Autologous Cell Therapy Market Size by Product, Type, Application, End-User, Region – Revenue Pool Analysis, Margin Structure Assessment, Capital Flow Trends, Competitive Benchmarking & Forecast to 2032
Overview
Autologous Cell Therapy Market size was valued at 7.11Bn in 2025 and the total Global Autologous Cell Therapy Market revenue is expected to grow at a CAGR of 21.54% from 2026 to 2032, reaching nearly USD 27.86 Bn by 2032.
Autologous Cell Therapy Market Overview:
Autologous cell therapy is a personalized treatment that uses a patient's own cells, which are harvested, processed (often genetically modified or expanded ex vivo), and then reintroduced into the same patient to treat diseases such as cancers, autoimmune disorders, or tissue damage. This approach reduces immune rejection risks and enables analog therapy. Autologous cell therapy market is a rapidly growing due to focus on individual remedies using his own cells, mainly for oncology (eg, CAR-T) and regenerative therapy (eg, chondocytes, stem cells).
Gilead Sciences (Kite Pharma), Novartis, and Bristol Myers Squibb are leading Global autologous cell therapy market, which leads to more than 75% of the global CAR-T revenue in 2024, respectively with Yasrata/Ticcortus, Kimiyah, and Breyanzi/Abcama. North America remains innovation leader (55% market share), operated by world class research centres and streamlines FDA approval, while Europe and Asia-Pacific Autologous cell therapy show thrilling development with expansion of manufacturing hubs and progressive regulatory structures. Hospital and Special Treatment Centres demand 70% of autologous cell therapy market led by adoption of CAR-T, while academic/research institutes contribute 20% through clinical trials and regenerative medical applications.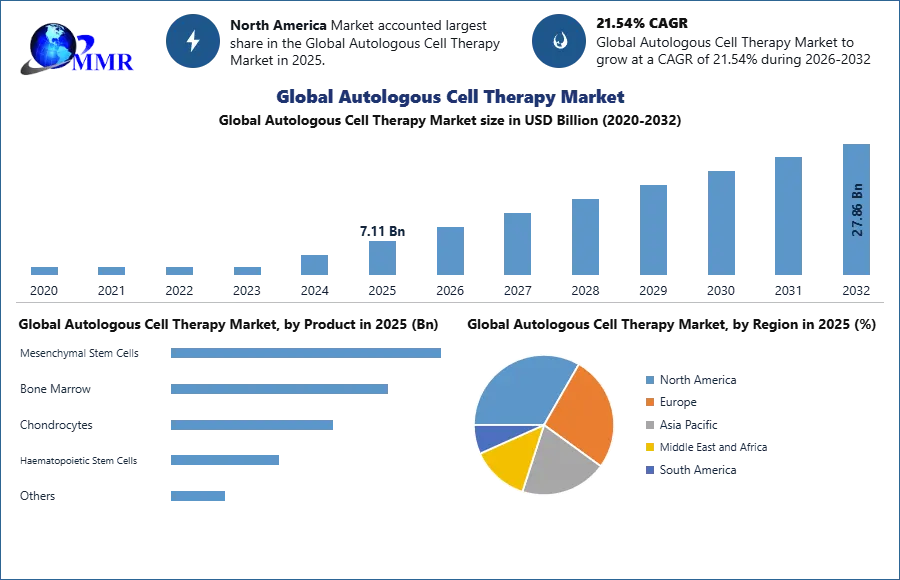 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
Autologous Cell Therapy Market Dynamics:
Growing Number of Diseases to Drive Autologous Cell Therapy Market
The prevalence of chronic diseases such as autoimmune diseases, cancer, blood disorders, and others is increasing. A rise in the number of people suffering from chronic diseases is also driving the autologous cell therapy market growth. MMR estimated the incidence of 38 cancers in 185 countries; there were an estimated 19,292,789 new cases of cancer diagnosed worldwide in 2020. Males accounted for 10,068,305 of the total diagnosed cancer cases, while females accounted for 9,229,484 cases. Male cancer cases were expected to reach 15,589,096 by 2040, while female cancer cases were expected to reach 13,310,846 by 2040.
