Pharmaceutical Cleaning Validation Market Size by Product Small Molecule Drug Peptides Proteins Cleaning Detergent, Validation Test Non-specific Tests Total Carbon Analysis (TC) Total Organic Carbon Testing (TOC) Non-purgeable Organic Carbon (NPOC) Conductivity Others Product-specific Analytical Tests Ultraviolet-Visible Spectroscopy (UV/VIS) High-Performance Liquid Chromatography (HPLC) Liquid Chromatography/ Mass Spectrometry (LC/MS) Others, Region – Revenue Pool Analysis, Margin Structure Assessment, Capital Flow Trends & Competitive Benchmarking
Overview
Global Pharmaceutical Cleaning Validation Market size was valued at US$ 17.20 Bn. in 2022 and the total revenue is expected to grow at 5.7% through 2023 to 2029, reaching nearly US$ 25.36 Bn.
Pharmaceutical Cleaning Validation Market Overview:
Cleaning Validation is a mechanism for ensuring that a cleaning procedure eliminates residues of active pharmaceutical components from a product made in a piece of equipment, as well as
cleaning aids and microbiological characteristics. Pharmaceutical cleaning validation is an important factor responsible to maintain the standard of medicine or pharmaceutical ingredients during the process of manufacturing.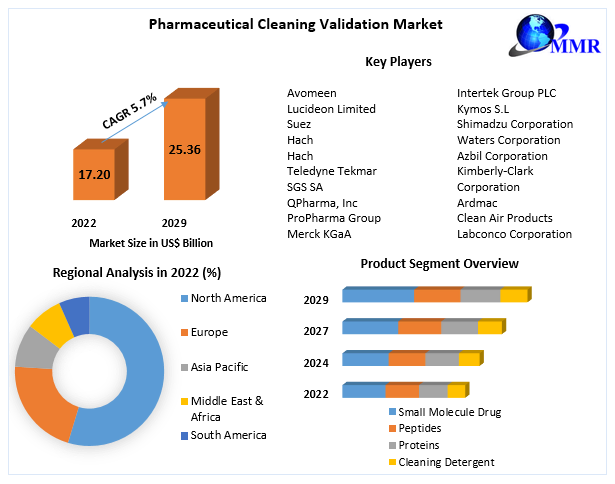 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
Market Dynamics:
The massive development of pharmaceutical manufacturing facilities, the rising prevalence of diseases, the growing global senile population increased healthcare spending, and increased market investments to meet regulatory standards set by international governing bodies such as the US FDA and the European Medicines Agency (EMA). The pharmaceutical manufacturing industry's developing regulations to limit cross-contamination have had a favorable effect on the market's growth. Furthermore, regulatory authorities are taking a more moderate approach towards rules, allowing the pharmaceutical sector to speed up their production and quality control procedures.
The market growth for pharmaceuticals is driving up the requirement for cleaning validation to assure product safety, monitor possible contamination, and detect contaminants. In September 2021, the International Society of Pharmaceutical Engineering hosted the ISPE APAC Pharmaceutical Manufacturing Conference to raise awareness about the relevance of the cleaning validation process. Supports the pharmaceutical industry's manufacturing and quality control procedures to be faster.
The rules and regulatory requirements for cleaning validation in medication production are mostly established by the US Food and Drug Administration (US FDA) and the European Medicines Agency (EMA). The European Medicines Agency (EMA) is a forerunner in developing risk-based cleaning validation criteria to prevent cross-contamination in manufacturing facilities. Following the EMA's lead, the Pharmaceutical Inspection Co-operation Scheme (PIC/S) created its own set of rules in shared production facilities, including Health-Based Exposure Limits (HBEL).
These reasons have boosted demand for cleaning validation among pharmaceutical producers, resulting in healthy market growth. In comparison to other sectors, the pharmaceutical business is more life-saving and life-endangering, and non-compliance can result in dangerous effects for human health, thus regulatory standards are more severe for the companies. Hence, these factors are estimated to fuel the market growth in the forecast timeframe.
