Post Herpetic Neuralgia Treatment Market by Treatment Type, End Users, and Distribution Channel – Global Market Size Estimation, Industry‑Wide Analysis, Competitive Landscape Assessment & Long‑Term Forecast to 2030
Overview
Post Herpetic Neuralgia Treatment Market is expected to grow at a CAGR of 3.2% during the forecast period. Post Herpetic Neuralgia Treatment Market is expected to reach US$ 4.05 Bn. by 2030.
Post Herpetic Neuralgia Treatment Market Overview
Post Herpetic Neuralgia (PHN) is the most challenging complication of the Herpes zoster virus. The condition is characterized by constant or intermittent burning, itching or aching. Other characteristic symptoms include numbness and tingling. It affects the nerve fibres and the skin as well. Despite the introduction of the vaccine against the Varicella Zoster virus, PHN continues to be a clinical problem. The treatment of PHN is based on an array of drugs and drug associations with proven efficacy. The drugs include Tricyclic antidepressants, the antiepileptic drug- Gabapentin, Pregabalin, and opioids.
People with ongoing conditions such as cancer or HIV are more likely to get affected. The geriatric population which is mostly immunosuppressant is likely to increase the screening rates for the disease. Along with these factors, the advancements in treatment techniques of Post Herpetic Neuralgia is a key driver to the Post Herpetic Neuralgia Treatment market. The development of steroids, antidepressants, and anticonvulsants have gained power in the market for treating the condition. Increasing awareness among people, government initiatives, and advances in healthcare infrastructure around the globe aid the market growth. Poor efficacy of drugs and their high cost is likely to restrain the PHN market. Inadequate knowledge of drug usage and overdose may lead to high rates of mortality hampering the market growth.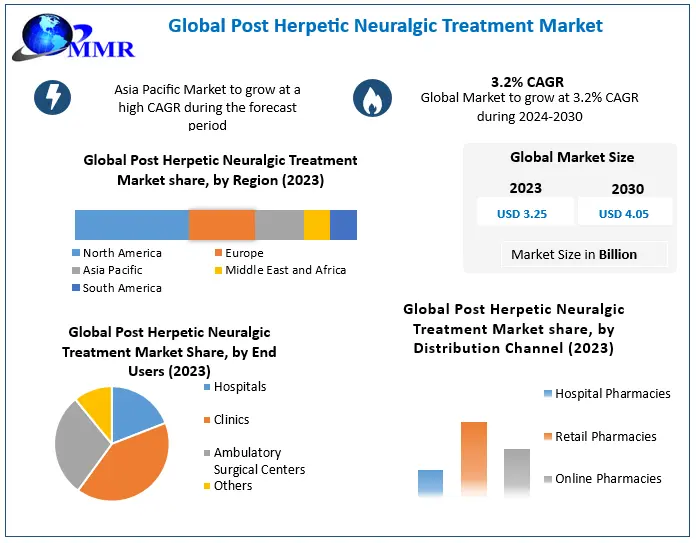 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
Post Herpetic Neuralgia Treatment Market Dynamics
Increasing Prevalence of Herpes Zoster Driving the Post Herpetic Neuralgia Treatment Market Growth
Herpes zoster, commonly known as shingles, is caused by the reactivation of the varicella-zoster virus (VZV), the same virus responsible for chickenpox. After a person has varicella, the virus remains dormant in the dorsal root ganglia and can reactivate later in life, causing herpes zoster. The characteristic rash of herpes zoster typically appears in one or two adjacent dermatomes, most commonly on the trunk or face. The rash is often accompanied by pain, itching, or tingling sensations.
The most common complication of herpes zoster is postherpetic neuralgia (PHN), which is characterized by persistent pain in the area where the rash occurred, lasting more than 90 days after rash onset. Thus, the increasing incidence of herpes zoster, increases the demand for treatment inturn driving the Post Herpetic Neuralgia Treatment Market growth. PHN can last for months or even years and is more common in older adults. Other complications of herpes zoster include herpes zoster ophthalmicus, disseminated zoster, bacterial superinfection of the lesions, and cranial and peripheral nerve palsies.
