Global mRNA vaccines Market size was valued at USD 11.4 Bn in 2022 and is expected to reach USD 21.79 Bn by 2029, at a CAGR of 9.7% over the forecast period.mRNA vaccines Market Overview
mRNA vaccines contain the genetic information in the form of mRNA, which encodes a specific protein. Unlike traditional vaccines that use a weakened or inactivated form of the pathogen, mRNA vaccines do not contain live virus, making them non-infectious. mRNA vaccines are designed and developed more quickly than traditional vaccines. This was evident in their rapid response to the COVID-19 pandemic. Due to the significant impact of the pandemic and the need for mass vaccination, the demand for COVID-19 vaccines, including mRNA vaccines, was substantial. Many governments and organizations placed orders for these vaccines to ensure their populations had access to vaccination, and boosted the mRNA vaccines Market growth. RNA vaccines, such as Pfizer-BioNTech's Comirnaty and Moderna's COVID-19 vaccine, were at the forefront of the global response to the COVID-19 pandemic. These vaccines demonstrated high efficacy rates and were widely used in many countries to curb the spread of the virus.To know about the Research Methodology :- Request Free Sample Report
mRNA vaccines Market Dynamics
Accelerated Development in vaccines to boost the mRNA vaccines Market growth Traditional vaccine development often takes years, but mRNA vaccines are developed and manufactured in a significantly shorter timeframe. The modular nature of mRNA sequences enables quick adaptation to new pathogens, reducing the time required for preclinical and clinical testing. The COVID-19 pandemic underscored the urgency of vaccine development and deployment. mRNA technology's ability to produce vaccines rapidly has positioned it as a critical tool in responding to emerging infectious diseases, which is expected to boost the mRNA vaccines market growth. The swift development of COVID-19 mRNA vaccines showcased their potential to address global health crises efficiently. The flexibility of mRNA technology enables researchers to tailor vaccines to different pathogens by adjusting the mRNA sequence. This adaptability facilitates the creation of vaccines for various diseases, potentially revolutionizing how we respond to diverse health threats. Unlike traditional vaccines that require growing live viruses or complex processes, mRNA vaccines can be produced through cell-free techniques. This streamlined manufacturing process holds the potential to enhance scalability, reduce production bottlenecks, and accelerate global vaccine distribution. The success of mRNA vaccines against COVID-19 has attracted substantial investment from governments, private companies, and philanthropic organizations. This influx of funds has spurred research, development, and innovation within the mRNA vaccine market. mRNA vaccine companies at the forefront of mRNA vaccine development often collaborate with academic institutions, research organizations, and other biotech companies. These partnerships accelerate knowledge exchange and advancement, fostering a collaborative environment within the industry. Beyond infectious diseases, mRNA technology offers a promising avenue for personalized medicine. It could revolutionize cancer treatment by enabling the development of personalized cancer vaccines tailored to an individual's unique genetic makeup. The success of mRNA vaccines in combating the COVID-19 pandemic has bolstered public confidence in the technology's safety and efficacy. This positive perception could influence greater acceptance of mRNA vaccines for other diseases. Efforts are underway to ensure equitable access to mRNA vaccines worldwide. Initiatives focusing on technology transfer and production capacity building could help address disparities in vaccine distribution and contribute to global health security. Manufacturing Complexity to restrain the mRNA vaccines Market growth The streamlined manufacturing process of mRNA vaccines compared to traditional methods, the technology still presents challenges in terms of scalability and large-scale production. Ensuring consistent and high-quality production at a global scale limits the mRNA vaccines market growth. Some mRNA vaccines require extremely low temperatures for storage, posing logistical challenges, especially in regions with limited cold chain infrastructure. The need for specialized storage conditions could hinder distribution and accessibility, particularly in resource-constrained areas. Developing and producing mRNA vaccines can be expensive, which might influence affordability