The increasing incidence of cancer among the global population is expected to drive market growth. Anemia, bleeding disorders such as hemophilia and blood clots, and blood cancers such as leukemia, lymphoma, and myeloma are examples of common chronic blood disorders. According to the Centers for Disease Control and Prevention's May 2022 update, sickle cell disease (SCD) affects approximately 100,000 Americans. It affects about 1 in every 365 Black or African American births. Effective treatment is the most effective way to save a patient's life who has a blood cell disorder.
Other factors are also playing critical roles in boosting the autologous cell therapy market forward, including ongoing drug development for new applications, which are expected to further propel the autologous cell therapy market's growth. During the forecast period, the market will grow due to an increase in the number of clinical trials and an increase in the global adoption of cell therapies.
Government support and initiatives to Boost the Autologous Cell Therapy Market
With multiple government efforts and approvals to perform clinical studies of biomaterials, the stem cell treatment category is expected to grow at the quickest CAGR. According to the Organization for Economic Cooperation and Development (OECD), Denmark's average per capita healthcare spending in 2020 was around USD 5,600 Bn.
Additionally, the Alzheimer Europe 2019 study indicates that the number of persons with dementia in Italy in 2018 was about 1,279,366 and that this figure is expected to quadruple to roughly 2,247,715 in 2050. Similarly, dementia will account for 4.13% of the general population in 2050, up from 2.12% in 2018. As the number of people suffering from dementia is predicted to increase in the near future, the autologous cell therapy market is likely to grow. A large number of research institutes, universities, and businesses from Europe, the United States, and Canada are also undertaking clinical studies for the use of stem cell-based therapeutics in nerve repair and regeneration. These countries are considered to have strong government support, a well-developed healthcare system, and a worldwide presence. As a result of the aforementioned factors, the category is expected to grow substantially during the forecast period.
Lack of Skilled Professionals to Restrain the Autologous Cell Therapy Market
Globally, there is a chronic shortage of skilled healthcare providers which hampers the market. India is dealing with serious shortages and unequal distributions of skilled healthcare personnel. According to the World Health Organization, around 28% of presently employed health professionals do not have the essential credentials. As the need for healthcare services grows and the sector embraces new technologies, there is a scarcity of personnel with digital skills, limiting their ability to manage nerve repair and regeneration technology.
The Covid-19 pandemic has also demonstrated that healthcare resources committed to pandemic management in urban and rural regions compete directly with critical medical, surgical, interventional, and interventional diagnostic services for other disorders, such as neurological ailments.
Increased Technological Advancement to Create Opportunities in the Autologous Cell Therapy Market
Government initiatives for cell therapy technology advancement and R&D were 46% in 2023 and 52% in 2024, which is driving the growth of the market. Furthermore, autologous anti-CD19 CAR T-cell therapy, such as Axicabtagene Ciloleucel, received FDA approval on April 1, 2022, for the treatment of adults with large B-cell lymphoma. Brexucabtagene autoleucel was approved by the Food and Drug Administration (FDA).
In 2024, for the treatment of mantle cell lymphoma. These approvals expand the market opportunity for autologous cell therapy. Furthermore, these approvals for autologous cell therapy boost the market growth. Slowly progressive and degenerative neurological diseases like Parkinson's disease (PD) and Alzheimer's disease (AD) prompted the search for new treatments. In recent years, research has shifted toward transplant therapy for these diseases, as well as autologous cell therapy, which is emerging to alleviate symptoms and even reverse disease progression. Thus, technological advancements for decentralizing autologous cell therapy manufacturing are expected to significantly benefit the market forecast.
Autologous Cell Therapy Market Segment Analysis:
Based on Source, Autologous Cell Therapy Market is segmented into Mesenchymal Stem Cells, Bone Marrow, Chondrocytes, Haematopoietic Stem Cells, etc. In 2024, the hematopoietic stem cells (HSCs) segment dominates the market, holding over 40% share driven primarily by their critical role in treating blood cancers (e.g., leukaemia, lymphoma) and bone marrow disorders. CAR-T therapies (like Brexucabtagene autoleucel) and autologous stem cell transplants (ASCTs) fuel this dominance, with over 25,000 annual procedures globally. Based on the Application, in terms of revenue, the cancer segment dominated the market in 2024, with a share of more than XX.XX%. The market is divided into cancer, neurodegenerative disorders, cardiovascular disorders, autoimmune disorders, orthopedics, wound healing, and others. Cancer is expected to kill nearly 10 million people by 2020, according to the WHO. MMR analyzed that there were 2.3 million new cancer cases diagnosed and 608,790 cancer deaths in the United States in 2023. With the growing burden of cases and demand for treatment and therapy, autologous cell therapy is expected to find its greatest demand in cancer applications.
