Global Small Interfering RNA (siRNA) Therapeutics Market: Growth, Trends, and Forecast (2026-2032) with insights into market dynamics and key players
Overview
The Small Interfering RNA (siRNA) Therapeutics Market Size was valued at USD 17.55 Billion in 2025, and the total revenue is expected to grow at a CAGR of 17.6% from 2026 to 2032, reaching USD 54.59 Billion by 2032. The Small Interfering RNA (siRNA) Therapeutics market is driven by increasing demand for precision medicine, FDA drug approvals, advancements in delivery systems, increased R&D investments, and expansion into new therapeutic areas.
Small Interfering RNA (siRNA) Therapeutics Market Overview
The Small Interfering RNA (siRNA) therapeutics are showing promise in treating cancer, ocular diseases, and metabolic disorders, with examples like QPI-1002 and TKM-PLK. Key challenges include efficient delivery and stability, but innovations in nanoparticle and liposome-based systems are addressing these issues. The market is driven by precision medicine demand, with companies such as Alnylam Pharmaceuticals and Arrowhead Pharmaceuticals leading the way.
Small Interfering RNA (siRNA) Therapeutics Market Key Highlights
• Olpasiran Clinical Trial: The trial demonstrated a 95% reduction in lipoprotein(a) levels in patients with cardiovascular disease, showcasing the potential of siRNA in treating cardiovascular conditions.
• Hereditary Transthyretin-mediated Amyloidosis: The approval of patisiran (ONPATTRO), the first siRNA-based drug, has paved the way for treatments in genetic diseases, contributing to the growth of the siRNA therapeutics market.
• Nanoparticle-Based Delivery Systems: Nanoparticle-based systems, such as lipid nanoparticles, are gaining preference for siRNA delivery, helping target tissues beyond the liver, including tumors and the brain, enhancing therapeutic efficacy.
• Alnylam Pharmaceuticals Expansion: Alnylam announced a $250 million investment to expand its U.S. manufacturing facility, supporting its growing siRNA pipeline for diseases like hypertension and obesity.
• Emerging Biotech Contributions: Companies like Silence Therapeutics and Sirnaomics are making significant strides in siRNA applications, with Silence Therapeutics focusing on mRNAi GOLD technology for precision delivery and Sirnaomics advancing treatments in oncology and fibrosis.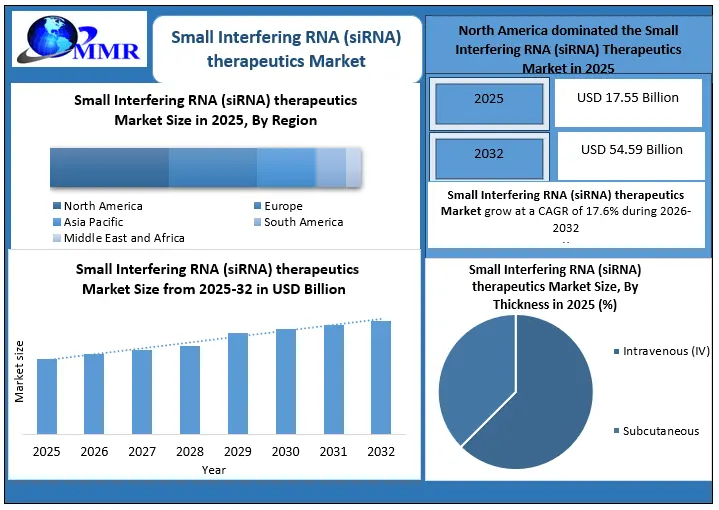 To Know About The Research Methodology :- Request Free Sample Report
To Know About The Research Methodology :- Request Free Sample Report
Small Interfering RNA (siRNA) Therapeutics Market Trend
Expansion of Sirna Applications Beyond the Liver
Therapeutic Applications of siRNA
• Cancer Therapy: siRNA targets oncogenes and biomarkers, like KRAS mutations in pancreatic cancer (siG12D-LODER) and polo-like kinase 1 (PLK1) in solid tumors (TKM-PLK). Preclinical and clinical trials show promising results in combination with chemotherapy and immunotherapy. For example, STAT3 siRNA improves efficacy when combined with immune checkpoint inhibitors.
