Global AI in Clinical Trials Market - Industry Structure Evaluation, Demand Drivers Analysis, Regional Growth Analysis and Identification, Competitive Positioning / Landscape Review & Global Market Size Forecast to 2032
Overview
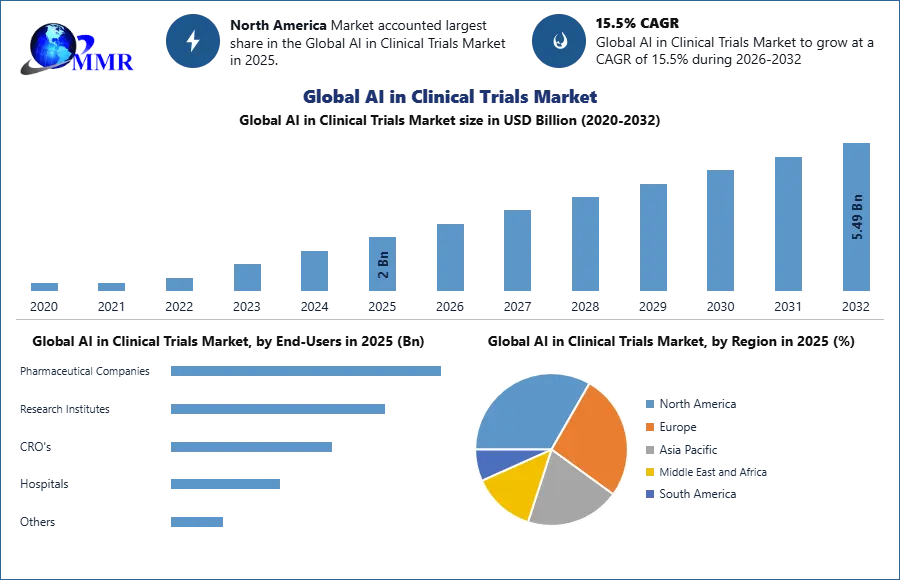
The Global AI in Clinical Trials Market size was valued at USD 2 Bn in 2025 and is expected to reach USD 5.49 Bn by 2032, at a CAGR of 15.5 %.
Overview of the AI in Clinical Trials Market
AI in Clinical Trials Market has the potential to enhance patient recruitment processes through the identification and screening of prospective participants in accordance with predefined inclusion and exclusion criteria. This capability holds the promise of diminishing both the temporal and financial resources traditionally expended in the patient recruitment phase, which has been a recurring challenge in clinical research. Wearable devices and various sensor technologies have the capacity to continuously gather real-time physiological data from patients.
AI is being used more and more in preclinical and clinical trials, with advanced algorithms able to rapidly analyze vast databases of chemical compounds and identify the ones that are most likely to bind to a target, enabling drug developers to explore the chemical world far more quickly. The graphical representation and structural exclusive information showed the dominating region of the AI in Clinical Trials Market. The detailed and constructive formation of key drivers, opportunities, and unique segmentation outputs structural and optimistic data. Validated using primary as well as secondary research methodology and scope of the AI in Clinical Trials Market.
To know about the Research Methodology :- Request Free Sample Report
AI in Clinical Trials Market Dynamics
Enhanced Precision in Data Analysis and Accelerated Time-to-Market are the major drivers in the AI in Clinical Trials Market
AI in Clinical Trials Market offers a substantial advantage by automating the time-consuming and resource-intensive tasks inherent in clinical trials. These tasks encompass data aggregation, analysis, cohort selection, and patient recruitment. For instance, AI algorithms excel in the meticulous examination of extensive patient data to pinpoint ideal candidates for clinical trials. As well as these algorithms can discern patient behavior patterns and offer trial design recommendations. AI-driven tools like Starmind expedite the journey to AI in Clinical Trials Market for research and development (R&D) initiatives by enhancing knowledge accessibility and fostering more efficient collaboration within organizations. When R&D teams seamlessly share and access crucial information across the enterprise, such as recruitment data and trial results, it leads to enhanced communication and collaboration, while redundant efforts are minimized.
