Hyperuricemia Drugs Market Size – Industry Structure Evaluation, Demand Drivers Analysis, Regional Growth Analysis and Identification, Competitive Positioning Review & Global Market Size Forecast to 2032
Overview
The Hyperuricemia Drugs Market size was valued at USD 5.98 Billion in 2025 and the total Hyperuricemia Drugs Market revenue is expected to grow at a CAGR of 8.52% from 2026 to 2032, reaching nearly USD 10.6 Billion by 2032.
Hyperuricemia Drugs Market Overview
“Hyperuricemia” is the medical term for having high uric acid levels in your body. Uric acid is a waste product that creates the body breaks down chemicals called purines in food and drinks. Most uric acid dissolves in your blood, passes through your kidneys, and leaves your body in your pee (urine). Hyperuricemia happens if too much uric acid stays in your body.
Hyperuricemia affects about 38 million Americans, over 11% of the population, and the incidence is increasing worldwide. Up to 21% of the general population and 25% of hospitalized patients are estimated to be asymptomatic, and uric acid levels be elevated 10 to 15 years before clinical manifestations of gout. Hyperuricemia does not indicate a pathological state because it is extremely prevalent in the general population and is asymptomatic in 90% to 95% of people.
The significant advancements in hyperuricemia drugs market strategies for managing gout, a condition characterized by elevated levels of uric acid in the blood. Treatment approaches lifestyle modifications, dietary changes, and increased physical activity, alongside pharmacological interventions aimed at reducing uric acid levels. The introduction of innovative therapies represents a significant step forward in the management of hyperuricemia in gout, offering hope for improved outcomes and quality of life for patients. As these agents progress through clinical trials and regulatory approval processes, they provide more effective and personalized treatment options for individuals living with gout.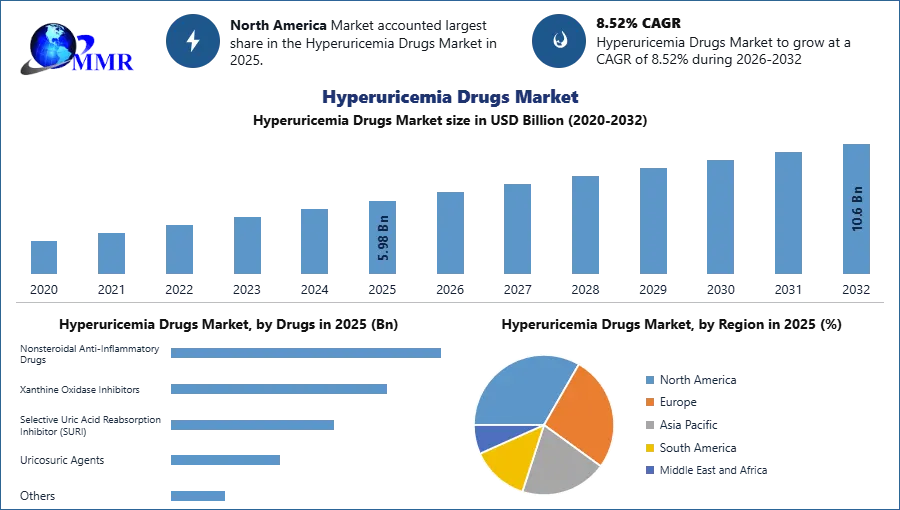 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
Hyperuricemia Drugs Market Trends:
Advancements in Technologies in Testing Influencing the Hyperuricemia Drugs Market
Since the mid-20th century, the gout therapeutics field came to an effective cessation during which no novel drugs were approved for therapeutic purposes. On the other hand, the early 21st century has seen a revitalization of gout treatment, commencement with febuxostat as xanthine oxidase inhibitor (XOI) in the hyperuricemia drugs market. Abbreviated New Drug Application (ANDA) with the U.S. Food and Drug Administration (FDA) for Febuxostat Tablets, 40 mg and 80 mg. This product is the generic version of Uloric®, which is indicated for the chronic management of hyperuricemia in patients with gout. It is not recommended for the treatment of asymptomatic hyperuricemia.
