beyond CAR-T/NK marks a next-gen trend in allogeneic cell therapy, enabling scalable, off-the-shelf treatments using versatile cell types like macrophages and γδ T cells for broader therapeutic applications.
Allogeneic Cell Therapy Market Key Developments
• Poseida Therapeutics (US): May 2024: Advanced P-BCMA-ALLO1, an off-the-shelf CAR-T for multiple myeloma, into Phase II trials, showcasing durable responses with its non-viral piggyBac® gene delivery platform.
• Autolus (UK): November 2024: Received FDA Fast Track designation for AUTO8, its allogeneic CAR-T for osteosarcoma, leveraging proprietary TARMAC™ technology to enhance persistence.
• Cellectis (France): September 2024: Partnered with Servier to launch UCART20x22, a dual-targeting allogeneic CAR-T for B-cell malignancies, entering Phase I/II trials with reduced risk of GvHD.
• CRISPR Therapeutics (Switzerland): January 2025: Reported positive Phase II data for CTX112™, an allogeneic CRISPR-edited CAR-T for autoimmune diseases (e.g., lupus), demonstrating 6-month remission in 60% of patients.
• Mesoblast (Australia): March 2025: Secured FDA approval for REXLEMestrocel-L, an allogeneic MSC therapy for chronic heart failure, marking the first allogeneic cell therapy approval for cardiovascular disease.
Allogeneic Cell Therapy Market Scope: Inquire before buying
| Global Allogeneic Cell Therapy Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020-2024 | Market Size in 2025: | 0.62 USD Billion. |
| Forecast Period 2025 to 2032 CAGR | 25.12% | Market Size in 2032: | 2.98 USD Billion |
| Segments Covered: | by Type | Stem Cell Therapies 1 Hematopoietic Stem Cell Therapies 2 Mesenchymal Stem Cell Therapies Non-stem Cell Therapies 1 Keratinocytes & Fibroblast-based Therapies 2 Others |
|
| by Therapeutic Area | Haematological disorders Dermatological disorders Others |
||
| by End User | Hospitals Speciality Clinics Research Organizations |
||
Global Allogeneic Cell Therapy Market, by Region
North America (United States, Canada, and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria, and the Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, ASEAN, Indonesia, Philippines, Malaysia, Vietnam, Thailand, ASEAN, Rest of Asia Pacific)
Middle East & Africa (South Africa, GCC, Nigeria, Rest of ME&A)
South America (Brazil, Argentina, Rest of South America)
Allogeneic Cell Therapy Market, Key Players
1. Allogene Therapeutics Inc.
2. Bluebird Bio Inc.
3. Mustang Bio Inc.
4. Nkarta Therapeutics Inc.
5. Precision BioSciences Inc.
6. Sangamo Therapeutics Inc.
7. WindMIL Therapeutics Inc.
8. Gilead Sciences Inc.
9. Iovance Biotherapeutics Inc.
10. Gamida Cell Ltd.
11. MaxCyte Inc
12. Poseida Therapeutics Inc.
13. Eureka Therapeutics Inc.
14. Atara Biotherapeutics Inc.
15. Adaptimmune Therapeutics plc
16. TC BioPharm Holdings plc
17. Autolus Therapeutics plc
18. Cellectis S.A.
19. Celyad Oncology SA
20. CRISPR Therapeutics AG
21. Mesoblast Limited
22. JCR Pharmaceuticals Co. Ltd.
23. Sumitomo Pharma Co. Ltd.
24. Takeda Pharmaceutical Company Limited
25. Tego Science Inc.
26. Stempeutics Research Pvt. Ltd.
27. Biosolution Co. Ltd.
28. MEDIPOST Co. Ltd.
29. Fate Therapeutics Inc.
30. Century Therapeutics Inc.
Others