AI algorithms empower researchers to expeditiously analyze extensive datasets, uncovering patterns and trends that may elude traditional clinical researchers or take significantly more time to discern. For instance, AI-based models predict the toxicity of potential drug candidates, thus eliminating unsuitable compounds from further development. This not only enhances safety but also conserves valuable time and resources that would otherwise be invested in unpromising endeavors. AI plays a pivotal role in identifying specific patient populations most likely to benefit from particular drugs, taking into account genetic makeup and lifestyle in the AI in Clinical Trials Industry. AI also contributes to personalized treatment by optimizing dosages and determining the appropriate quantity and frequency based on individual patient characteristics, including personal and family medical history.
The proliferation of AI Integration and Focus on Diagnostics and Treatment are the prominent trends in the AI in Clinical Trials Market
Over the past six years, there has been a notable surge in the integration of AI tools and technologies within clinical trials. Approximately 600 completed or ongoing clinical trials have harnessed AI's potential to assess drugs and therapies across various therapeutic areas. This trend highlights the significant dedication and efforts of researchers to leverage AI's capabilities in the healthcare domain. The majority of these clinical studies have been strategically designed to cater to diagnostics and treatment. AI is increasingly being employed to enhance the precision and efficacy of diagnostic procedures, as well as to optimize treatment strategies. The utilization of AI in Clinical Trials is not limited to specific therapeutic areas. Researchers are exploring AI's applicability across a wide range of medical conditions and diseases. This trend showcases the versatility of AI in Clinical Trials Industry, with its potential to contribute to advancements in various healthcare domains.
AI in Clinical Trials Market Segment Analysis
Clinical Trial Phases:
AI exerts a profound influence across Phase I, Phase II, and Phase III. During Phase I, which emphasizes safety and dosage evaluations, AI plays a pivotal role in optimizing dose-finding studies, identifying potential adverse events, and assisting in the selection of the most promising drug candidates. As clinical trials progress to Phase II, AI technologies prove instrumental in assessing treatment efficacy by harnessing data analysis to identify patient subgroups that respond favorably to specific therapies, thus expediting the drug development process in the AI in Clinical Trials Market. In Phase III trials, which demand extensive data analysis and validation of treatment effectiveness, AI-driven efficiencies come to the forefront, streamlining patient recruitment, optimizing trial designs, and facilitating real-time monitoring of safety and efficacy endpoints.
In metabolic disorders like diabetes and obesity, AI optimizes insulin dosing, monitors glucose levels, and tailors personalized dietary and exercise regimens based on individual patient data. Furthermore, AI significantly impacts oncological disorders, encompassing tumor detection, classification, and prediction of treatment responses, thereby enabling more precise and effective cancer therapies. Beyond these areas, AI demonstrates its versatility across diverse therapeutic domains, aiding in data analysis, patient stratification, and treatment optimization for rare diseases, autoimmune conditions, and respiratory ailments to boosts the AI in Clinical Trials Market Growth.
End-Users:
The primary end-users within the AI in Clinical Trials market predominantly comprise pharmaceutical and biotechnology companies, positioning themselves at the forefront of AI integration. AI technologies bolster drug discovery, streamline clinical trial operations, and enhance post-market surveillance, elevating their capabilities in research and development. Additionally, other key end-users include academic research institutions, government agencies, and contract research organizations (CROs), all harnessing AI's data analytics proficiency, patient recruitment efficiency, and overall enhancement of clinical trial processes to drive advancements in healthcare research.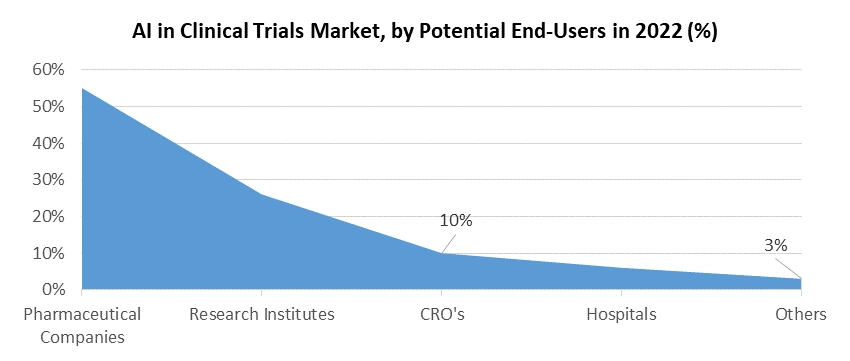
AI in Clinical Trials Market Regional Analysis
North America, led by the United States, stands as a prominent hub for AI integration in clinical trials. This region benefits from a robust healthcare infrastructure, a well-established pharmaceutical industry, and significant investments in AI research. The FDA's supportive stance on AI's incorporation in clinical trials fosters innovation and adoption. Collaboration between pharmaceutical giants and tech companies further harnesses AI's potential for expediting drug development. European nations, including the UK, Germany, and France, are experiencing rapid AI adoption in the context of clinical trials. Renowned pharmaceutical companies, robust regulatory bodies, and government initiatives to promote AI in healthcare collectively drive market growth.