Recombinant zoster vaccine (RZV, Shingrix) is recommended to prevent shingles and related complications. The risk of developing herpes zoster and related complications increases with age and in people with compromised or suppressed immune systems. An estimated one million cases of herpes zoster occur annually in the United States, with approximately 10% to 18% of people developing PHN. Complications from herpes zoster result in hospitalization, particularly in older adults and those with compromised immune systems.
High Cost of Treatment Restraining the Post Herpetic Neuralgia Treatment Market Growth
The cost of treatment for PHN is substantial, especially for patients who require long-term therapy or interventional procedures. Medications such as anticonvulsants and antidepressants, which are commonly used to manage PHN are expensive, particularly for patients without adequate insurance coverage. The interventional procedures such as nerve blocks and spinal cord stimulation, while effective, can be cost-prohibitive for many patients. The high cost of treatment acts as a significant barrier to access, particularly for patients with limited financial resources, thereby restraining Post Herpetic Neuralgia Treatment Market growth.
Post-herpetic neuralgia (PHN) is a painful and often disabling condition that can occur following acute shingles caused by the herpes simplex virus. Treatment of PHN typically involves a range of drug therapies, and patients may require multiple visits to general practitioners and hospital outpatient departments. This study aimed to estimate the costs and consequences of treating PHN in the UK National Health Service (NHS) and to assess the impact of the condition on patients' lives, including pain and time off usual activities such as work. The study found that PHN can be a costly consequence of acute shingles, with lifetime treatment costs estimated at $821.09 for patients attending a tertiary referral centre. For a 1-year incidence cohort of shingles patients in the UK, the lifetime treatment costs of PHN ranged from $5.12 million to $19.09 million. Efforts to reduce the incidence or severity of PHN are needed to alleviate the economic burden on the healthcare system.
Post Herpetic Neuralgia Treatment Market Segment Analysis
Based on Treatment Type: The market is segmented into Drugs, Patches and Steroid Injectables. The Drugs segment held the largest Post Herpetic Neuralgia Treatment Market share in 2023 and is expected to grow rapidly during the forecast period. The development of new drugs, as a definitive curative treatment has yet to be discovered. Government support for products aimed at treating postherpetic neuralgia, particularly through initiatives and investments in research and development, is expected to drive market growth.
For example, Cipla's subsidiary, InvaGen Pharmaceuticals, has received final approval from the United States Food and Drug Administration (USFDA) for Pregabalin Capsules, a generic therapeutic equivalent version of Pfizer's Lyrica. The drug is indicated for managing neuropathic pain associated with certain illnesses, including diabetic peripheral neuropathy, postherpetic neuralgia, and fibromyalgia, among others. Available in strengths ranging from 25mg to 300mg, the product is set to tap into a significant market, with Lyrica having an estimated market size of $5.4 billion for the 12 months ending March 2019, according to IQVIA data. The approved product is available for shipping immediately.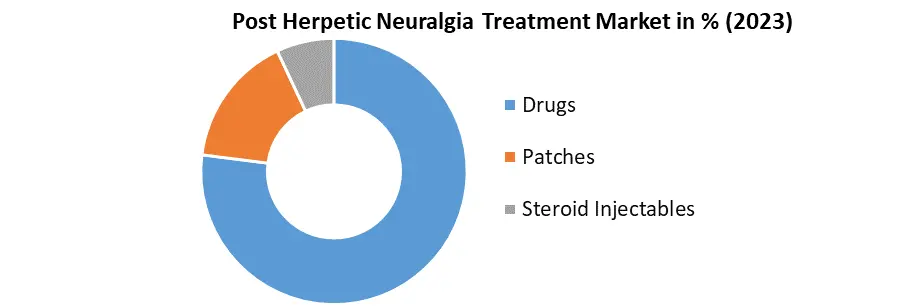
Post Herpetic Neuralgia Treatment Market Regional Insights
Asia Pacific Post Herpetic Neuralgia Treatment Market is expected to grow at a high rate during the forecast period. Upcoming changes to the Australian government initiative i.e. National Immunisation Program (NIP) will take effect from 1 July 2023. The hexavalent vaccine Vaxelis® will be added to the NIP, offering protection against diphtheria, tetanus, pertussis, hepatitis B, poliomyelitis, and invasive infections caused by Haemophilus influenzae type b (DTPa-hepB-IPV-Hib). Vaxelis® will be administered to children at 2, 4, and 6 months of age, and it will also be funded for catch-up vaccination in children under 10 years who have not previously received DTPa-hepB-IPV-Hib vaccination.
