Amniotic Products Market – Industry Structure Evaluation, Demand Drivers Analysis, Regional Growth Analysis and Identification, Competitive Positioning Review & Global Market Size Forecast to 2030
Overview
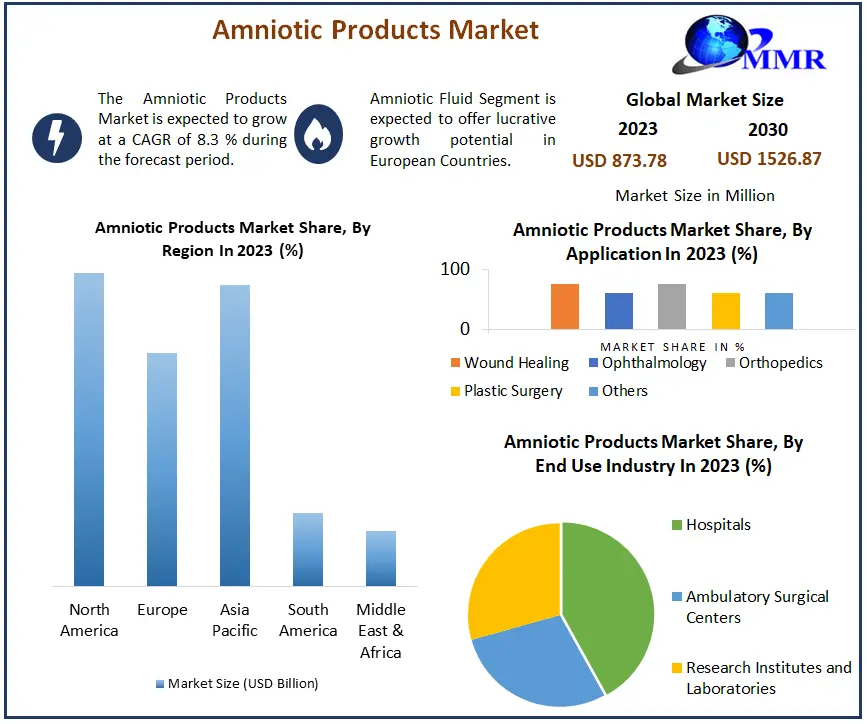
The Amniotic Products Market size was valued at USD 873.78 Million in 2023 and the total Amniotic Products revenue is expected to grow at a CAGR of 8.3% from 2024 to 2030, reaching nearly USD 1526.87 Million by 2030.
Amniotic products are biological materials sourced from the amniotic fluid or membrane of the gestational sac, prized for their regenerative properties and used in various medical applications like wound healing and tissue repair. The rising prevalence of chronic diseases, increasing awareness about regenerative medicine, and the growing adoption of minimally invasive procedures driving the growth of Amniotic Products Market. Amniotic products, derived from the amniotic membrane or amniotic fluid, offer various therapeutic benefits such as anti-inflammatory, antimicrobial, and wound healing properties. These products find extensive applications in wound care, ophthalmology, orthopedic, and soft tissue repair. Advancements in technology have led to the development of innovative products with enhanced efficacy, further propelling market growth. Key players in the market are actively engaged in strategic initiatives such as mergers and acquisitions, collaborations, and product launches to strengthen their market position and expand their product portfolios. For instance, in recent years, major players have focused on launching novel amniotic tissue-based products, investing in research and development activities, and expanding their distribution networks to capitalize on emerging market opportunities.
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
The market is experiencing a trend towards the development of personalized therapies and the integration of biotechnology and stem cell technology, offering immense growth potential. Regulatory challenges associated with product approval and reimbursement policies may hinder Amniotic Products Market growth to some extent. Nevertheless, with increasing investments in healthcare infrastructure and the growing demand for advanced regenerative therapies, the Amniotic Products Market is poised for significant expansion in the coming years.
