Gene Therapy Market– Industry Structure Evaluation, Demand Drivers Analysis, Regional Growth Analysis and Identification, Competitive Positioning Review & Global Market Size Forecast to 2032
Overview
The Gene Therapy Market size was valued at USD 10.03 Billion in 2024 and the total Gene Therapy revenue is expected to grow at a CAGR of 16% from 2025 to 2032, reaching nearly USD 32.89 Billion.
Gene Therapy Market Overview
Gene therapy is a medical technique that aims to cure or stop disease by fixing the genetic makeup and genetic issue beneath the surface. Gene therapy methods enable physicians to address a medical condition by changing an individual's genetic composition rather than resorting to medications or operations.
The First method of gene therapy, often called gene transfer or gene addition, was
developed to:
• Introduce a new gene into cells to help fight a disease.
• Introduce a non-faulty copy of a gene to stand in for the altered copy causing Disease.
The worldwide gene therapy market is quickly enlarging and is expected to experience substantial growth in the future. The gene therapy market aims to offer treatment for genetic disorders which are currently incurable. For ex: (Huntington's disease, Sickle Cell Disease) The gene therapy industry is dedicated to creating novel treatments that target the underlying cause of these illnesses.
The rising incidence of genetic disorders is the main driving force behind the growth of the gene therapy market. The rising occurrence of genetic disorders worldwide is causing a higher need for gene therapy. Developing gene therapies is a costly and intricate process, leading to high expenses for patients. For ex: (Voretigene neparvovec-rzyl injection is indicated for the treatment of patients with confirmed biallelic RPE65 mutation-associated retinal dystrophy with viable retinal cells. Costs $850 000 for one time treatment). The success of gene therapies relies heavily on the delivery mechanism, must translocate the healthy gene to specific cells within the body. Moreover, the immune system identify the gene therapy as an external threat, triggering an immune reaction that diminishes the therapy's efficacy. The pharmaceutical companies have a great opportunity to create groundbreaking therapies in the gene therapy market.
• As per the report published by Alliance for Regenerative Medicine, there are more than 1400 companies globally focusing on CGT (Cell Gene Therapy) and over 3500 therapies in preclinical and clinical development.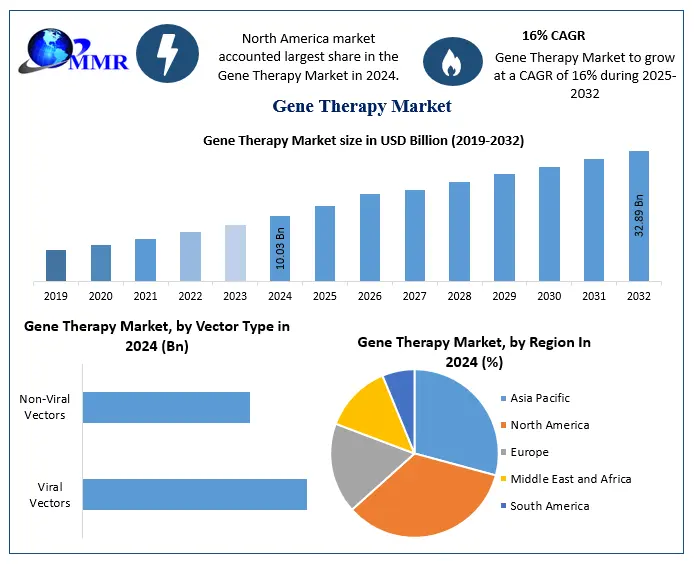 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
Gene Therapy Market Dynamics
Increase In The Demand For Gene Therapy Interventions Drives The Gene Therapy Market
The increase in genetic conditions like cystic fibrosis, sickle cell anemia, and muscular dystrophy increase the demand for gene therapy interventions. The increasing occurrence highlights the need for creative treatment methods. Gene therapy provides possible solutions by focusing on the underlying causes of these disorders, offering a hopeful path for meeting medical needs in affected group.
Innovations like hemophilia gene therapy and CRISPR-Cas9 have transformed the field of gene editing by enhancing accuracy, effectiveness, and cost-effectiveness. This advancement drives expansion in the gene therapy industry by enabling precise genetic alterations for treating diseases. These developments result in better treatments, prompting investments and leading to the development of creative gene therapy options for various medical issues.
High Expenses Associated With Gene Therapy Limits The Growth of Gene Therapy Market
Immune reactions to viral vectors or foreign gene products hamper the effectiveness of gene therapy and lead to negative outcomes. Approaches to deal with the obstacle involve immunomodulatory treatments and modified vectors created to avoid detection by the immune system. It is essential to conquer these reactions to guarantee the safety and efficacy of gene therapy treatments.