and accessibility, particularly in low-income countries. Cost considerations could impact the equitable distribution of these vaccines. Despite the success of mRNA vaccines against COVID-19, concerns about novel vaccine technologies and misinformation could lead to vaccine hesitancy among certain populations. Building public trust and confidence is essential for the broader acceptance of mRNA vaccines. Regulatory agencies might face challenges in adapting to the unique characteristics of mRNA vaccines. Ensuring that safety and efficacy standards are appropriately met while expediting the regulatory process could be complex. As mRNA vaccine technology becomes more prominent, disputes over patents and intellectual property could arise, potentially slowing down innovation and hindering technology sharing. The long-term safety and efficacy of mRNA vaccines require continued monitoring and research. While the short-term data has been encouraging, ongoing surveillance is essential to assess any potential rare or long-term adverse effects. The emergence of new variants of viruses might necessitate frequent updates to mRNA vaccine formulations. Adapting to evolving pathogens quickly could be a challenge, especially if regulatory approval processes need to be repeated for each new formulation. Ensuring equitable access to mRNA vaccines across all regions and populations is a challenge. Disparities in access could perpetuate health inequalities and hinder global efforts to control infectious diseases.mRNA vaccines Market Regional Insight
North America region dominated the market in 2022 and is expected to hold the largest mRNA Vccines market share over the forecast period. The North American mRNA vaccine industry has undergone remarkable transformation, driven primarily by the development and distribution of mRNA-based vaccines to combat the COVID-19 pandemic. The two prominent players in this market are Pfizer-BioNTech and Moderna, both of which introduced groundbreaking vaccines utilizing messenger RNA technology. The North American mRNA vaccine market is characterized by high demand, leading to a race to secure doses and distribution infrastructure. Governments in the United States, Canada, and other North American countries negotiated contracts with Pfizer-BioNTech and Moderna to secure millions of vaccine doses. These agreements aimed to ensure timely access to vaccines for their populations. The authorization of Pfizer-BioNTech's and Moderna's vaccines for emergency use by regulatory agencies marked a significant milestone in the fight against COVID-19. The United States Food and Drug Administration (FDA) granted Emergency Use Authorization (EUA) to both vaccines in late 2020, allowing for their distribution and administration to priority groups and boost the market growth. The success of mRNA Vaccines against COVID-19 also paved the way for broader applications of mRNA technology. Researchers and pharmaceutical companies began exploring the potential of mRNA Vaccines in addressing other infectious diseases and even certain cancers. This opened up new avenues for innovation in the North American healthcare and biotechnology sectors.The European mRNA vaccine market has been a crucial focal point in the global response to the COVID-19 pandemic, with significant developments in the production, distribution, and administration of mRNA-based vaccines. This market has been dominated by key players such as Pfizer-BioNTech and Moderna, whose mRNA Vaccines have played a pivotal role in the region's efforts to combat the spread of the virus. The European mRNA vaccine industry faced logistical challenges similar to those in other regions. The Pfizer-BioNTech vaccine, for instance, required ultra-cold storage, presenting distribution hurdles, particularly in rural or less-developed areas. Adequate cold chain infrastructure had to be established to ensure the vaccines' stability during transportation and storage. Despite these challenges, Europe made significant progress in its vaccination campaign, with millions of doses administered to its population. Mass vaccination centers, collaboration with healthcare providers, and public awareness campaigns played essential roles in achieving high vaccination rates in many European countries. The success of mRNA Vaccines in Europe also led to increased interest in vaccination research and development within the region. European pharmaceutical companies and research institutions explored the potential of mRNA technology in addressing other infectious diseases, such as influenza, and in developing personalized cancer vaccines.