Amniotic Products Market Dynamics:
Growing trend towards minimally invasive procedures is boosting the Amniotic Products Market
The increasing prevalence of chronic diseases such as diabetic foot ulcers and osteoarthritis is acting as a significant driver for the growth of the Amniotic Products Market. With approximately 33% of diabetic patients developing foot ulcers, companies like MiMedx Group, Inc. and Osiris Therapeutics, Inc. are experiencing heightened demand for their amniotic tissue-based products, particularly for wound healing in diabetic patients, thus propelling market expansion. The aging global population, susceptible to age-related conditions like osteoarthritis and age-related macular degeneration (AMD), is further fueling the demand for amniotic products. For instance, AMD affects over 2 million Americans aged 50 and above, leading to the development of innovative amniotic membrane-based products by companies like Amnio Technology, LLC, for ocular surface reconstruction and AMD treatment, thereby contributing to market growth.
The trend towards minimally invasive surgeries and treatments is also driving the market for amniotic products, especially in orthopedic procedures where techniques utilizing amniotic membrane allografts offer quicker recovery times and reduced post-operative complications. Organogenesis Inc.'s offerings in this domain are gaining popularity, bolstering market growth. Additionally, the increasing awareness of the benefits of regenerative medicine is spurring demand for amniotic products as patients seek alternatives to traditional treatments. Skye Biologics, Inc., for instance, is witnessing heightened demand for its amniotic fluid-based products for regenerative medicine applications like tissue repair and wound healing, contributing to market expansion.
Technological advancements are playing a crucial role in enhancing the efficacy of amniotic products, with companies like Surgenex, LLC, leveraging advanced processing techniques to produce products with higher concentrations of growth factors, thus accelerating tissue repair and regeneration. Furthermore, rising investments in research and development activities aimed at exploring new therapeutic applications of amniotic products are fostering innovation and market growth. Companies such as Katena Products, Inc. are investing in clinical trials to evaluate the efficacy of amniotic membrane grafts for treating conditions like glaucoma and corneal ulcers, driving the evolution and expansion of the market landscape.
High-cost treatment Hindering the market growth
The stringent regulatory requirements for the approval of amniotic products emerge as a significant constraint impeding market growth. Instances such as the FDA's warning letters to companies like Amniotic Therapies LLC for marketing unapproved products highlight compliance challenges, leading to setbacks in market progression. Moreover, the high cost associated with amniotic products and related procedures presents affordability hurdles for both patients and healthcare systems. For instance, the prohibitive pricing of amniotic membrane grafts for ophthalmic surgeries, exemplified by Bio-Tissue, Inc.'s offerings, restricts market penetration and adoption, exacerbating the growth constraints.
Limited insurance coverage and reimbursement policies for amniotic products further hamper patient access and adoption rates. With Medicare's non-reimbursement stance on certain amniotic tissue-based injections, patients face financial barriers, as evidenced by cases documented by the American Medical Association, hindering market expansion. The absence of standardized protocols and robust quality control measures in amniotic product manufacturing contributes to variability in efficacy and safety, exemplified by instances of contamination and inconsistency reported in FDA warning letters to companies like Liveyon LLC, emphasizing the pressing need for improved industry standards and regulatory oversight. Additionally, the dearth of long-term clinical data on the safety and efficacy of amniotic products raises concerns among healthcare providers and patients alike. The FDA's safety communication regarding potential risks associated with certain amniotic fluid-derived products underscores the prevailing uncertainty and hesitancy among stakeholders, further restraining market growth.
Amniotic Products Market Segment Analysis:
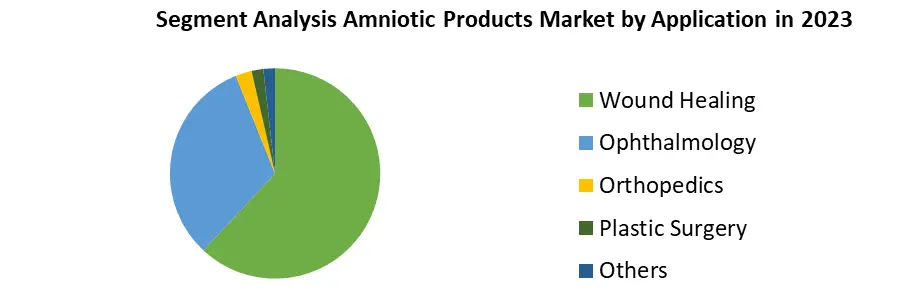
Based on Application, Wound healing segment dominated the Amniotic Products Market in 2023, benefiting from the regenerative properties of amniotic products, particularly in chronic wound management and tissue repair. Ophthalmology follows closely, with increasing adoption in ocular surface reconstruction and the treatment of conditions like dry eye syndrome and corneal defects. Orthopedics utilizes amniotic products for their anti-inflammatory and tissue regenerative properties, aiding in the management of musculoskeletal injuries and conditions such as osteoarthritis. In plastic surgery, amniotic products find applications in tissue augmentation, wound closure, and scar reduction, contributing to post-operative outcomes. Other applications encompass a diverse range of specialties, including gynecology, dentistry, and urology, albeit with comparatively lower adoption rates.