The high expenses associated with gene therapy come from complex manufacturing processes, thorough clinical trials, and strict regulatory requirements. The financial obstacles companies face restrict their ability to invest and impede their ability to innovate and enter new markets. It is crucial to find ways to lower the expensive costs of gene therapy to advance the field and make it more accessible to a larger number of patient market report includes information on recent developments, trade regulations, import-export analysis, production analysis, optimizing the value chain, market share, impact of domestic and localized market players, opportunities for emerging revenue pockets, changes in market regulations, strategic analysis for market growth, market size, category market growth, application niches and dominance, product approvals, product launches, geographic expansions, and technological innovations in the market.
Increase In Personalized Treatment Creates Opportunities In The Gene Therapy Market
Gene therapy enables personalized treatment by focusing on genetic mutations and providing customized solutions for each patient. Focusing on each individual's specific genetic characteristics drives the growth of the market by offering personalized and highly efficient treatments, ultimately transforming the field of medical care.
Organizations are more and more creating strategic alliances to create gene therapy products for identifying genetic disorders. Collaborations make it easier to create new technologies and platforms for detecting diseases. Extended contracts allow businesses to provide flexible pricing for gene therapy products, meeting the needs and demand of customers. These collaborations open up chances for both parties to grow and benefit in the market.
Gene therapy provides customized treatment possibilities that are individualized for each patient according to their genetic composition. With the increasing demand for personalized medicine, it anticipated that the gene therapy market see significant growth.
Maintaining The Accuracy For Delivering The Therapeutic Gene To Target Cells Efficiently
One of the biggest challenges is delivering the therapeutic gene to the target cells efficiently. Different delivery methods, such as viral vectors and non-viral vectors, each have their own limitations including immunogenicity, toxicity, and difficulty in targeting specific tissues or cells. The body's immune system recognizes the vectors used in gene therapy as foreign invaders, leading to immune responses that neutralize the therapy or cause adverse reactions in patients. These immune responses limits the effectiveness of repeated dosing. And It indicates that the safety of gene therapy is very crucial. Viral vectors, in particular, carries the risks such as Insertional mutagenesis, it is a condition where the introduced gene disrupts the function of an existing gene, potentially leading to cancer or other adverse effects.
There is a risk of unintended gene expression or disruption of normal gene function, leading to unforeseen side effects. Ensuring that the therapeutic gene integrates into the genome at the intended site and does not interfere the function of other genes is a significant challenge. Gene therapy involves novel technologies and poses unique regulatory challenges. Regulatory agencies needs to balance the need for safety and efficacy with the urgency of bringing potentially life-saving treatments to patients.
Gene Therapy Market Segment Analysis
Based on Type, Viral vectors are developed from viruses that have been altered to translocate therapeutic genes to specific cells. Popular types of viral vectors are retroviruses, adenoviruses, adeno-associated viruses (AAVs), and lentiviruses. Viral vectors are currently the most popular in the gene therapy industry because of their strong ability to efficiently transfer genes to both dividing and non-dividing cells. AAVs (adeno-associated viruses) have become increasingly popular because of their excellent safety record and ability to provide lasting gene expression. Despite their benefits, viral vectors trigger immune reactions, have restricted cargo space, and present dangers like insertional mutagenesis. Non-viral vectors are different delivery systems that do not use viral components. These vectors could consist of DNA without packaging, liposomes, nanoparticles, and polymer-based delivery methods. Non-viral vectors are becoming more popular in the gene therapy industry due to their ability to reduce immune responses, be easily scaled, and accommodate various cargo sizes. Nevertheless, they frequently demonstrate reduced transduction effectiveness when contrasted with viral vectors. Non-viral vectors encounter difficulties in achieving effective gene delivery, including obstacles like degradation by nucleases and restricted cellular uptake.
Non-viral vectors are different delivery systems that do not use viral components. These vectors could consist of DNA without packaging, liposomes, nanoparticles, and polymer-based delivery methods. Non-viral vectors are becoming more popular in the gene therapy industry due to their ability to reduce immune responses, be easily scaled, and accommodate various cargo sizes. Nevertheless, they frequently demonstrate reduced transduction effectiveness when contrasted with viral vectors. Non-viral vectors encounter difficulties in achieving effective gene delivery, including obstacles like degradation by nucleases and restricted cellular uptake.