• Naked siRNA: Directly delivered siRNA without carriers, like QPI-1002 (acute kidney injury) and SYL040012 (ocular diseases like AMD), offers a more cost-effective alternative but faces challenges in stability and delivery efficiency.
Distribution of Small Interfering RNA (siRNA) Therapeutic Development Across Phases by Therapeutic Area
| Therapeutic Area | Preclinical | Phase I | Phase II | Phase III |
| Cardiovascular disorders | 34 | 11 | 8 | 1 |
| Neurological disorders | 31 | 4 | 3 | 1 |
| Cancer | 28 | 4 | 2 | 0 |
| Metabolic disorders | 26 | 7 | 2 | 0 |
| Immunological disorders | 14 | 1 | 2 | 0 |
| Musculoskeletal disorders | 10 | 0 | 0 | 0 |
| Sensory disorders | 9 | 2 | 0 | 0 |
| Infectious diseases | 7 | 2 | 6 | 1 |
Key Advancements in siRNA Delivery Beyond the Liver
• Targeting New Tissues: Progress in delivering siRNA to tissues like the brain, lungs, and adipose tissue promises expanded therapeutic uses in neurological diseases and metabolic disorders. For example, QPI-1007 targets caspase 2 for ocular diseases like POAG.
Overview of RNA-Based Therapeutic Approaches and Their Targets
| Category | Example | Disease/Target | Therapeutic Approach |
| Cancer Therapy | siG12D-LODER, TKM-PLK | KRAS, PLK1 | Targeting oncogenes |
| Naked siRNA | QPI-1002, SYL040012 | p53, VEGFR1 | Acute kidney injury, ocular diseases |
| Ocular Diseases | QPI-1007 | Caspase 2 | Non-arteritic ischemic optic neuropathy, POAG |
| Other Applications | QPI-1002, SYL040012 | Age-related macular degeneration | Inhibition of angiogenesis |
Small Interfering RNA (siRNA) Therapeutics Market Dynamics
Increasing Demand for Precision Medicine to boost Small Interfering RNA (siRNA) Therapeutics Market
The development in siRNA therapeutics is the Olpasiran clinical trial, where researchers at Brigham and Women's Hospital reported a 95% reduction in lipoprotein(a) levels in patients with cardiovascular disease. The study, which included about 300 patients, demonstrated the potential of siRNA in reducing a key cholesterol protein that contributes to atherosclerosis. This reduction was sustained over 36 weeks, emphasising the effectiveness of RNA interference (RNAi) technologies in cardiovascular treatments.
Several emerging biotech companies are making significant strides in siRNA therapeutics. Silence Therapeutics is focusing on precision siRNA delivery using its mRNAi GOLD platform, which offers promising new approaches to targeted therapy. Sirnaomics is advancing siRNA therapies in the areas of oncology and fibrosis, exploring both topical and injectable formulations. Dicerna Pharmaceuticals, part of Novo Nordisk, has developed GalXC technology, which allows for liver-targeted siRNA delivery, offering a foundation for novel treatments for liver diseases.
Challenge of Effective Drug Delivery
The delivery of siRNA efficiently to the target tissues and cells remains one of the most complex obstacles in its development. Despite the approval of siRNA-based therapies like ONPATTRO (patisiran) and GIVLAARI (givosiran) by Alnylam Pharmaceuticals, which demonstrated the potential of siRNA in treating genetic disorders, the path to these successes was not easy. These therapies rely on the GalNAc (N-acetylgalactosamine)-siRNA conjugate, a critical platform that allows siRNA to be effectively delivered to the liver. However, the complexity of ensuring the stability, activity, specificity, and minimising off-target effects of siRNA still presents substantial challenges. While these therapies are able to be administered less frequently—quarterly or even twice-yearly—which is an advantage over traditional small molecules and antibodies, the issue of delivering siRNA to the correct tissues in a manner that is both safe and efficient remains a major hurdle.