Cross-border collaborations and research partnerships contribute to the stimulation of AI innovations in clinical research. The Asia-Pacific region is emerging as a significant player in AI in Clinical Trials market. Countries like China and India, boasting substantial patient populations and expanding pharmaceutical sectors, are exploring AI to enhance the efficiency and affordability of clinical trials. Japan, with its advanced technology ecosystem and aging population, presents opportunities for AI-driven healthcare solutions to boost the AI in Clinical Trials Market growth.
Latin America, led by Brazil and Mexico, is gradually embracing AI in the context of clinical trials. The pharmaceutical industry in this region is expanding, and regulatory frameworks are evolving to accommodate AI applications. Collaborations between local research institutions and global pharmaceutical companies are driving the development of the AI in Clinical Trials market. The Middle East and Africa are witnessing promising but nascent growth in AI adoption for clinical trials. Countries like South Africa and the United Arab Emirates are investing in healthcare infrastructure and technology. Collaborations with international organizations and pharmaceutical companies are instrumental in propelling AI adoption in the region.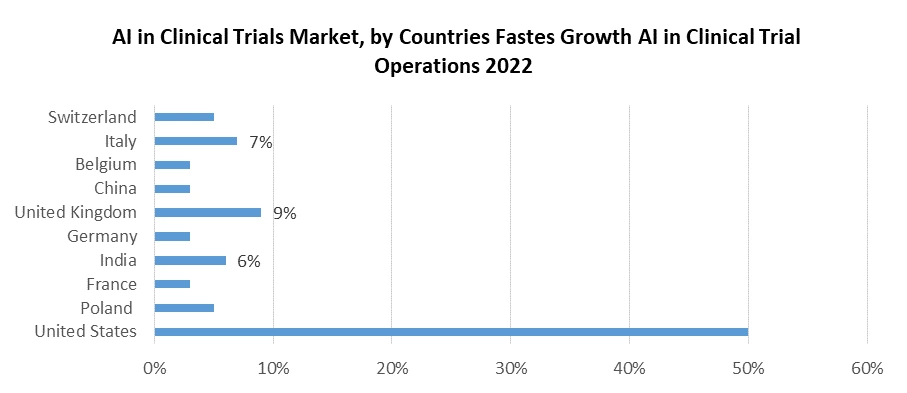
AI in Clinical Trials Market Competitive Landscape
In August 2023, Texas Tech University Health Sciences Center embarked on a noteworthy collaboration with Deep 6 AI, aimed at the establishment of an innovative AI program tailored for clinical trials. This partnership underscores the increasing recognition of AI's potential to revolutionize clinical trial processes. By leveraging Deep 6 AI's expertise, Texas Tech University Health Sciences Center aims to enhance patient recruitment, streamline data analysis, and ultimately accelerate the pace of clinical research. This development signifies a commitment to harnessing AI for more efficient and effective clinical trials in the AI in Clinical Trials Market.