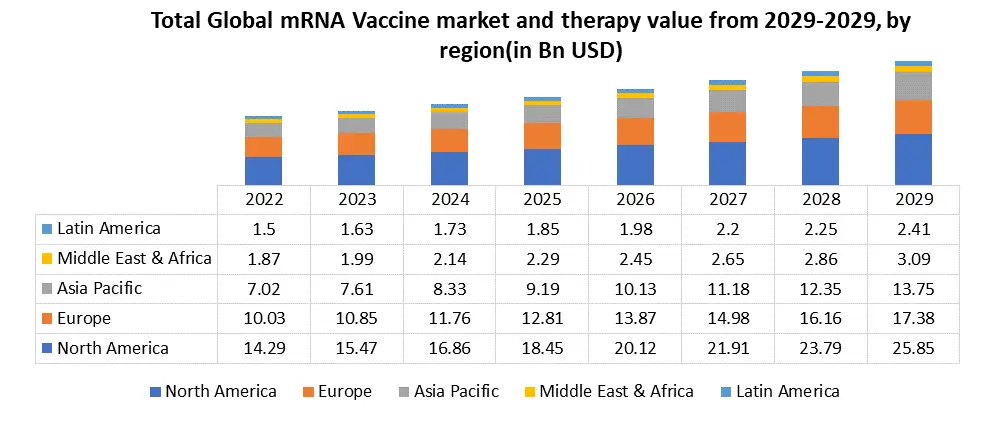
mRNA vaccines Market Segment Analysis
Based on Type, the market is segmented into Self-Amplifying mRNA based vaccines, and Conventional Non- Amplifying mRNA based vaccines. Self-Amplifying mRNA based vaccines segment dominated the market in 2022 and is expected to hold the largest mRNA vaccines market share over the forecast period. SaRNA vaccines are designed to include not only the genetic code for the target antigen but also additional genetic elements derived from RNA viruses. These elements enable the saRNA to replicate within the host cell once it's taken up. This replication lead to the generation of more copies of the target antigen mRNA, resulting in a higher expression of the antigen. The amplified antigen production in saRNA vaccines stimulate more robust immune response compared to conventional non-amplifying mRNA vaccines. This enhanced immune response lead to improved efficacy, especially when it comes to generating strong and long-lasting protection against the target pathogen. SaRNA vaccine technology offers scalability, which is crucial in responding to pandemics or outbreaks. The ability to rapidly produce large quantities of vaccine using saRNA platforms could be vital in addressing emerging infectious diseases and is expected to boost the segment growth in the mRNA vaccines market growth. Based on Distribution Channels, the market is segmented into Government Procurement, Private Healthcare Providers, and Public Health Initiatives. Government Procurement and Public Health Initiatives segment dominated the market in 2022 and is expected to continue the dominance over the forecast period. Governments play a central role in procuring vaccines on behalf of the population. They negotiate contracts with vaccine manufacturers to secure doses for their citizens, often purchasing large quantities. Government procurement ensures equitable distribution of vaccines to the population, with a focus on priority groups and vulnerable populations, which is expected to boost the mRNA vaccines industry growth. During the COVID-19 pandemic, governments worldwide, including those in North America, Europe, and many other regions, were actively engaged in procuring vaccines for their citizens. This played a significant role in the initial phases of vaccination campaigns. Public health initiatives, often led by public health agencies, focus on large-scale vaccination campaigns to reach a significant portion of the population efficiently. Mass vaccination centers, mobile vaccination units, and targeted outreach efforts are common components of public health initiatives. Such initiatives are particularly relevant during pandemics or when the goal is to vaccinate a substantial portion of the population quickly and most mRNA vaccines manufacturers are benefited by this initiative.mRNA vaccines Market Competitive Landscape
Several mRNA vaccines companies and organizations are working on developing mRNA vaccines for various diseases beyond COVID-19. The success of mRNA technology in combating the pandemic likely attracted new players and increased investment in this field. The success of mRNA vaccines for COVID-19 highlighted the potential of this technology for other infectious diseases and even non-infectious diseases such as cancer. This success could lead to the emergence of new companies and collaborations focusing on mRNA vaccine development. Pfizer, a well-established pharmaceutical company, partnered with BioNTech, a German biotechnology firm known for its mRNA expertise. Pfizer's extensive distribution network and global presence allowed for widespread delivery of the vaccine. Pfizer-BioNTech vaccine was one of the first mRNA vaccines authorized for emergency use and gained a significant market share early on. Moderna, an American biotechnology company, developed an mRNA-based COVID-19 vaccine that showed high efficacy rates in clinical trials. Moderna's vaccine received authorization around the same time as Pfizer-BioNTech's, providing an alternative option for vaccination efforts. The company's technology platform and ongoing research indicated a commitment to mRNA-based therapeutics.mRNA vaccines Market Scope: Inquire before buying
mRNA vaccines Market Report Coverage Details Base Year: 2022 Forecast Period: 2023-2029 Historical Data: 2018 to 2022 Market Size in 2022: US $ 11.4 Bn. Forecast Period 2023 to 2029 CAGR: 9.7% Market Size in 2029: US $ 21.79 Bn. Segments Covered: by Type Self-Amplifying mRNA based vaccines Conventional Non- Amplifying mRNA based vaccines by Application Cancer Immunotherapy Allergy Tolerization Therapeutic Vaccines Protein Replacement Others by Distribution Channels Government Procurement Private Healthcare Providers Public Health Initiatives by End-User Hospitals & Clinics Vaccination centers Research institutes & Biotechnology companies mRNA Vaccines Market by Region
