Australia Growth Hormone Deficiency Clinical Trials Market Size by Product, Application, Distribution Channel - Revenue Pool Analysis, Margin Structure Assessment, Capital Flow Trends, Competitive Benchmarking & Forecast to 2030
Overview
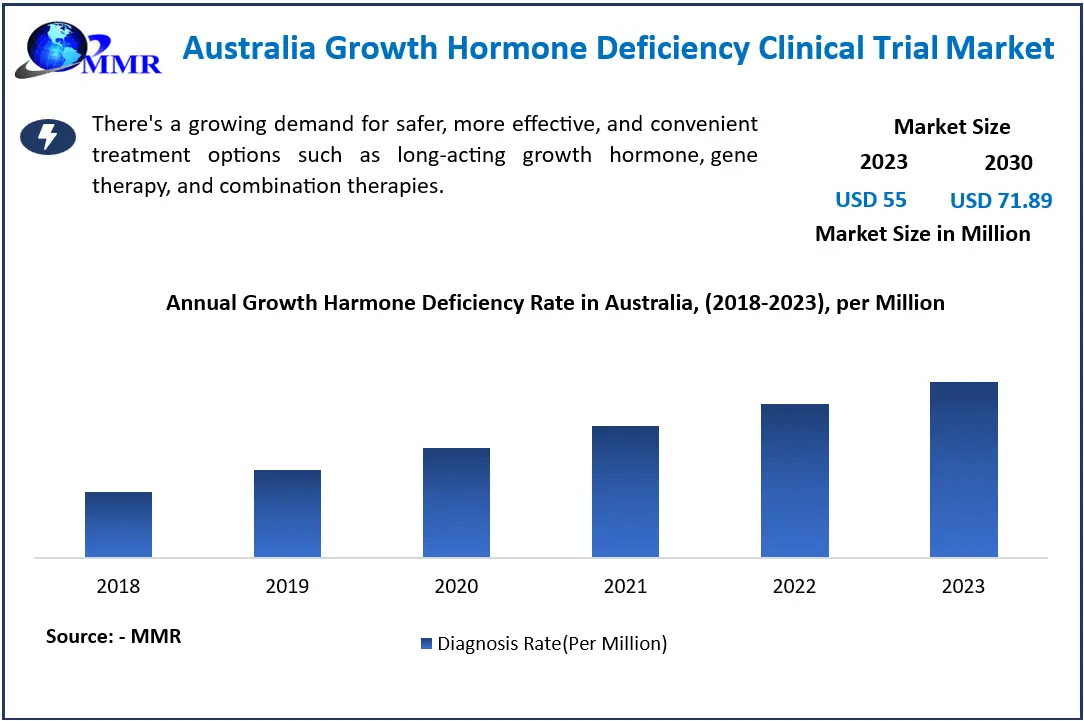
The Australian Growth Hormone Deficiency Clinical Trial Market size was valued at USD 55 million in 2023. The total Australia Growth Hormone Deficiency Clinical Trial Market revenue is expected to grow at a CAGR of 3.9% from 2023 to 2030, reaching nearly USD 71.89 Million.
Australia Growth Hormone Deficiency Clinical Trial Market Analysis
A Growth Hormone Deficiency (GHD) Clinical Trial refers to a systematic and scientific investigation conducted to evaluate the safety, efficacy, and potential benefits of therapeutic interventions for individuals with Growth Hormone Deficiency. Investing in the initial stages of businesses developing breakthrough Growth Hormone Deficiency (GHD) therapeutics, such as gene therapy or long-acting GH formulations, has a high growth potential. These revolutionary techniques have the potential to transform GHD approaches to treatment and also provide investors with attractive profits. Supporting specialist Contract Research Organizations (CROs) with experience in GHD trials and efficient patient recruitment tactics is another interesting investment opportunity. Their expertise accelerates trial processes, increasing overall trial success. Also, the growing market for digital health platforms that enable patient participation and remote data gathering represents a fast-developing opportunity, reflecting the industry's shift toward advanced technology and comprehensive healthcare solutions.
| 1. The MMR Studies have shown that EDC systems can reduce data entry errors by up to 70% and improve data quality by 20-30%. |
| 1. Research suggests that remote monitoring is expected to reduce the number of clinic visits by up to 50%, leading to significant cost savings |
| 1. AI-powered platforms have been shown to improve the accuracy of predicting patient response to treatment by up to 15%, paving the way for personalized medicine approaches |
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
Advancement in GHD Therapies
Continuous research and development in GHD medicines is expected to result in the discovery of innovative therapy modalities, such as gene therapies and unique drug delivery techniques, to improve patient outcomes. Gene therapy represents an innovative advancement in GHD treatment by potentially replacing lifelong daily injections with a single, definitive therapy. The initiative not only promises to lower healthcare expenses but also to improve patient quality of life. The market is expected to grow, attracting new patient populations with the promise of long-term efficiency and convenience. Still, ethical considerations, such as a comprehensive examination of safety data, need to be prioritized before widespread implementation. Novel dosage strategies that provide convenience and reduced frequency are expected to increase patient compliance, improving treatment outcomes. Targeted delivery systems have the potential to increase Growth Hormone (GH) consumption, hence improving therapy efficacy. The development additionally improves the patient experience and diversifies the GHD treatment landscape by responding to preferences and meeting unexpected market needs.