Small Interfering RNA (siRNA) Therapeutics Market Segment Analysis
By Type, the market is segmented into the Liposome Based Systemic Therapy, Nanoparticle Based Systemic Therapy and Others. Nanoparticle Based Systemic Therapy held the largest Small Interfering RNA (siRNA) Therapeutics Market share in 2025. It is increasingly preferred for siRNA delivery due to its ability to deliver small interfering RNA beyond the liver into complex tissues such as tumours and the brain, a key trend in North America and global RNAi research. Nanoparticle approaches (including lipid nanoparticles, polymer or inorganic particles) enhance stability, controlled release, and site-specific targeting of siRNA molecules, addressing core delivery challenges like degradation and cellular uptake that limit unmodified siRNAs.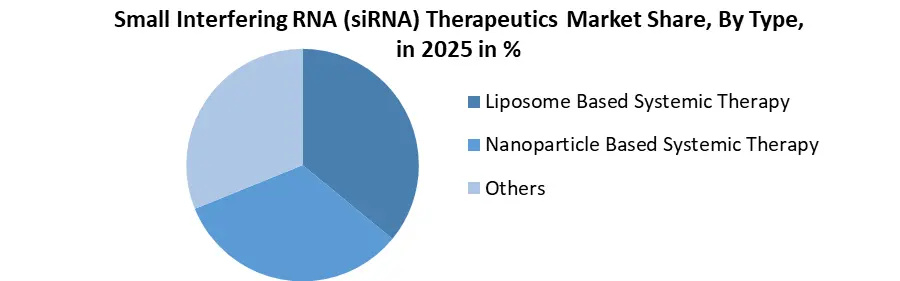
By Indication, the market is categorised into the Hereditary Transthyretin-mediated Amyloidosis, Acute Hepatic Porphyria, Primary Hyperoxaluria Type 1, Primary Hypercholesterolemia and Others. Hereditary Transthyretin-mediated Amyloidosis dominates Small Interfering RNA (siRNA) Therapeutics Market. It is a key indication in siRNA therapeutics, marked by the first FDA-approved siRNA drug, patisiran (Onpattro), targeting mutant TTR genes to treat amyloid protein buildup. Additional siRNA drugs, including givosiran, lumasiran, inclisiran, and vutrisiran, validate the clinical importance of this segment and expand siRNA use into rare genetic and metabolic disorders.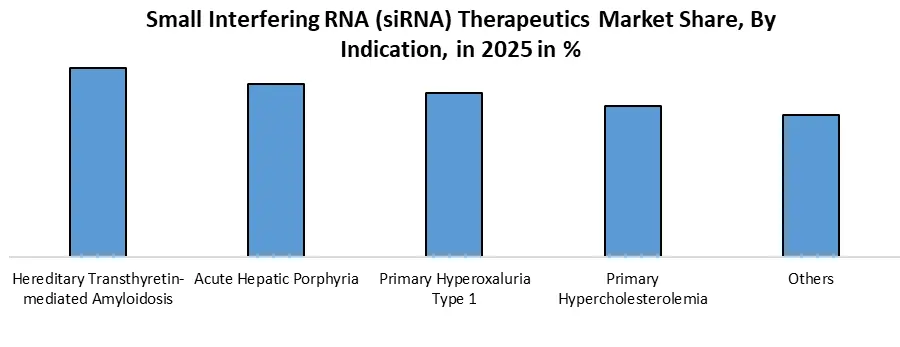
Small Interfering RNA (siRNA) Therapeutics Market Regional Insights
North America dominated the Small Interfering RNA (siRNA) Therapeutics Market in 2025 and is expected to continue its dominance over the forecast period.
• Growth in siRNA Therapeutics: Since the FDA approved patisiran (2018) for hereditary transthyretin-mediated amyloidosis, siRNA therapeutics have expanded to treat genetic diseases. Notable drugs include givosiran for acute hepatic porphyria and lumasiran for primary hyperoxaluria type 1.
• Investment in U.S. Manufacturing: Alnylam Pharmaceuticals is investing $250 million to expand its U.S. manufacturing facility to support the growing siRNA pipeline, particularly targeting diseases like hypertension, obesity, and other metabolic conditions.