An emerging trend in the hyperuricemia drugs market involves strategic partnerships aimed at advancing novel therapies for the treatment of conditions such as gout and hyperuricemia. A recent example of this trend is the agreement between Kissei Pharmaceutical Co., Ltd. and Pfizer Inc. In this partnership, Kissei grants Pfizer exclusive worldwide rights (excluding Japan) to develop and commercialize the investigational therapy KUX-1151 for gout and hyperuricemia. KUX-1151, discovered by Kissei, represents a promising new approach to the treatment of these conditions. It works by inhibiting both xanthine oxidase, which regulates uric acid production, and uric acid transporter (URAT1), responsible for uric acid reabsorption.
As part of the agreement, Kissei receives an upfront payment and eligible for milestone payments and royalties based on product sales. Pfizer's Neusentis Research Unit spearheads the advancement of the KUX-1151 program, leveraging its expertise in pain and associated conditions.
Hyperuricemia Drugs Market Dynamics:
The Rising Incidence of chronic renal diseases
The hyperuricemia drugs market significantly grows by various factors. The increasing incidence of chronic renal diseases impairs the kidneys' ability to eliminate uric acid efficiently. Additionally, lifestyle factors like consuming purine-rich diets, excessive alcohol intake, obesity, and hypertension contribute to rising hyperuricemia cases. The widespread adoption of uricosuric agents enhances uric acid excretion and alleviates symptoms. Joint replacement therapy is fueling hyperuricemia drugs market growth by addressing pain and improving mobility in affected individuals. Investments in research and development by key industries like Pfizer Inc., Mylan N.V., and Bristol Myers Squibb Company are leading to the development of novel recombinant uricase preparations, offering effective control over elevated uric acid levels in critical conditions. The innovative anti-hyperuricemia therapies, alongside dietary modifications like reducing purine intake and increasing hydration, are expected to drive the hyperuricemia drugs market.
Growing Geriatric Population Fuels Hyperuricemia Drugs Market
The aging population is a key driver behind the rapid growth of the hyperuricemia drugs market. As individuals age, their kidney function naturally declines, impacting the body's ability to effectively filter and eliminate uric acid. This diminished function leads to elevated uric acid levels in the bloodstream, indicating hyperuricemia. Changes in dietary habits and physical activity levels. Unhealthy practices such as excessive alcohol consumption and consumption of purine-rich foods become more prevalent with age, further exacerbating uric acid levels. With the demographic trend toward an aging population, the prevalence of hyperuricemia is expected to rise significantly.
For example, in January 2022, the European Union had an estimated population of 446.7 million, with individuals aged 65 or older comprising 21.1% of the total population. This demographic shift underscores the need for effective management strategies for hyperuricemia among the elderly and drives the ongoing development of the hyperuricemia drugs market.
Hyperuricemia Drugs Market Regional Insights:
North America dominates the hyperuricemia drugs market due to the advancement of technology for kidney diseases and increasing initiatives taken by the pharmaceutical organizations to generate novel formulation. Gout is the most common rheumatic disease of adulthood, with a self-reported prevalence of more than 8 million cases in the United States, affecting 3.9% of adults, with a male-to-female ratio of 3:1.5 However, at an older age the incidence of gout in women approaches the incidence in men. Followed by Europe and Asia pacific are growing the hyperuricemia drugs market with the higher awareness of hyperuricemia and advanced health care system.
Hyperuricemia Drugs Market Competitive Landscape:
14 Feb 2024- The U.S. Food and Drug Administration (FDA) has granted Priority Review to hyperuricemia drugs market key player Bristol Myers Squibb’s application for Augtyro™, a groundbreaking treatment targeting ROS1-positive or NTRK-positive locally advanced or metastatic solid tumors, including non-small cell lung cancer (NSCLC). Augtyro (TPX-0005, BMS-986472) is a next-generation tyrosine kinase inhibitor (TKI), that addresses significant medical needs for patients in this category.
Among the 351 patients who received Augtyro, 35% experienced increased alanine transaminase (ALT) and 40% experienced increased aspartate aminotransferase (AST). Grade 3 or 4 increased ALT occurred in 2%, while increased AST occurred in 2.6%. The median time to onset of increased ALT or AST was 15 days, with some cases appearing within a day and others up to almost two years. Instances of increased ALT or AST necessitating dose interruptions or reductions were observed in 2.8% and 1.4% of patients, respectively, while hyperbilirubinemia leading to dose interruptions occurred in 0.6%.