Pharmaceutical Cleaning Validation Market Segment Analysis:
The small molecule segment is dominating the Product Type segment of the Pharmaceutical Cleaning Validation Market:
The small Molecule segment is estimated to gain more attraction in the market and accounted for the largest market share of 45.7% in the year 2022. In 2018, the FDA authorized 59 new medicines, with 71% of them being small molecules and 29% being biologics. Small molecule medicines also accounted for 75% of the pharmaceutical industry in 2022. As a result, the growing need for cleaning validation is being fuelled by the increased manufacturing levels of small molecule medicines. Small-molecule medicines are also important in the treatment of chronic illnesses and regenerative medicine. Hence, growing chronic diseases and their treatment options are estimated to drive the growth of the market.
The product-specific analytical tests are considered to supplement the growth of the Pharmaceutical Cleaning Validation Market.
In 2022, the product-specific analytical tests segment led the pharmaceutical cleaning validation market, accounting for 65.3 % of total revenue. Cleaning validation frequently employs analytical tests such as ultraviolet spectroscopy, high-performance liquid chromatography, liquid chromatography/mass spectrometry, near-infrared spectroscopy, mid-infrared spectroscopy, capillary zone electrophoresis, atomic absorption, ion mobility spectrometry, and enzyme-linked immunosorbent assay.
The most widely used technique of cleaning validation is High-performance Liquid Chromatography (HPLC), which accounted for 50.9 % of the revenue share in the product-specific analytical testing sector in 2022. This is due to the benefits of employing HPLC, which includes the ability to utilize a variety of swabs and different detection techniques like UV, fluorescence, photodiode array, are some of them. Furthermore, it provides quantitative findings, peak separation, and the ability to leverage several solvents for extraction solutions.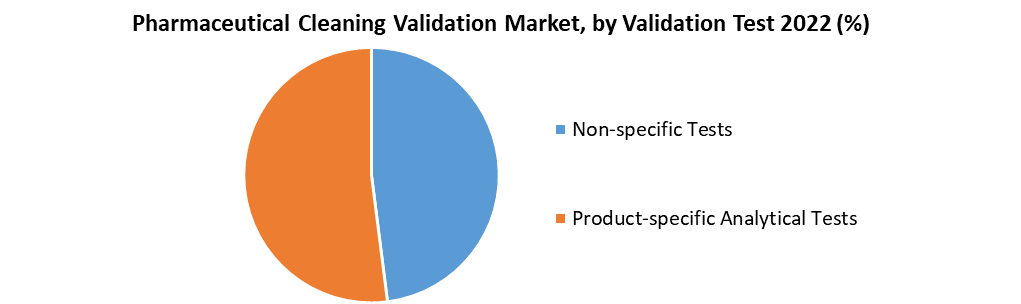
Regional Insights:
North America is dominating the growth of the pharmaceutical cleaning validation market. Owing to the uptick adoption of cleaning validation by pharmaceutical, manufacturing firms to comply with strict regulatory requirements laid down by the U.S. FDA and Health Canada, North America led the market and accounted for the biggest revenue share of 40.2 % in 2022. The geographical revenue share is influenced by the presence of local and foreign pharmaceutical businesses, rising healthcare expenditure on pharmaceutical medications, rising illness burden, and rising awareness levels.
Asia Pacific is estimated to gain more attractions owing to the high demand for pharmaceutical cleaning validation processes and is growing at a 9.3% CAGR. The existence of significant pharmaceutical production units, as well as increased investments by multinational pharmaceutical firms, are key factors driving regional revenue growth. Further, investment in the healthcare industry and rapid innovations are estimated to dominate the market growth.
The objective of the report is to present a comprehensive analysis of the Pharmaceutical Cleaning Validation Market to the stakeholders in the industry. The past and current status of the industry with the forecasted market size and trends are presented in the report with the analysis of complicated data in simple language. The report covers all the aspects of the industry with a dedicated study of key players that include market leaders, followers, and new entrants.