Lack of public perception and awareness
Global clinical trial participation is 4%, and Germany's GHD trials are estimated to have comparable low rates due to information gaps and concerns. It reduces patient diversity and jeopardizes outcome generalizability. Insufficient recruiting has a detrimental impact on research progress, causing delays and stifling advancements in Growth Hormone Deficiency treatment alternatives, limiting potential discoveries in the field. Limited knowledge about GHD, its symptoms, and treatment choices discourages participation in related research. Potential volunteers are hesitant to participate because they have concerns about unknown side effects, invasive procedures, and long-term consequences. Misinformation and distrust, exacerbated by bad media depictions and previous unethical research procedures, add to public doubts, reducing willingness to engage in clinical studies.
Australia Growth Hormone Deficiency Clinical Trial Market Segments Analysis
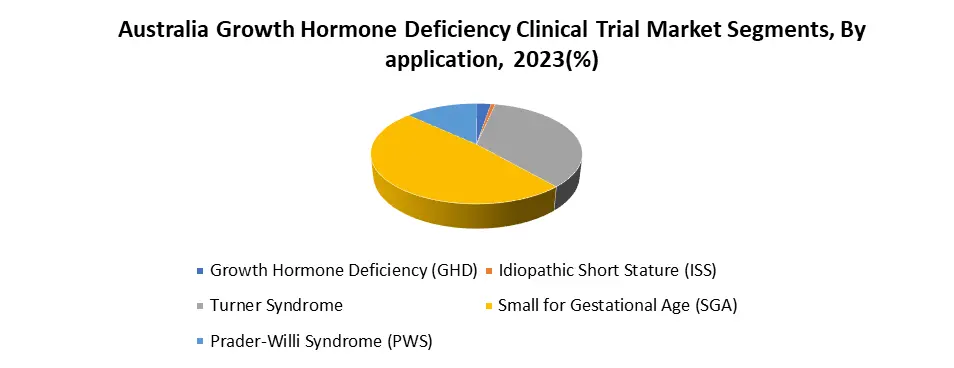
By Application, the growth Hormone Deficiency segment accounts for an estimated 60% of the overall Australian growth Hormone Deficiency Clinical Trial Market. The high frequency of Growth Hormone Deficiency (GHD) in both children and adults creates a large pool of potential clinical trial patients, driving more funding and research activity. The changing climate supports an active market ecosystem, with numerous ongoing studies investigating various therapeutic options. The huge demand for effective GHD medicines drives ongoing innovation and development, encouraging pharmaceutical companies and researchers to spend resources. The increasing focus on the GHD segment additionally drives overall market growth however, it also encourages patient-centricity, reflecting the demands of a vast and diverse patient population. The focus on patient-centricity leads to more relevant and meaningful research outcomes, which benefit the entire GHD community.
Australia Growth Hormone Deficiency Clinical Trials Market Scope: Inquire Before Buying
| Australia Growth Hormone Deficiency Clinical Trials Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2023 | Forecast Period: | 2024-2030 |
| Historical Data: | 2018 to 2023 | Market Size in 2023: | US $ 55 Mn. |
| Forecast Period 2024 to 2030 CAGR: | 3.9% | Market Size in 2030: | US $ 71.89 Mn. |
| Segments Covered: | by Product | Powder Solvent |
|
| by Application | Growth Hormone Deficiency Idiopathic Short Stature Turner Syndrome Small For Gestational Age Prader-Willi Syndrome |
||
| by Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies |
||
Key Players of the Growth Hormone Deficiency Clinical Trial in Australia
1. Merck KGaA
2. Eli Lily and Company
3. Novartis AG
4. Novo Nordisk A/S
5. Pfizer Inc.
6. Ipsen S.A.
7. LG Chem Ltd.
8. Anhui Anke Biotechnology (Group) Co. Ltd
9. Ferring B.V.
10. Genetech Inc. (Roche Group)
11. QuintilesIMS
FAQs:
1. What are the different types of GHD clinical trials conducted in Australia?
Ans. Phase I, Phase II, Phase III, and Phase IV trials are conducted for developing and testing new GHD therapies, including
2. What are the regulatory guidelines for conducting GHD clinical trials in Australia?
Ans. The Therapeutic Goods Administration (TGA) oversees clinical trials and requires adherence to strict ethical and safety standards.
3. What is the projected market size & and growth rate of the Australia Growth Hormone Deficiency Clinical Trial Market?
Ans. The Australian Growth Hormone Deficiency Clinical Trial Market size was valued at USD Million in 2023. The total Australian Growth Hormone Deficiency Clinical Trial market revenue is expected to grow at a CAGR of % from 2023 to 2030, reaching nearly USD Million By 2030.