• Challenges and Breakthroughs: Key challenges for siRNA include degradation, low cellular uptake, and immune responses. However, chemical modifications and advanced delivery systems like lipid nanoparticles (LNPs) and targeted conjugates are improving the stability and effectiveness of siRNA.
• Market Landscape: Over 260 siRNA drug candidates are in clinical development, with North America leading in RNAi innovations aimed at precision medicine.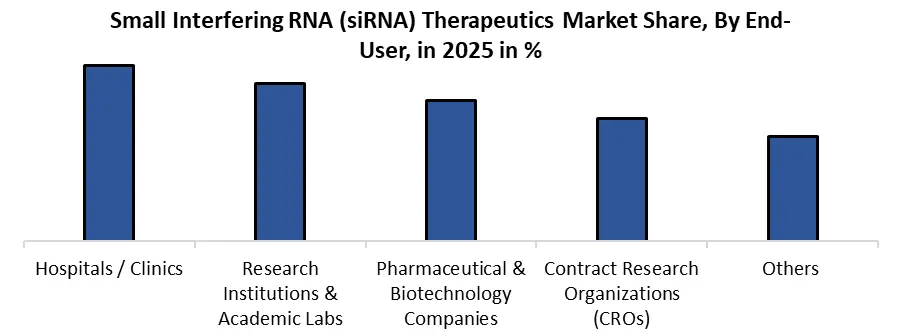
Small Interfering RNA (siRNA) Therapeutics Market Competitive Landscape
The Small Interfering RNA (siRNA) Therapeutics Market is highly competitive, driven by key players like Alnylam Pharmaceuticals, Novartis, and Arrowhead Pharmaceuticals, focusing on genetic disorders, oncology, and cardiovascular diseases. These companies are advancing RNA-based therapeutics, leveraging Lipid Nanoparticle (LNP) delivery systems and GalNAc conjugates to improve gene silencing effectiveness. Emerging players are exploring new siRNA delivery technologies, while large pharmaceutical companies are investing heavily in clinical trials and partnerships to gain market share.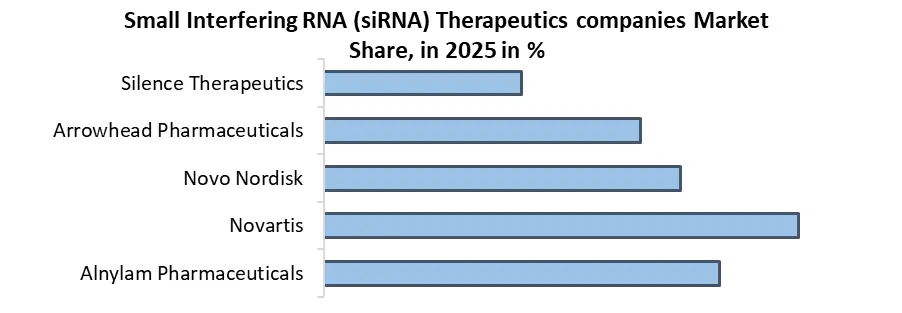
Recent Developments
• June 9, 2025 – Alnylam Pharmaceuticals continues to lead the siRNA space, expanding beyond liver-targeted therapies with FDA approvals for Amvuttra and Qfitlia. Boehringer Ingelheim signed a $2B agreement with Suzhou Ribo Life Science to develop siRNA for metabolic diseases. GSK invested $1.2B in Boston Pharmaceuticals to pair siRNA with FGF21 analog for non-alcoholic steatohepatitis. Arrowhead Pharmaceuticals reports promising Phase I/II results for siRNA targeting obesity and lung conditions.
• December 17, 2025 – Alnylam Pharmaceuticals announced a $250 million investment to expand its U.S. manufacturing facility in Norton, Massachusetts. The expansion will introduce the siRELIS enzymatic-ligation platform, enabling scalable, efficient siRNA production. This move aims to boost capacity, lower production costs, and support the growing RNAi therapeutics pipeline, including treatments for hypertension, type 2 diabetes, and obesity. The platform has been accepted into the FDA’s Emerging Technology Program, accelerating global regulatory engagement.