PORTER, SVOR, PESTEL analysis with the potential impact of micro-economic factors of the market have been presented in the report. External as well as internal factors that are supposed to affect the business positively or negatively have been analyzed, which will give a clear futuristic view of the industry to the decision-makers.
The report also helps in understanding the Pharmaceutical Cleaning Validation Market dynamics, structure by analyzing the market segments and project the Pharmaceutical Cleaning Validation Market size. Clear representation of competitive analysis of key players by Grade, price, financial position, Grade portfolio, growth strategies, and regional presence in the Pharmaceutical Cleaning Validation Market make the report investor’s guide.
Pharmaceutical Cleaning Validation Market Scope: Inquire before buying
| Pharmaceutical Cleaning Validation Market | ||||
|---|---|---|---|---|
| Base Year | 2022 | Forecast Period | 2023-2029 | |
| Historical Data | CAGR | Market Size in 2022 | Market Size in 2029 | |
| 2018 to 2022 | 5.7% | US$ 17.20 Bn | US$ 25.36 Bn | |
| Segments Covered | ||||
| by Product Small Molecule Drug Peptides Proteins Cleaning Detergent |
by Validation Test Non-specific Tests Total Carbon Analysis (TC) Total Organic Carbon Testing (TOC) Non-purgeable Organic Carbon (NPOC) Conductivity Others Product-specific Analytical Tests Ultraviolet-Visible Spectroscopy (UV/VIS) High-Performance Liquid Chromatography (HPLC) Liquid Chromatography/ Mass Spectrometry (LC/MS) Others |
|||
| Regions Covered | ||||
| North America United States Canada Mexico |
Europe UK France Germany Italy Spain Sweden Austria Rest of Europe |
Asia Pacific China S Korea Japan India Australia Indonesia Malaysia Vietnam Taiwan Bangladesh Pakistan Rest of APAC |
Middle East and Africa South Africa GCC Egypt Nigeria Rest of ME&A |
South America Brazil Argentina Rest of South America |
Pharmaceutical Cleaning Validation Market Key Players are:
1. Avomeen
2. Lucideon Limited
3. Suez
4. Hach
5. Hach
6. Teledyne Tekmar
7. SGS SA
8. QPharma, Inc
9. ProPharma Group
10.Merck KGaA
11.Intertek Group PLC
12.Kymos S.L
13.Shimadzu Corporation
14.Waters Corporation
15.Azbil Corporation
16.Kimberly-Clark Corporation
17.Ardmac
18.Clean Air Products
19.Labconco Corporation
20.Dynarex Corporation
FAQs:
1. What is the Pharmaceutical Cleaning Validation Market share in 2022?
Ans: Pharmaceutical Cleaning Validation Market share in 2022 was estimated as 17.20 Billion USD.
2. What is the Pharmaceutical Cleaning Validation Market growth?
Ans: The Pharmaceutical Cleaning Validation Market is anticipated to grow with a CAGR of 5.7% in the forecast period and is likely to reach USD 25.36 Billion by the end of 2029.
3. Which Product Type segment is expected to dominate the Pharmaceutical Cleaning Validation Market during the forecast period?
Ans: the small molecule drug segment dominated the market and accounted for the largest revenue share of 46.9%. The large volumes of small drugs are manufactured every year accountable to the highest market share of the segment.
4. Which region is expected to dominate the global Pharmaceutical Cleaning Validation during the forecast period?
Ans: North America dominated the pharmaceutical cleaning validation market and accounted for the largest revenue share of 40.2% in 2022.
5. What is the key driving factor for the growth of the Pharmaceutical Cleaning Validation Market?
Ans: Key factors that are driving the pharmaceutical cleaning validation market growth include stringent regulatory requirements and a rise in pharmaceutical manufacturing.