North America (United States, Canada and Mexico) Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe) Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan and Rest of APAC) Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A) South America (Brazil, Argentina Rest of South America)mRNA Vaccines Key players
1. Pfizer-BioNTech 2. Moderna 3. CureVac 4. Arcturus Therapeutics 5. eTheRNA Immunotherapies 6. BioNTech SE 7. Acuitas Therapeutics 8. VaxartFrequently Asked Questions:
1] What is the growth rate of the Global mRNA Vaccines Market? Ans. The Global mRNA Vaccines Market is growing at a significant rate of 9.7% over the forecast period. 2] Which region is expected to dominate the Global mRNA Vaccines Market? Ans. North America region is expected to dominate the mRNA Vaccines Market over the forecast period. 3] What is the expected Global mRNA Vaccines Market size by 2029? Ans. The market size of the mRNA Vaccines Market is expected to reach USD 21.29 Bn by 2029. 4] Who are the top players in the Global mRNA Vaccines Industry? Ans. The major key players in the Global mRNA Vaccines Market are Pfizer-BioNTech, Moderna, CureVac, and Therapeutics. 5] Which factors are expected to drive the Global mRNA Vaccines Market growth by 2029? Ans. Accelerated Development in vaccines is expected to drive the mRNA Vaccines Market growth over the forecast period (2023-2029).
1. MRNA Vaccines Market Introduction 1.1. Study Assumption and Market Definition 1.2. Scope of the Study 1.3. Executive Summary 2. MRNA Vaccines Market: Dynamics 2.1. MRNA Vaccines Market Trends by Region 2.1.1. Global MRNA Vaccines Market Trends 2.1.2. North America MRNA Vaccines Market Trends 2.1.3. Europe MRNA Vaccines Market Trends 2.1.4. Asia Pacific MRNA Vaccines Market Trends 2.1.5. Middle East and Africa MRNA Vaccines Market Trends 2.1.6. South America MRNA Vaccines Market Trends 2.2. MRNA Vaccines Market Dynamics by Region 2.2.1. North America 2.2.1.1. North America MRNA Vaccines Market Drivers 2.2.1.2. North America MRNA Vaccines Market Restraints 2.2.1.3. North America MRNA Vaccines Market Opportunities 2.2.1.4. North America MRNA Vaccines Market Challenges 2.2.2. Europe 2.2.2.1. Europe MRNA Vaccines Market Drivers 2.2.2.2. Europe MRNA Vaccines Market Restraints 2.2.2.3. Europe MRNA Vaccines Market Opportunities 2.2.2.4. Europe MRNA Vaccines Market Challenges 2.2.3. Asia Pacific 2.2.3.1. Asia Pacific MRNA Vaccines Market Drivers 2.2.3.2. Asia Pacific MRNA Vaccines Market Restraints 2.2.3.3. Asia Pacific MRNA Vaccines Market Opportunities 2.2.3.4. Asia Pacific MRNA Vaccines Market Challenges 2.2.4. Middle East and Africa 2.2.4.1. Middle East and Africa MRNA Vaccines Market Drivers 2.2.4.2. Middle East and Africa MRNA Vaccines Market Restraints 2.2.4.3. Middle East and Africa MRNA Vaccines Market Opportunities 2.2.4.4. Middle East and Africa MRNA Vaccines Market Challenges 2.2.5. South America 2.2.5.1. South America MRNA Vaccines Market Drivers 2.2.5.2. South America MRNA Vaccines Market Restraints 2.2.5.3. South America MRNA Vaccines Market Opportunities 2.2.5.4. South America MRNA Vaccines Market Challenges 2.3. PORTER’s Five Forces Analysis 2.4. PESTLE Analysis 2.5. Value Chain Analysis 2.6. Regulatory Landscape by Region 2.6.1. Global 2.6.2. North America 2.6.3. Europe 2.6.4. Asia Pacific 2.6.5. Middle East and Africa 2.6.6. South America 2.7. MRNA Vaccines Clinical Trial Analysis for MRNA Vaccines 2.8. Key Opinion Leader Analysis For MRNA Vaccines Industry 2.9. Analysis of Government Schemes and Initiatives For MRNA Vaccines Industry 2.10. The Global Pandemic Impact on MRNA Vaccines Market 2.11. MRNA Vaccines Price Trend Analysis (2021-22) 3. MRNA Vaccines Market: Global Market Size and Forecast by Segmentation for Demand and Supply Side (by Value in USD Million) (2022-2029) 3.1. MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 3.1.1. Self-Amplifying mRNA based vaccines 3.1.2. Conventional Non- Amplifying mRNA based vaccines 3.2. MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 3.2.1. Cancer Immunotherapy 3.2.2. Allergy Tolerization 3.2.3. Therapeutic Vaccines 3.2.4. Protein Replacement 3.2.5. Others 3.3. MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 3.3.1. Government Procurement 3.3.2. Private Healthcare Providers 3.3.3. Public Health Initiatives 3.4. MRNA Vaccines Market Size and Forecast, by End User (2022-2029) 3.4.1. Hospitals & Clinics 3.4.2. Vaccination centers 3.4.3. Research institutes & Biotechnology companies 3.5. MRNA Vaccines Market Size and Forecast, by Region (2022-2029) 3.5.1. North America 3.5.2. Europe 3.5.3. Asia Pacific 3.5.4. Middle East and Africa 3.5.5. South America 4. North America MRNA Vaccines