In July 2023, Insilico Medicine made a groundbreaking announcement with the commencement of a phase II clinical trial for INSO18_055, a drug designed entirely through AI. This represents a significant milestone in drug discovery and development, as it marks one of the first instances where a fully AI-designed drug advances to clinical trial stages. INSO18_055 is being evaluated for its efficacy in treating idiopathic pulmonary fibrosis, a challenging and often life-threatening condition. This development reflects the growing confidence in AI's ability to expedite drug discovery and potentially lead to novel therapies for complex diseases in the AI in Clinical Trials Market.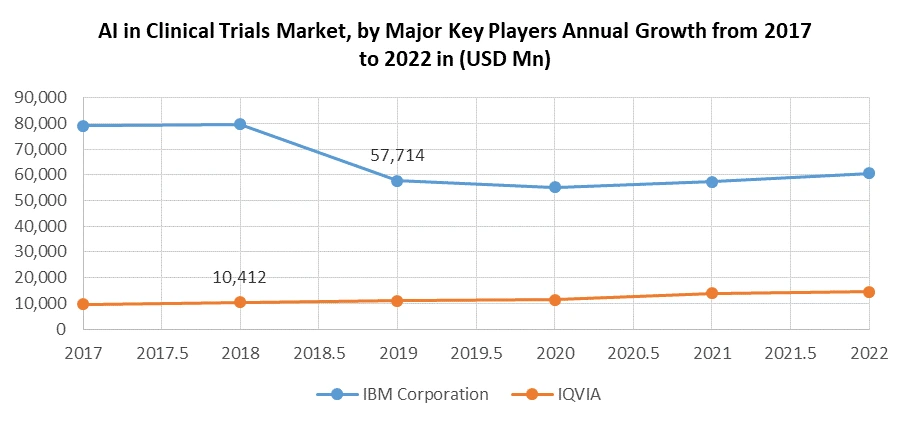 In June 2023, Anavex Life Sciences forged a strategic partnership with Partex Group, aimed at harnessing Partex's proprietary AI technology to advance the development of Anavex's drug pipeline. This collaboration underscores the synergy between pharmaceutical companies and AI-focused entities, as they seek to leverage AI's capabilities to optimize drug development processes. By incorporating AI into its drug pipeline, Anavex Life Sciences aims to enhance the efficiency of preclinical and clinical research, from target identification to patient recruitment and safety monitoring. This partnership highlights the strategic use of AI as a valuable tool in the pursuit of innovative drug therapies in the AI in Clinical Trials Industry.
In June 2023, Anavex Life Sciences forged a strategic partnership with Partex Group, aimed at harnessing Partex's proprietary AI technology to advance the development of Anavex's drug pipeline. This collaboration underscores the synergy between pharmaceutical companies and AI-focused entities, as they seek to leverage AI's capabilities to optimize drug development processes. By incorporating AI into its drug pipeline, Anavex Life Sciences aims to enhance the efficiency of preclinical and clinical research, from target identification to patient recruitment and safety monitoring. This partnership highlights the strategic use of AI as a valuable tool in the pursuit of innovative drug therapies in the AI in Clinical Trials Industry.
Global AI in Clinical Trials Market Scope: Inquire before buying
| Global AI in Clinical Trials Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 2 USD Billion |
| Forecast Period 2026-2032 CAGR: | 15.5% | Market Size in 2032: | 5.49 USD Billion |
| Segments Covered: | by Trial Phase | Phase I Phase II Phase III |
|
| by Technology | Machine Learning Natural Language processing Deep Learning Unspecified |
||
| by Application | Patient Recruitment Data Analysis Safety Monitoring Drug Discovery Personalized Medicine |
||
| by End-Users | Pharmaceutical Companies Research Institutes CRO's Hospitals Others |
||
AI in Clinical Trials Market, by Region
North America (United States, Canada and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan and Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A)
South America (Brazil, Argentina Rest of South America)
Key Players of the AI in Clinical Trials Market
1. Euretos
2. Biosymetric
3. Unlearn.AI,Inc.
4. Exscientia
5. IMB Corporation
6. AiCure
7. Antidote Technologies
8. Deep 6 AI
9. Innoplexus
10. IQVIA
11. Median Technologies
12. Medidata
13. Mendel.ai
14. Phesi
15. Saama Technologies
16. Signant Health
17. Trials.ai
Frequently Asked Questions and Answers about AI in Clinical Trials Market
1. How AI can improve clinical trials?
Ans: AI can improve clinical trials by expediting patient recruitment, enhancing data analysis accuracy, personalizing treatment plans, and optimizing trial designs.
2. What are the challenges associated with the integration of AI in clinical trials?
Ans: Challenges associated with integrating AI in clinical trials include data privacy concerns, regulatory compliance, data quality, and the need for AI expertise.
3. What is the role of AI in electronic health records of clinical trials data?
Ans: AI plays a role in electronic health records of clinical trials data by facilitating data management, analysis, and insights for more efficient and informed decision-making.
4. What are the upcoming trends in AI in clinical trial market?
Ans: Upcoming trends in the AI in clinical trial market include decentralized trials, AI-driven patient monitoring, real-world evidence utilization, and AI-enhanced drug discovery.
5. Which region captures the largest share in the AI in clinical trials market?
Ans: North America captures the largest share in the AI in clinical trials market, driven by its robust healthcare infrastructure and significant investments in AI research.