Amniotic Products Market Regional Insights:
North America Dominated the Amniotic Products Market
North American region dominated the Amniotic Products Market in 2023, driven by substantial healthcare expenditure, robust healthcare infrastructure, and recent advancements by industry stakeholders. As per data released by the United States Centers for Medicare & Medicaid Services (CMS) in February 2023, national healthcare spending increased by 2.7% to USD 4.3 trillion in 2021 compared to the preceding year.
The North America Amniotic Products Market is expected growth with the anticipated impact of technological innovations by Amniotic Products Market leaders. For instance, TissueTech received clearance from the Food and Drug Administration (FDA) in March 2021 to conduct a Phase 2 study employing morselized Cryopreserved Amniotic Membrane (CAM) and Cryopreserved Umbilical Cord (CUC) Investigational New Drug (IND) TTBT01. Similarly, in May 2021, Vivex Biologics, Inc., a prominent figure in regenerative medicine, introduced Viagenex, a suite of amniotic allografts meticulously designed to maintain the intrinsic mechanical properties of amniotic tissue while harboring a rich assortment of extracellular matrix, growth factors, and cytokines.
Amniotic Products Market Scope: Inquire before buying
| Global Amniotic Products Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2023 | Forecast Period: | 2024-2030 |
| Historical Data: | 2018 to 2023 | Market Size in 2023: | US 873.78 Mn. |
| Forecast Period 2024 to 2030 CAGR: | 8.3 % | Market Size in 2030: | US 1526.87 Mn. |
| Segments Covered: | by Product Type | Amniotic Membrane Amniotic Fluid Amniotic Tissue |
|
| by Application | Wound Healing Ophthalmology Orthopaedics Plastic Surgery Others |
||
| by End-Use Industry | Hospitals Ambulatory Surgical Centers Research Institutes and Laboratories |
||
Amniotic Products Market, by Region
North America (United States, Canada and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan and Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A)
South America (Brazil, Argentina Rest of South America)
Amniotic Products Market Key Players:
Major Contributors in the Amniotic Products Industry:
1. Tissue Regenix Group plc, York, United Kingdom
2. Biotissue AG, Zug, Switzerland
3. Celestica HealthTech (formerly known as IZON Group), Auckland, New Zealand
4. CellCura ASA, Oslo, Norway
5. CollPlant Biotechnologies Ltd., Rehovot, Israel
6. Medskin Solutions Dr. Suwelack AG, Billerbeck, Germany
7. Corestem, Inc., Seoul, South Korea
8. Cryoport Asia Pacific Pte Ltd., Singapore
9. Genesis BPS Pty Ltd., Sydney, Australia
10. Japan Tissue Engineering Co., Ltd., Gamagori, Aichi Prefecture, Japan
11. LifeCell International Pvt. Ltd., Chennai, Tamil Nadu, India
12. CryoCell International, Inc., Oldsmar, Florida, USA
13. MiMedx Group, Inc., Marietta, Georgia, USA
14. Organogenesis Inc., Canton, Massachusetts, USA
15. Osiris Therapeutics, Inc., Columbia, Maryland, USA
FAQs:
1] Which region is expected to hold the highest share in the Global Amniotic Products Market?
Ans. North America region is expected to hold the highest share in the Amniotic Products Market.
2] What is the market size of the Global Amniotic Products Market by 2030?
Ans. The market size of the Amniotic Products Market by 2030 is expected to reach US$ 1526.87 Million.
3] What is the forecast period for the Global Market?
Ans. The forecast period for the Market is 2024-2030.
4] What was the market size of the Global Market in 2023?
Ans. The market size of the Market in 2023 was valued at US$ 873.78 Million.