Gene Therapy Market Regional Insights
North America, especially the United States, leads the gene therapy market Thanks to its strong research infrastructure, supportive regulations, and significant investments in biotech and pharmaceutical sectors. Regulatory approval is given to gene therapy products, are now more commonly used in clinical practice for treating genetic disorders, cancer, and rare diseases. Institutions and companies in North America lead the way in gene therapy research, spearheading advancements in vector development, gene editing technologies, and clinical trial design. Organizations like the FDA are essential for monitoring the progression and authorization of gene therapy products to uphold safety, effectiveness, and quality criteria.
Europe Countries like the UK, Germany, and France are at the forefront of research and clinical development in gene therapy, making Europe a crucial market for the technology. Regulatory approval is granted in Europe for gene therapy products, are now being more commonly used in healthcare systems, especially for treating rare diseases and specific types of cancer. European academic institutions, biotech companies, and pharmaceutical firms are engaged in gene therapy research, working together on clinical trials and investigating innovative therapeutic methods. The European Medicines Agency (EMA) monitors gene therapy products in the European Union to ensure they meet strict regulatory standards for safety, effectiveness, and quality.
Gene Therapy Market Competitive Landscapes
• In December 2023, Libmeldy received approval from the Swiss Agency for Therapeutic Products to treat early-onset metachromatic leukodystrophy, marking a significant advancement in addressing the rare disorder's therapeutic needs.
• In May 2023, Krystal Biotech's VYJUVEK gene therapy gained approval for treating Dystrophic Epidermolysis Bullosa, offering hope to patients with the debilitating condition.
• In January 2023, Voyager Therapeutics and Neurocrine Biosciences initiated a strategic collaboration to commercialize and develop Voyager's GBA1 program and other innovative gene therapies targeting neurological disorders, promising advancements in the critical field of medicine.
Gene Therapy Industry Ecosystem
Gene Therapy Market Scope: Inquiry Before Buying
| Gene Therapy Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2024 | Forecast Period: | 2025-2032 |
| Historical Data: | 2019 to 2024 | Market Size in 2024: | USD 10.03 Billion |
| Forecast Period 2025 to 2032 CAGR: | 16% | Market Size in 2032: | USD 32.89 Billion |
| Segments Covered: | by Delivery Method | In Vivo Ex Vivo |
|
| by Vector Type | Viral Vectors Non-Viral Vectors |
||
| by Therapeutic Area | Oncology Neurological Disorders Cardiovascular Diseases Others |
||
| by Route of Administration | Intravenous Others |
||
| by End User | Hospitals and Clinics Academic and Research Institutes Biotechnology and Pharmaceutical Companies Others |
||
Gene Therapy Market by Region
North America (United States, Canada, and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, and the Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Indonesia, Malaysia, Australia, Vietnam, Taiwan, Bangladesh, Pakistan, and the Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria, and the Rest of ME&A)
South America (Brazil, Argentina Rest of South America)
Gene Therapy Market Key Players
1. Organogenesis
2. JCR Pharmaceuticals
3. Japan Tissue Engineering
4. AVITA Medical
5. CHIESI Farmaceutici
6. Stempeutics Research
7. Biosolution
8. MolMed
9. APAC Biotech
10. Gilead Sciences
11. Medipost
12. JW CreaGene
13. Vericel
14. Takeda Pharmaceutical Company
15. Novartis AG
16. Osiris Therapeutics
17. AnGes
18. Tego Sciences
19. Dendreon
20. CollPlant
Frequently Asked Questions:
1] What segments are covered in Global Gene Therapy Market report?
Ans. The segments covered in Global Gene Therapy Market report are based on method, Vector Type and End User.
2] Which region is expected to hold the highest share in the Global Gene Therapy Market?
Ans. North America is expected to hold the highest share in the Global Gene Therapy Market.
3] Who are the top key players in the Global Gene Therapy Market?
Ans. CHIESI Farmaceutici, Stempeutics Research, Biosolution, APAC Biotech, Gilead Sciences, Medipost, JW CreaGene, Vericel, Takeda Pharmaceutical Company are the key players in Gene Therapy Market.
4] Which segment holds the largest market share in the Global Gene Therapy market by 2032?
Ans. Spinal Muscular Atrophy (SMA) Indication segment will hold the largest market share in the Global Gene Therapy market by 2032.
5] What is the market size of the Global Gene Therapy market by 2032?
Ans. The market size of the Global Gene Therapy market is USD 32.89 Bn. by 2032.
6] What was the Global Gene Therapy Market size in 2024?
Ans: The Global Gene Therapy Market size was USD 10.03 Billion in 2024.