Additionally, from 1 November 2023, Shingrix® vaccine will replace Zostavax® vaccine on the NIP for the prevention of herpes zoster (shingles) and post-herpetic neuralgia, pending final regulatory approvals. Shingrix® will be provided as a two-dose primary course for eligible cohorts, including all adults aged 70 years, First Nations Australian adults aged 50 years and older, and immunocompromised individuals aged 18 years and older with conditions at 'high risk' of herpes zoster infection. These changes follow recommendations from the Pharmaceutical Benefits Advisory Committee (PBAC), the Australian Technical Advisory Group on Immunisation, and other clinical experts.
As per the study, Latin America and Caribbean (LAC) is expected to grow in the future. The economic burden of herpes zoster (HZ) in the Latin America and Caribbean (LAC) region has been studied by analyzing healthcare resource use and cost outcomes. The primary direct medical resources reported in the studies included doctor visits, transportation, hospital days, nursing, medication schedules, and physical therapy. Direct total costs per patient ranged from $99.99 to $4177.91, with Brazil reporting the highest costs. Direct costs were on average 81.39% higher than indirect costs. The economic burden of HZ and its complications in the LAC region is considerable, particularly among high-risk populations and older age groups. Preventative strategies such as vaccination could help alleviate this burden.
Post Herpetic Neuralgia Treatment Market Scope: Inquiry Before Buying
| Post Herpetic Neuralgia Treatment Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2023 | Forecast Period: | 2024-2030 |
| Historical Data: | 2018 to 2023 | Market Size in 2023: | US $ 3.25 Bn. |
| Forecast Period 2024 to 2030 CAGR: | 3.2% | Market Size in 2030: | US $ 4.05 Bn. |
| Segments Covered: | by Treatment Type | Drugs Anticonvulsants Antidepressants Opioids Others Patches Lidocaine Skin Patches Capsaicin Skin Patches Steroid Injectables |
|
| by End Users | Hospitals Clinics Ambulatory Surgical Centers Others |
||
| by Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies |
||
Post Herpetic Neuralgia Treatment Market by Region:
North America (United States, Canada, and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria, and the Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan, and the Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria, and the Rest of ME&A)
South America (Brazil, Argentina Rest of South America)
Post Herpetic Neuralgia Treatment Key Players
1. Pfizer Inc
2. Teikoku Pharma USA, Inc
3. Teva Pharmaceutical Industries Ltd.
4. Endo Pharmaceuticals Inc
5. Daiichi Sankyo
6. Acorda Therapeutics
7. Novartis AG
8. Merck & Co., Inc
9. Sun Pharmaceutical Industries Ltd.
10. Astellas Pharma Inc.
11. Mallinckrodt Pharmaceuticals
12. Others
Frequently Asked Questions:
1. Which region held the largest share in the Global Post Herpetic Neuralgia Treatment Market in 2023?
Ans: North American region held the highest share of the global market in 2023.
2. What is the growth rate of the Global Post Herpetic Neuralgia Treatment Market?
Ans: The Global Post Herpetic Neuralgia Treatment Market is growing at a CAGR of 3.2% during the forecast period 2024-2030.
3. What is the scope of the Global Post Herpetic Neuralgia Treatment Market report?
Ans: The Global Post Herpetic Neuralgia Treatment Market report helps with the PESTLE, PORTER, COVID-19 Impact analysis, Recommendations for Investors & Leaders, and market estimation of the forecast period.
4. Who are the key players in the Global Post Herpetic Neuralgia Treatment Market?
Ans: The important key players in the Global Post Herpetic Neuralgia Treatment Market are – Pfizer Inc, Teikoku Pharma USA, Inc, Teva Pharmaceutical Industries Ltd., Endo Pharmaceuticals Inc, Daiichi Sankyo, Acorda Therapeutics, Novartis AG, Merck & Co., Inc., Sun Pharmaceutical Industries Ltd., Astellas Pharma Inc. and Mallinckrodt Pharmaceuticals.
5. What is the study period of this Market?
Ans: The Global Post Herpetic Neuralgia Treatment Market is studied from 2018 to 2030.