Market Size and Forecast by Segmentation for Demand and Supply Side (by Value in USD Million) (2022-2029) 4.1. North America MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 4.1.1. Self-Amplifying mRNA based vaccines 4.1.2. Conventional Non- Amplifying mRNA based vaccines 4.2. North America MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 4.2.1. Cancer Immunotherapy 4.2.2. Allergy Tolerization 4.2.3. Therapeutic Vaccines 4.2.4. Protein Replacement 4.2.5. Others 4.3. North America MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 4.3.1. Government Procurement 4.3.2. Private Healthcare Providers 4.3.3. Public Health Initiatives 4.4. North America MRNA Vaccines Market Size and Forecast, by End User (2022-2029) 4.4.1. Hospitals & Clinics 4.4.2. Vaccination centers 4.4.3. Research institutes & Biotechnology companies 4.5. North America MRNA Vaccines Market Size and Forecast, by Country (2022-2029) 4.5.1. United States 4.5.1.1. United States MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 4.5.1.1.1. Self-Amplifying mRNA based vaccines 4.5.1.1.2. Conventional Non- Amplifying mRNA based vaccines 4.5.1.2. United States MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 4.5.1.2.1. Cancer Immunotherapy 4.5.1.2.2. Allergy Tolerization 4.5.1.2.3. Therapeutic Vaccines 4.5.1.2.4. Protein Replacement 4.5.1.2.5. Others 4.5.1.3. United States MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 4.5.1.3.1. Government Procurement 4.5.1.3.2. Private Healthcare Providers 4.5.1.3.3. Public Health Initiatives 4.5.1.4. United States MRNA Vaccines Market Size and Forecast, by End User (2022-2029) 4.5.1.4.1. Hospitals & Clinics 4.5.1.4.2. Vaccination centers 4.5.1.4.3. Research institutes & Biotechnology companies 4.5.2. Canada 4.5.2.1. Canada MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 4.5.2.1.1. Self-Amplifying mRNA based vaccines 4.5.2.1.2. Conventional Non- Amplifying mRNA based vaccines 4.5.2.2. Canada MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 4.5.2.2.1. Cancer Immunotherapy 4.5.2.2.2. Allergy Tolerization 4.5.2.2.3. Therapeutic Vaccines 4.5.2.2.4. Protein Replacement 4.5.2.2.5. Others 4.5.2.3. Canada MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 4.5.2.3.1. Government Procurement 4.5.2.3.2. Private Healthcare Providers 4.5.2.3.3. Public Health Initiatives 4.5.2.4. Canada MRNA Vaccines Market Size and Forecast, by End User (2022-2029) 4.5.2.4.1. Hospitals & Clinics 4.5.2.4.2. Vaccination centers 4.5.2.4.3. Research institutes & Biotechnology companies 4.5.3. Mexico 4.5.3.1. Mexico MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 4.5.3.1.1. Self-Amplifying mRNA based vaccines 4.5.3.1.2. Conventional Non- Amplifying mRNA based vaccines 4.5.3.2. Mexico MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 4.5.3.2.1. Cancer Immunotherapy 4.5.3.2.2. Allergy Tolerization 4.5.3.2.3. Therapeutic Vaccines 4.5.3.2.4. Protein Replacement 4.5.3.2.5. Others 4.5.3.3. Mexico MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 4.5.3.3.1. Government Procurement 4.5.3.3.2. Private Healthcare Providers 4.5.3.3.3. Public Health Initiatives 4.5.3.4. Mexico MRNA Vaccines Market Size and Forecast, by End User (2022-2029) 4.5.3.4.1. Hospitals & Clinics 4.5.3.4.2. Vaccination centers 4.5.3.4.3. Research institutes & Biotechnology companies 5. Europe MRNA Vaccines Market Size and Forecast by Segmentation for Demand and Supply Side (by Value in USD Million) (2022-2029) 5.1. Europe MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 5.2. Europe MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 5.3. Europe MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 5.4. Europe MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 5.5. Europe MRNA Vaccines Market Size and Forecast, by Country (2022-2029) 5.5.1. United Kingdom 5.5.1.1. United Kingdom MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 5.5.1.2. United Kingdom MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 5.5.1.3. United Kingdom MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 5.5.1.4. United Kingdom MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 5.5.2. France 5.5.2.1. France MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 5.5.2.2. France MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 5.5.2.3. France MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 5.5.2.4. France MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 5.5.3. Germany 5.5.3.1. Germany MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 5.5.3.2. Germany MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 5.5.3.3. Germany MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 5.5.3.4. Germany MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 5.5.4. Italy 5.5.4.1. Italy MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 