Drug Repurposing Market Size by Type Of Approach, Therapeutic Area, Drug Molecules, Region, Industry-Wide Analysis, Competitive Landscape Assessment & Long-Term Forecast to 2032
Overview
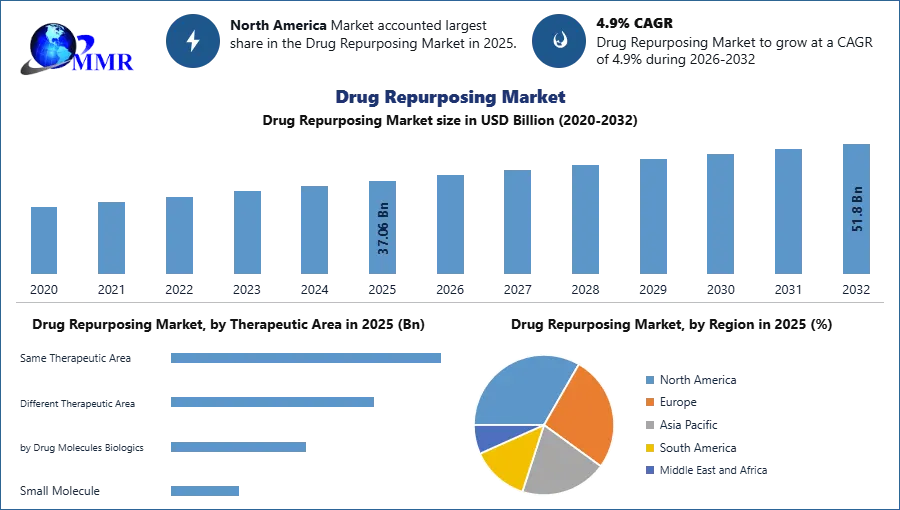
The Drug Repurposing Market was valued at USD 37.06 Bn in 2025 is expected to grow at a CAGR of 4.9% from 2025 to 2032 and reach USD 51.80 Bn by the end of 2032.
Drug Repurposing Market Overview:
Drug repurposing, also recognized as drug repositioning or drug reprofiling, is a tactical methodology in drug discovery that recognizes new therapeutic practices for current or investigational drugs. By leveraging prior clinical data, such as off-label prescriptions or biological experiments like gene expression profiling, drug repurposing deals a quicker and cost-effective substitute to traditional drug discovery. This method quickens the improvement of treatments for unmet medical necessities.
Drug Repurposing acknowledged as a worldwide approach for addressing neglected diseases, IT offers numerous benefits, including fewer clinical trial stages, lesser costs, and earlier Drug Repurposing Market access. Current supply chains enable formulation and delivery, while drug combinations improve treatment effectiveness. Moreover, repurposing supports the encounter of new mechanisms of action, overwhelming early-stage research obstacles.
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
Developments in genomics, network biology, and chemoproteomics have additionally supported targeted repurposing by recognizing gene interfaces. Drugs with genetically supported targets are twice as expected to thrive.
According to the National Institutes of Health, above 35% of transformative drugs sanctioned by the US FDA are repurposed. In recent years, antifungal agents such as ketoconazole and clotrimazole have been examined for anti-trypanosome therapy. Effective antimicrobial repositioning has also directed the discovery of broad-spectrum therapeutics effective against several infections.
Drug repositioning monitors three principal methodologies: disease-centric, target-centric, and drug-centric. The disease-centric method, presently holding around 42.6% Drug Repurposing Market share, recognizes contacts between old and new signals. Target-centric repositioning connects recognized drugs and their identified targets to new indications, although drug-centric approaches associate prevailing drugs with new targets and their particular signals.
With the growing worldwide population and growing disease occurrence, drug inventors face the stress of accelerating time to Drug Repurposing industry while lessening R&D costs. Drug repurposing has increased popularity for its shorter development timelines, recognized safety profiles, and higher success rates. In 2020, 12 out of 28 drugs accepted by the US FDA were repurposed, contributing to 25%-40% of annual pharmaceutical revenue. Especially, many COVID-19 treatments, containing chloroquine, hydroxychloroquine, and remdesivir, were repurposed.