Augtyro's efficacy and safety profile presents a significant advancement in the treatment landscape for ROS1-positive or NTRK-positive locally advanced or metastatic solid tumors. Despite potential adverse events, the overall benefit-risk profile positions Augtyro as a promising option for patients facing these challenging conditions. Its acceptance for Priority Review by the FDA underscores the urgent need for such therapeutic innovations in addressing medical needs within the hyperuricemia drugs market.
Hyperuricemia Drugs Market Recent Industry Developments (2025–2026)
| Date | Company | Development | Impact |
|---|---|---|---|
| 28 March 2025 | Shanton Pharma | The company announced positive topline data from its Phase 2b study of SAP-001, a first-in-class kidney transporter inhibitor for refractory gout. | Nearly 100% of patients reached the therapeutic target of serum uric acid (sUA) levels below 6mg/dL by the third month of treatment. |
| 15 May 2025 | LG Chem | The company successfully completed its EURELIA2 Phase 3 study evaluating Tigulixostat, a novel xanthine oxidase inhibitor, against allopurinol. | Completion of this large-scale trial involving 2,202 patients accelerates the timeline for a New Drug Application (NDA) submission. |
| 16 July 2025 | China Medical System (CMS) | CMS secured exclusive commercialization rights for ABP-671, a potent URAT1 inhibitor for hyperuricemia, as it dual-listed on the Singapore Exchange. | The deal strengthens CMS’s position in the chronic disease market and facilitates the transition of ABP-671 into Phase 3 clinical trials. |
| 25 July 2025 | Shanton Pharma | The U.S. FDA granted Fast Track Designation to SAP-001 for the treatment of hyperuricemia in adult patients with refractory gout. | This designation allows for expedited regulatory reviews and rolling submission of the marketing application to address unmet medical needs. |
| 21 August 2025 | Arthrosi Therapeutics | The company achieved full enrollment for its REDUCE 1 Phase 3 trial of Pozdeutinurad (AR882), a next-generation URAT1 inhibitor. | With 750 patients enrolled, the company remains on track to deliver pivotal data in Q2 2026 to support its planned NDA submission. |
| 28 October 2025 | Atom Bioscience | Data presented at ACR Convergence 2025 demonstrated that ABP-671 achieved an 82.1% maximum mean sUA decrease in hyperuricemia subjects. | The high efficacy and dose-dependent safety profile position the drug as a potentially best-in-class urate-lowering therapy. |
Hyperuricemia Drugs Market Scope: Inquiry Before Buying
| Hyperuricemia Drugs Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 5.98 USD Billion |
| Forecast Period 2026-2032 CAGR: | 8.52% | Market Size in 2032: | 10.6 USD Billion |
| Segments Covered: | by Type | Asymptomatic Hyperuricemia Symptomatic Hyperuricemia Others |
|
| by Diseases | Gout Kidney Stone Others |
||
| by Drugs | Nonsteroidal Anti-Inflammatory Drugs Xanthine Oxidase Inhibitors Selective Uric Acid Reabsorption Inhibitor (SURI) Uricosuric Agents Others |
||
| by End-User | Hospitals Homecare Specialty Clinics Others |
||
Hyperuricemia Drugs Market by Region:
1. North America (United States, Canada, and Mexico)
2. Europe (UK, France, Germany, Italy, Spain, Sweden, Russia, and the Rest of Europe)
3. Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan, and the Rest of APAC)
4. Middle East and Africa (South Africa, GCC, and the Rest of MEA)
5. South America (Brazil, Argentina, Rest of South America)
Hyperuricemia Drugs Market key players
- Pfizer Inc (United States)
- Mylan N.V. (Viatris Inc.)
- Bristol Myers Squibb Company
- Fresenius Kabi AG
- Hikma Pharmaceuticals PLC
- F. Hoffmann-La Roche Ltd (Switzerland)
- Novartis AG
- Teva Pharmaceutical Industries Ltd.
- GSK plc
- Bayer AG
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Endo International plc
- Takeda Pharmaceutical Company Limited
- AstraZeneca plc
- Horizon Therapeutics plc (Amgen Inc.)
- Boehringer Ingelheim International GmbH
- Sanofi S.A.
- Merck & Co. Inc.
- Teijin Pharma Limited
- Arthrosi Therapeutics Inc.
- Jiangsu Atom Bioscience and Pharmaceutical Co. Ltd.
- Kissei Pharmaceutical Co.Ltd.
- LG Life Sciences
- Selecta Biosciences Inc.