5.5.4.2. Italy MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 5.5.4.3. Italy MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 5.5.4.4. Italy MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 5.5.5. Spain 5.5.5.1. Spain MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 5.5.5.2. Spain MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 5.5.5.3. Spain MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 5.5.5.4. Spain MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 5.5.6. Sweden 5.5.6.1. Sweden MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 5.5.6.2. Sweden MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 5.5.6.3. Sweden MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 5.5.6.4. Sweden MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 5.5.7. Austria 5.5.7.1. Austria MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 5.5.7.2. Austria MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 5.5.7.3. Austria MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 5.5.7.4. Austria MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 5.5.8. Rest of Europe 5.5.8.1. Rest of Europe MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 5.5.8.2. Rest of Europe MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 5.5.8.3. Rest of Europe MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 5.5.8.4. Rest of Europe MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 6. Asia Pacific MRNA Vaccines Market Size and Forecast by Segmentation for Demand and Supply Side (by Value in USD Million) (2022-2029) 6.1. Asia Pacific MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 6.2. Asia Pacific MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 6.3. Asia Pacific MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 6.4. Asia Pacific MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 6.5. Asia Pacific MRNA Vaccines Market Size and Forecast, by Country (2022-2029) 6.5.1. China 6.5.1.1. China MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 6.5.1.2. China MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 6.5.1.3. China MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 6.5.1.4. China MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 6.5.2. S Korea 6.5.2.1. S Korea MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 6.5.2.2. S Korea MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 6.5.2.3. S Korea MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 6.5.2.4. S Korea MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 6.5.3. Japan 6.5.3.1. Japan MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 6.5.3.2. Japan MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 6.5.3.3. Japan MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 6.5.3.4. Japan MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 6.5.4. India 6.5.4.1. India MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 6.5.4.2. India MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 6.5.4.3. India MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 6.5.4.4. India MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 6.5.5. Australia 6.5.5.1. Australia MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 6.5.5.2. Australia MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 6.5.5.3. Australia MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 6.5.5.4. Australia MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 6.5.6. Indonesia 6.5.6.1. Indonesia MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 6.5.6.2. Indonesia MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 6.5.6.3. Indonesia MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 6.5.6.4. Indonesia MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 6.5.7. Malaysia 6.5.7.1. Malaysia MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 6.5.7.2. Malaysia MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 6.5.7.3. Malaysia MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 6.5.7.4. Malaysia MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 6.5.8. Vietnam 6.5.8.1. Vietnam MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 6.5.8.2. Vietnam MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 6.5.8.3. Vietnam MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 6.5.8.4. Vietnam MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 6.5.9. Taiwan 6.5.9.1. Taiwan MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 6.5.9.2. Taiwan MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 6.5.9.3. Taiwan MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 6.5.9.4. Taiwan MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 6.5.10. Rest of Asia Pacific 6.5.10.1. Rest of Asia Pacific MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 6.5.10.2. Rest of Asia Pacific MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 6.5.10.3. Rest of Asia Pacific MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 6.5.10.4. Rest of Asia Pacific MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 7. Middle East and Africa MRNA Vaccines Market Size and Forecast by Segmentation for Demand and Supply Side (by Value in USD Million) (2022-2029 7.1. Middle East and Africa MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 7.2. Middle East and Africa MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 7.3. Middle East and Africa MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 7.4. Middle East and Africa MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 7.5. Middle East and Africa MRNA Vaccines Market Size and Forecast, by Country (2022-2029) 7.5.1. South Africa 7.5.1.1. South Africa MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 7.5.1.2. South Africa MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 7.5.1.3. South Africa MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 7.5.1.4. South Africa MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 7.5.2. GCC 7.5.2.1. GCC MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 7.5.2.2. GCC MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 7.5.2.3. GCC MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 7.5.2.4. GCC MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 7.5.3. Nigeria 7.5.3.1. Nigeria MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 7.5.3.2. Nigeria MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 7.5.3.3. Nigeria MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 7.5.3.4. Nigeria MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 7.5.4. Rest of ME&A 7.5.4.1. Rest of ME&A MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 7.5.4.2. Rest of ME&A MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 7.5.4.3. Rest of ME&A MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 7.5.4.4. Rest of ME&A MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 8. South America MRNA Vaccines Market Size and Forecast by Segmentation for Demand and Supply Side (by Value in USD Million) (2022-2029 8.1. South America MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 8.2. South America MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 8.3. South America MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 8.4. South America MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 8.5. South America MRNA Vaccines Market Size and Forecast, by Country (2022-2029) 8.5.1. Brazil 8.5.1.1. Brazil MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 8.5.1.2. Brazil MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 8.5.1.3. Brazil MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 8.5.1.4. Brazil MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 8.5.2. Argentina 8.5.2.1. Argentina MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 8.5.2.2. Argentina MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 8.5.2.3. Argentina MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 8.5.2.4. Argentina MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 8.5.3. Rest Of South America 8.5.3.1. Rest Of South America MRNA Vaccines Market Size and Forecast, by Type (2022-2029) 8.5.3.2. Rest Of South America MRNA Vaccines Market Size and Forecast, by Application (2022-2029) 8.5.3.3. Rest Of South America MRNA Vaccines Market Size and Forecast, by Distribution Channels (2022-2029) 8.5.3.4. Rest Of South America MRNA Vaccines Market Size and Forecast, by End User(2022-2029) 9. Global MRNA Vaccines Market: Competitive Landscape 9.1. MMR Competition Matrix 9.2. Competitive Landscape 9.3. Key Players Benchmarking 9.3.1. Company Name 9.3.2. Product Segment 9.3.3. End-user Segment 9.3.4. Revenue (2022) 9.3.5. Company Locations 9.4. Market Analysis by Organized Players vs. Unorganized Players 9.4.1. Organized Players 9.4.2. Unorganized Players 9.5. Leading MRNA Vaccines Market Companies, by market capitalization 9.6. Market Structure 9.6.1. Market Leaders 9.6.2. Market Followers 9.6.3. Emerging Players 9.7. Mergers and Acquisitions Details 10. Company Profile: Key Players 10.1. Pfizer-BioNTech 10.1.1. Company Overview 10.1.2. Business Portfolio 10.1.3. Financial Overview 10.1.4. SWOT Analysis 10.1.5. Strategic Analysis 10.1.6. Scale of Operation (small, medium, and large) 10.1.7. Details on Partnership 10.1.8. Regulatory Accreditations and Certifications Received by Them 10.1.9. Awards Received by the Firm 10.1.10. Recent Developments 10.2. Moderna 10.3. CureVac 10.4. Arcturus Therapeutics 10.5. eTheRNA Immunotherapies 10.6. BioNTech SE 10.7. Acuitas Therapeutics 10.8. Vaxart 11. Key Findings 12. Industry Recommendations 13. MRNA Vaccines Market: Research Methodology 14. Terms and Glossary