Contract research organizations (CROs) play a crucial role in supporting drug inventors through clinical trials, drug discovery operations, and consultancy facilities. Outsourcing actions in drug repurposing are anticipated to rise by 20%-25%, contributing to significant Drug Repurposing Market development in the upcoming years.
Global Drug Repurposing Market Dynamics:
Cost-Effectiveness to boost Drug Repurposing Market Growth: The Benefits of Drug Repurposing in Pharmaceutical Expansion
Drug repurposing deals pharmaceutical companies an additional proficient and effective preference compared to traditional drug development. Evolving new medications from scratch is time-consuming and costly, however repurposing present drugs for new uses is faster, less expensive, and often hastens regulatory approval. This lessens financial stress and conveys treatments to the market quicker. Moreover, supportive policies and incentive programs from governments and philanthropic organizations in together developed and developing countries, alongside better government investments in healthcare and medical segments, promote and drive the development of drug repurposing initiatives.
AI-Powered Drug Repurposing and Increasing Therapeutic Applications to boost Drug Repurposing Market growth
Growing therapeutic applications through drug repurposing offers companies with chance to raise their target patient base, arrive at new markets, and exploit the usage of prevailing pharmaceutical compounds. AI and bioinformatics tools have altered drug repurposing by empowering the investigation of huge datasets, containing genomic and proteomic data, to proficiently and precisely recognize potential drug candidates. This expertise improves the drug discovery practice, permitting researchers to forecast new therapeutic applications for prevailing drugs more quickly. Furthermore, developments in Deep Learning, Machine Learning (ML), and AI are motivating the efficient identification of repurposed drugs for polypharmacology. Joined with improved R&D investments from governments and leading industry players, predominantly in cancer treatment development, these trends are estimated to enhance the Drug Repurposing Market profitability.
Driving Therapeutic Innovation: The Potential of Drug Repurposing:
Drug repurposing provides a quicker and safer result for infrequent diseases and orphan conditions by operating prevailing drugs with well-known safety profiles. By significantly decreasing development risks and timelines, it speeds up Drug Repurposing industry entrance. Regulatory support through streamlined authorization practices, financial incentives, and regulatory flexibility boosts drug accessibility for rare conditions. Moreover, drug repurposing bolsters pandemic readiness by permitting the quick identification of antiviral properties in prevailing drugs. The COVID-19 pandemic emphasized the prominence of this method, with repurposed drugs rapidly measured for their effectiveness. With fewer novel drugs presented in recent years, pharmaceutical companies are progressively turning to repositioning to encounter urgent medical demands. By concentrating on earlier marketed drugs, those without exclusive indications, and withdrawn drugs because of budget constrictions, companies lessen early-stage development costs and exploit return on investment (ROI). This attitude is predominantly noticeable in oncology, where drugs are frequently tested on tumors with like characteristics, leading to lengthened indications. Examples like Merck’s Keytruda (pembrolizumab) and Novartis’ Arzerra (ofatumumab) demonstrate the achievement of drug repurposing. Keytruda is now approved for 14 cancer types, though Arzerra is underestimation for numerous sclerosis treatments, representing how drug repositioning reveal new therapeutic.
Directing Challenges and Limitations in Drug Repurposing:
A foremost challenge in drug repurposing is directing intellectual property rights. Present drugs is thriving by patents, and legal difficulties arise when repurposing them for new indications. This lead to extended negotiations, and legal disputes, or even inhibit repurposing overall, thus restrictive Drug Repurposing Market potential. Financial incentives for drug repurposing also be less appealing associated with emerging new drugs, as there are no exclusive rights and the threat of generic competition. These concerns disappoint pharmaceutical companies from investing substantial resources into repurposing struggles, hindering the investigation of prevailing drugs for new therapeutic applications.