Based on the Application, in terms of revenue, the cancer segment dominated the market in 2024, with a share of more than XX.XX%. The market is divided into cancer, neurodegenerative disorders, cardiovascular disorders, autoimmune disorders, orthopedics, wound healing, and others. Cancer is expected to kill nearly 10 million people by 2020, according to the WHO. MMR analyzed that there were 2.3 million new cancer cases diagnosed and 608,790 cancer deaths in the United States in 2023. With the growing burden of cases and demand for treatment and therapy, autologous cell therapy is expected to find its greatest demand in cancer applications.
Based on the End-use, in terms of revenue, hospitals and clinics dominated the autologous cell therapy market in 2024, accounting for more than XX.XX% of the total. The market is divided into hospitals and clinics, ambulatory centres, academics and research, and others based on end-use. The rising prevalence of cancer, autoimmune diseases, cardiovascular disease, and neurodegenerative disorders has increased the demand for treatment and therapies.
The academics & research segment is expected to witness growth at a CAGR of XX.XX% over the estimated timeframe. Autologous cell therapy is garnering an increasing amount of interest worldwide and is being extensively studied. It is an emerging field of science that uses the living cellular to treat and prevent disease. Academics and research mostly conduct research in bone marrow, epidermis, mesenchymal stem cells, hematopoietic stem cells, and chondrocytes.
For instance, in October 2024, Allogene Therapeutics mentioned the new survey of the U.S academic institutes that specialized in the administration of CAR T, which discovered 82.5% of respondents agreed that CAR T therapies have assisted in the management of cancers.
Autologous Cell Therapy Market Regional Insights:
North America dominated the market in 2025, accounting for xx% of total revenue. The market in this region is highly competitive because most of the major market players, such as Brain Storm Cell Therapeutics and Bristol Myers Squibb, are present. Furthermore, the growing geriatric population, numerous immunological benefits, and the reduced risk of autologous cell therapy rejection have created new growth opportunities for the autologous cell therapy market. With rising disease burden, strong research activities in autologous cell therapies, the presence of a well-established biotechnology sector, developing healthcare facilities, and growing awareness for cell therapies, Asia Pacific is expected to grow at the fastest CAGR of xx% during the forecast period. For example, the Atara Biotherapeutics phase 1 clinical study was conducted in the company's collaboration with the University of Queensland and the QIMR Berghofer Medical Research Institute in November 2018. It describes outcomes in patients with progressive multiple sclerosis who received autologous EBV-specific T-cell immunotherapy from Atara.
Autologous Cell Therapy Competitive Landscape:
In 2024, Bristol Myers Squibb (BMS) strengthened its position as a top 3 leader in the market, operated by its CAR-T portfolio- Breyanzi (for large B-cell lymphoma) and Abecma (for multiple myeloma), which collectively develops USD 1.8 billion in revenue, 20% YoY development. BMS expanded Breyanzi’s superior safety profile (Grade 3+ neurotoxicity in just 4% of patients vs. competitors’ 10%+) and FDA approval, which included the use of earlier-line. Strategic investment in manufacturing scalability reduced production time to 14 days, which shut down the gap with a 7-day turnaround. While facing competition from Gilead’s Yescarta ($2.4B revenue) and Legend Biotech’s Carvykti (70% growth in myeloma), BMS’s pipeline advancements—like next-gen CAR-Ts for solid tumours—and 85% therapy access coverage in U.S. hospitals position it for sustained growth, targeting USD 2.2B+ in 2025 CAR-T sales.
Autologous Cell Therapy Market Key Trends:
• Expanding Clinical Applications: Autologous therapies are gaining approvals for new cancer types (e.g., solid tumors) and autoimmune diseases, broadening revenue potential beyond hematologic malignancies.