Small Interfering RNA (siRNA) Therapeutics Market Report: Inquire before buying
| Small Interfering RNA (siRNA) Therapeutics Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | USD 17.55 Bn. |
| Forecast Period 2026 to 2032 CAGR: | 17.6% | Market Size in 2032: | USD 54.59 Bn. |
| Segments Covered: | by Type | Liposome Based Systemic Therapy Nanoparticle Based Systemic Therapy Others |
|
| by Indication | Hereditary Transthyretin-mediated Amyloidosis Acute Hepatic Porphyria Primary Hyperoxaluria Type 1 Primary Hypercholesterolemia Others |
||
| by RoA | Intravenous (IV) Subcutaneous |
||
| by Thickness/ Molecule | Hereditary Transthyretin-mediated Amyloidosis Acute Hepatic Porphyria Primary Hyperoxaluria Type 1 Primary Hypercholesterolemia Others |
||
| by Application | Oncology Genetic Disorders Cardiovascular Diseases Infectious Diseases Metabolic Disorders Neurological Disorders Immunology Others |
||
| by End-Use | Hospitals / Clinics Research Institutions & Academic Labs Pharmaceutical & Biotechnology Companies Contract Research Organizations (CROs) Others |
||
Small Interfering RNA (siRNA) Therapeutics Market Key Players
1. Alnylam Pharmaceuticals
2. Novartis
3. Novo Nordisk
4. Arrowhead Pharmaceuticals
5. Silence Therapeutics
6. Sanofi Genzyme
7. Quark Pharmaceuticals
8. Dicerna Pharmaceuticals
9. Johnson & Johnson
10. GSK (GlaxoSmithKline)
11. Aro Biotherapeutics
12. GE Dharmacon
13. OPKO Health
14. Arbutus Biopharma
15. Genecon Biotechnologies
16. Sylentis S.A
17. OliX Pharmaceuticals, Inc.
18. Ionis Pharmaceuticals
19. Benitec Biopharma
20. Bio‑Path Holdings
21. Calando Pharmaceuticals
22. Avidity Biosciences
23. Code Biotherapeutics
24. Effigene Pharmaceuticals
25. Sirna Therapeutics
26. Patisiran (ONPATTRO)
27. Givosiran
28. Lumasiran
29. Inclisiran
30. Vutrisiran
Frequently Asked Questions
1. What are the growth drivers for the Small Interfering RNA (siRNA) Therapeutics Market?
Answer: The growth of the siRNA therapeutics market is driven by the increasing demand for precision medicine, advancements in RNA interference (RNAi) technologies, and the growing approval of siRNA-based therapies for rare genetic diseases, including hereditary transthyretin-mediated amyloidosis and acute hepatic porphyria.
2. What are the major restraints for the growth of the Small Interfering RNA (siRNA) Therapeutics Market?
Answer: Key restraints for the siRNA therapeutics market include challenges related to effective drug delivery, ensuring stability, minimizing off-target effects, and overcoming cellular uptake barriers for siRNA molecules.
3. Which region is expected to lead the global Small Interfering RNA (siRNA) Therapeutics Market during the forecast period?
Answer: North America is expected to dominate the global siRNA therapeutics market, driven by significant investments in siRNA research, advancements in RNAi technologies, and a high number of FDA-approved siRNA-based drugs in the region.
4. What was the market size in 2025 and growth rate of the Small Interfering RNA (siRNA) Therapeutics Market?
Answer: The Small Interfering RNA (siRNA) Therapeutics Market was valued at approximately USD 17.55 billion in 2025 and is expected to grow at a CAGR of 17.6 %, reaching an estimated USD 54.59 billion by 2032.
5. What segments are covered in the Small Interfering RNA (siRNA) Therapeutics Market report?
Answer: The siRNA therapeutics market report covers segments including Type (liposome-based, nanoparticle-based, and others), Indication (e.g., hereditary transthyretin-mediated amyloidosis, metabolic disorders), Route of Administration (IV and subcutaneous), Application (oncology, genetic disorders, etc.), and End-Use (hospitals, pharmaceutical companies, CROs).