Exposing Opportunities: AI-Driven Drug Repurposing for Market Enlargement
Growing market share of Pharmaceutical companies is deliberately repositioning present drugs into new therapeutic areas, predominantly underserved diseases like neurodegenerative conditions, erratic cancers, and infectious diseases, leveraging AI and bioinformatics to forecast drug-protein interactions and recognize molecular relationships, by this means to speed up discovery. Applying real-world records from EMRs and claims data, joined with advanced NLP to investigate vast datasets, enriches perceptions into off-label uses and novel drug applications, improving drug portfolio management and spreading the development of approved drugs. This AI-driven method delivers cost-effective treatment explanations for infrequent and neglected diseases, supported by government enterprises and funding, generating significant market development potential.
Global Drug Repurposing Market Segmentation:
Segmentation in Drug Repositioning: Exploring Disease-Centric, Target-Centric, and Drug-Centric Approaches
Based on Type of approach the market is segmented into disease-centric, Target centric, and drug-centric approaches. The disease-centric approach has Dominated the market in 2024 and is expected to hold the largest market share over the forecast period.
In the drug repurposing market, the disease-centric approach is predominant, accounting for more than 60% of cases, as it identifies the relationship between existing drugs and new indications, thereby decreasing development time and cost. This approach allows researchers and pharmaceutical companies to concentrate on s definite disease and underlying mechanisms, leading to more targeted and efficient drug development stages. In comparison, the target-centric approach contributes nearly 30% to 35% of cases while the drug-centric approach is less common.
Drug Repurposing in Segmentation: Biologics vs. Small Molecules
Based on the Type of Drug Molecule the market is segmented into Biologics and small molecule. The Biologic Type has Dominated the market in 2025 and is expected to hold the largest Drug Repurposing Market share over the forecast period. concerning
Developments in biotechnology and analytical methods have been major contributors to Biologics segment's volatile growth, and both stay to do so as it remains to pick up speed.
Its explosive growth over the last ten years has been driven by the rapid development of biologics-based medications and the enlarged association of businesses. As drugs, biologics have several benefits over small molecules. Unlike small molecules, which commonly have non-specific side effects that call for non-definite solutions, biologics have superior specificity concerning target specificity, permitting side effects that support therapeutic outcomes more rapidly than is usually the case.
Monoclonal antibodies (mAbs), such as trastuzumab (Herceptin), is one example of this. Breast cancer that is HER2-positive is treated with trastuzumab. It primarily targets the HER2 receptor, which is present on the surface of some breast cancer cells in huge numbers. Trastuzumab merely targets cancer cells, as opposed to small molecule chemotherapy medications that harm together diseased and healthy cells. This helps to lessen common side effects of chemotherapy, like nausea and hair loss.
Global Drug Repurposing Market Regional Analysis:
In 2025, North America has undergone noteworthy progression in the drug repurposing segment, driven by improved research and development accomplishments directed at repurposing prevailing medications. This rush in R&D efforts has contributed to the region's rising Drug Repurposing industry share. Moreover, the subdivision's resilience remains obvious despite the ongoing pandemic, as healthcare professionals have progressively supported the use of supplements and repurposed drugs, solidifying the market's importance. The report covers major countries in this region, including the US and Canada.
Europe holds the second-largest Drug Repurposing Market share in the drug repurposing division. The region's strong R&D proficiencies, government support for innovation, and well-established regulatory context have complete it a crucial player in the industry. Amongst the European countries, the UK has experienced the fastest development, although Germany preserves the major market share. Further noteworthy providers include France, Italy, and Spain.
Global Drug Repurposing Market Competitive Landscape
To boost market existence, businesses in the pharmaceutical division are engaging a range of strategies, which contains partnerships, mergers and acquisitions, strategic associations, regional extension, and the introduction of new products and services.
Novartis AG, headquartered in Basel, Switzerland, is one of the prime pharmaceutical companies worldwide. In 2025, Novartis experienced a 4.8% development in sales, acquiring fourth place in PharmExec's Top 50 Companies survey. The Novartis collaborated with Ares Genetics to explored AI-based drug repurposing for antimicrobial resistance. Moreover, in Branaplam, originally for spinal muscular atrophy, was investigated for Huntington’s disease but discontinued due to safety concerns.