• Improved Manufacturing Efficiency: Innovations like AI-driven automation and decentralized production are reducing costs (30%+) and turnaround times (under 7 days), enhancing patient access.
• Strong Reimbursement & Commercial Adoption: Medicare and private insurers now cover CAR-T broadly, while hospitals invest in certified treatment centers, driving 25% annual market growth.
Autologous Cell Therapy Market Key Developments:
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 09 December 2025 | Abeona Therapeutics | The FDA approved ZEVASKYN (prademagene zamikeracel), a first-of-its-kind, autologous gene-corrected cell therapy for RDEB. | This provides a definitive regenerative treatment for chronic wounds in patients with recessive dystrophic epidermolysis bullosa. |
| 04 December 2025 | Bristol Myers Squibb | The FDA granted approval to Breyanzi (lisocabtagene maraleucel) for the treatment of relapsed or refractory marginal zone lymphoma (MZL). | The expansion broadens the therapeutic reach of this CAR-T cell therapy to patients who have failed at least two prior lines of treatment. |
| 08 December 2025 | Orchard Therapeutics | The FDA approved Waskyra (etuvetidigene autotemcel), an autologous hematopoietic stem cell-based gene therapy for Wiskott-Aldrich syndrome. | This is the first cell-based gene therapy for this condition, offering a curative option where HLA-matched donors are unavailable. |
| 06 February 2026 | Bambino Gesù Children's Hospital | Clinical research validated the use of autologous CD19 CAR-T cells to induce drug-free remission in pediatric patients with severe, treatment-resistant autoimmune disorders. | This breakthrough suggests a transformative shift in using CAR-T technology to reset the immune system beyond oncology. |
Autologous Cell Therapy Market Ecosystem
Autologous Cell Therapy Market Scope: Inquiry Before Buying
| Global Autologous Cell Therapy Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 7.11 USD Billion |
| Forecast Period 2026-2032 CAGR: | 21.54% | Market Size in 2032: | 27.86 USD Billion |
| Segments Covered: | by Product | Mesenchymal Stem Cells Bone Marrow Chondrocytes Haematopoietic Stem Cells Others |
|
| by Type | Autologous Stem Cell Therapies Car T Cell Therapies Autologous Non-Stem Cell Therapies Tumour Infiltrating Lymphocyte Therapies |
||
| by Application | Cancer Orthopaedics Cardiovascular/Autoimmune Disorders Neurodegenerative Disorders Wound Healing Others |
||
| by End-User | Hospitals & Clinics Academics & Research Ambulatory Centres |
||
Autologous Cell Therapy by region
North America (United States, Canada and Mexico)
Europe (United Kingdom, France, Germany, Italy, Spain, Sweden, Russia, Rest of Europe)
Asia Pacific (China, Japan, South Korea, India, Australia, Malaysia, Thailand, Vietnam, Indonesia, Philippines, Rest of APAC)
Middle East and Africa (South Africa, GCC, Nigeria, Egypt, Turkey, Rest of MEA)
South America (Brazil, Argentina, Colombia, Chile, Peru, Rest of South America)
Autologous Cell Therapy Key Players are
1. Bristol Myers Squibb Company
2. Gilead Sciences Inc. (Kite Pharma)
3. Bluebird Bio Inc.
4. Novartis AG
5. Johnson & Johnson (Janssen Biotech Inc.)
6. Vertex Pharmaceuticals Inc.
7. Caladrius Biosciences Inc.
8. Vericel Corporation
9. Castle Creek Biosciences Inc.
10. Lineage Cell Therapeutics Inc.
11. Catalent Inc.
12. Corning Incorporate
13. Autolus Therapeutics plc
14. Bayer AG
15. Sartorius AG
16. Lonza Group AG
17. Holostem Terapie Avanzate S.r.l.
18. Bio Elpida
19. Pharmicell Co. Ltd.
20. Tego Science Inc.
21. Daiichi Sankyo Company Limited
22. Takeda Pharmaceutical Company Limited
23. Sumitomo Pharma Co. Ltd.
24. Regeneus Ltd.
25. JW Therapeutics
26. Celyad Oncology SA
27. Mesoblast Limited
28. Fate Therapeutics Inc.
29. Adaptimmune Therapeutics plc
30. Orchard Therapeutics plc
Others