Biovista Inc., a private drug development services firm based in Charlottesville, Virginia, specializes in drug repositioning and de-risking. Its main services include disease cohort analysis, adverse event prediction, and clinical hold analysis. Moreover, Biovista is Expanding its proprietary platform to grow pharmaceutical repositioning programs pointing to oncology, diabetes, obesity, ophthalmic diseases, and central nervous system (CNS) disorders.
AbbVie Inc. has effectively repurposed Ritonavir (Norvir), originally developed as an HIV protease inhibitor, to boost the efficacy of other HIV drugs.
Large pharmaceutical companies, including Pfizer, Novartis, Johnson & Johnson, and Roche, successfully influence their wide-ranging resources to discover new applications for prevailing drugs. They stabilize their efforts through big data analysis, associations with contract research organizations (CROs), and technology-driven methods. Although mid-sized companies also allocate their focus through these areas, they may highlight specific technologies or specialized CRO services.
In comparison, smaller firms often focus more deeply on big data and technology-based techniques, showcasing their power in specialized analysis and innovation with less dependence on traditional CRO models. Notable companies excelling in fields like AI-driven drug discovery, preclinical services, and progressive data analytics include Recursion Pharmaceuticals, BenevolentAI, Charles River Laboratories, WuXi AppTec, GNS Healthcare, and NuMedii.
Drug Repurposing Market Scope: Inquire Before Buying
| Drug Repurposing Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 37.06 USD Billion |
| Forecast Period 2026-2032 CAGR: | 4.9% | Market Size in 2032: | 51.8 USD Billion |
| Segments Covered: | by Type Of Approach | Disease-centric Target-centric Drug-centric |
|
| by Technology Used | Computational Drug Repurposing Artificial Intelligence & Machine Learning Network-based Analysis High-throughput Screening Bioinformatics Platforms |
||
| by Indication / Disease Type | Oncology Neurological Disorders Infectious Diseases Cardiovascular Diseases Rare Diseases Metabolic Disorders |
||
| by End User | Pharmaceutical Companies Biotechnology Companies Contract Research Organizations (CROs) Academic & Research Institutes Government Research Organizations |
||
Global Drug Repurposing Market, by Region
North America (United States, Canada and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Russia and Rest of Europe)
Asia Pacific (China, S. Korea, India, Japan, Australia, Indonesia, Malaysia, Philippians, Thailand, Vietnam, Rest of Asia Pacific)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria, Rest of MEA)
South America (Brazil, Argentina, Colombia, Chile, Rest of South America)
Key Players of the Global Drug Repurposing Market includes:
1. Novartis – Switzerland
2. Takeda Pharmaceutical Company – Japan
3. Teva Pharmaceutical Industries – Israel
4. Gilead Sciences – United States
5. Eli Lilly and Company – United States
6. AstraZeneca – United Kingdom/Sweden
7. Pfizer – United States
8. Merck & Co. – United States
9. Bristol-Myers Squibb – United States
10. Johnson & Johnson – United States
11. Roche – Switzerland
12. GlaxoSmithKline – United Kingdom
13. Sanofi – France
14. AbbVie – United States
15. Bayer AG – Germany
16. Eisai Co., Ltd. – Japan
17. Medivir AB – Sweden
18. RxAll Inc. – United States
19. Summit Therapeutics – United States
20. Akeso Biopharma – China
21. Little Green Pharma – Australia
22. Shijiazhuang Yuanmai Biotechnology Co. Ltd. – China
23. Celltrion – South Korea
24. Aragen Life Sciences – India
25. Mylan – United States
26. Eva Pharma – Egypt
27. Ferozsons Laboratories – Pakistan
28. Chinook Therapeutics – United States
29. Gyroscope Therapeutics – United States
30. Cambrian Biopharma – United States
