Myelodysplastic Syndrome Treatment Market – Global Market Size, Strategic Growth Drivers, Risk Assessment Framework, Regulatory Landscape Review, Competitive Intensity Mapping & Long-Term Industry Outlook to 2030
Overview
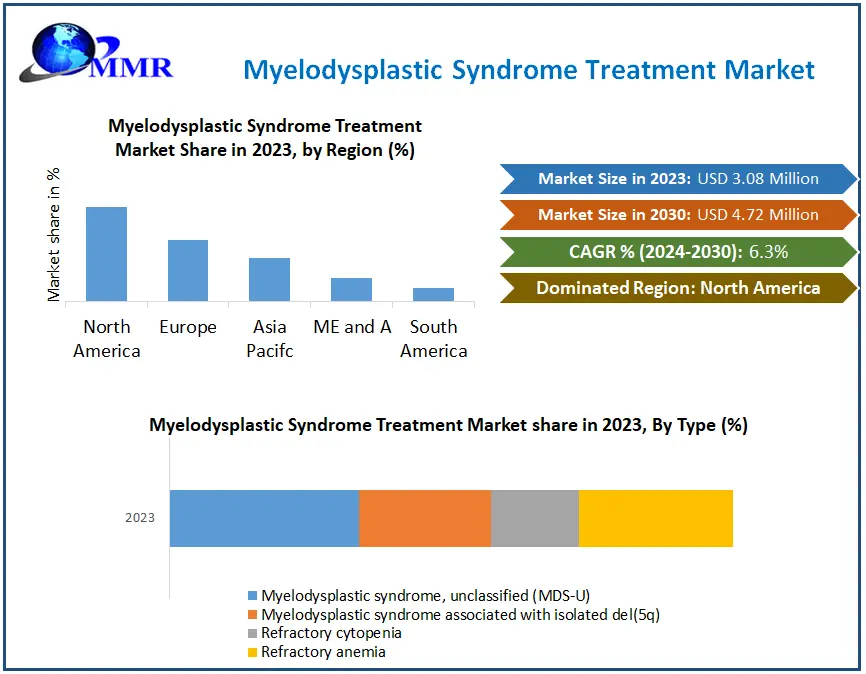
The Myelodysplastic Syndrome Treatment Market size was valued at USD 3.08 Million in 2023 and the total Myelodysplastic Syndrome Treatment Market revenue is expected to grow at a CAGR of 6.3% from 2024 to 2030, reaching nearly USD 4.72 Million by 2030.
Myelodysplastic Syndrome Treatment Market Overview
Myelodysplastic neoplasms (MDS), formerly known as myelodysplastic syndromes, are clonal hematopoietic malignancies that cause morphologic bone marrow dysplasia along with anemia, neutropenia, or thrombocytopenia. MDS are associated with an increased risk of acute myeloid leukemia (AML). The yearly incidence of MDS is approximately 4 per 100 000 people in the United States and is higher among patients with advanced age. The market outlook for Myelodysplastic Syndrome Treatment is influenced by several factors, including advancements in medical technology, increasing awareness, and evolving treatment paradigms.
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
Key trends in the Myelodysplastic Syndrome Treatment Market include the growing emphasis on personalized medicine, a shift towards targeted therapies and immunotherapies, aiming to enhance treatment efficacy while minimizing adverse effects. These trends invent numerous opportunities for stakeholders in the Myelodysplastic Syndrome Treatment Market. The demand for Myelodysplastic Syndrome Treatment is propelled by the rising prevalence of MDS globally, particularly among the aging population. As awareness about early diagnosis and treatment options increases, the demand for effective therapeutic interventions grows. Patients and healthcare providers alike seek treatments that not only improve survival rates but also enhance quality of life, driving demand for innovative therapies and supportive care options.
Myelodysplastic Syndrome Treatment Market Trends:
Stem Cell Transplantation Is New Trends in Myelodysplastic Treatments Market
Trends in the Myelodysplastic Syndrome (MDS) Treatment market reflect advancements and challenges in stem cell transplant (SCT) procedures, which currently offer the most viable option for curing MDS. One trend involves the increasing exploration and utilization of allogeneic SCTs, where patients receive blood-forming stem cells from a donor. Efforts are directed toward improving matching processes between donors and recipients, enhancing treatment efficacy, and reducing the risk of complications. The development of innovative techniques to develop the pool of potential donors for allogeneic Stem Cell transplantation, particularly for patients lacking closely matched related donors, alternative donor sources such as unrelated donors, and umbilical cord blood.
The focus is on optimizing Stem Cell Transplantation protocols to improve outcomes and reduce the incidence of serious side effects, particularly in older patients less tolerant of intensive chemotherapy and radiation. Reduced-intensity conditioning regimens are being increasingly utilized in older patient populations to minimize toxicity while still facilitating engraftment of donor cells. R&D finds novel therapeutic approaches beyond Stem Cell Transplantation, including targeted therapies, immunotherapies, and combination treatments. These approaches aim to address the heterogeneity of MDS and improve patient outcomes, particularly not eligible for or have access to Stem Cell Transplantation. Overall, these trends underscore the evolving landscape of MDS treatment toward more personalized, effective, and accessible therapeutic options.
Myelodysplastic Syndrome Treatment Market Dynamics:
Advancement in Technology and Drug Development in Myelodysplastic Syndrome Treatment Market
A prominent driver for the Myelodysplastic Syndrome (MDS) Treatment Market is the response to the increasing demand for enhanced treatment processes. With approximately 20,000- 24000 new cases diagnosed annually in the United States alone and a prevalence rate of around 3.4 per 104,657 individuals in the general population, the need for effective therapeutic interventions is evident. Particularly among older patient populations, where the incidence rises to approximately 65 instances per 130,000 patients over 65 years old, the urgency to address MDS becomes apparent, especially considering the average age of MDS patients is 75-77.
The association between MDS and the heightened risk of developing acute leukemia further underscores the necessity for improved treatment options. The utilization of chemotherapy, similar to that used for acute myeloid leukemia patients, highlights the overlapping nature of treatment modalities and the potential for cross-application in addressing MDS.
The myelodysplastic syndrome treatment market is also driven by collaborations among MDS treatment companies like pfizer, Otsuka Holdings Co., Ltd., and Bristol-Myers Squibb aimed at fostering better development and innovation. Another factors such as rising incomes, advanced medical treatments, technological infrastructures, and the launch of new, effective products contribute to market growth.
The Limited Number of Approved Drugs for Myelodysplastic Syndrome Treatment (MDS) Facing Challenges
With the limited number of approved myelodysplastic syndrome treatment drugs and the inconstant efficacy of available treatments an insistent need for more effective therapeutic options. This limitation restricts the treatment choices available to healthcare providers and patients, potentially leading to suboptimal outcomes and unmet medical needs within the market. The high-risk nature of MDS, coupled with the limited duration of response to current therapies such as hypomethylating agents (HMAs), poses a considerable challenge. Patients with higher-risk myelodysplastic syndrome treatment experience short-lived responses to treatment, followed by disease progression or transformation to acute myeloid leukemia (AML). This highlights the urgency to enhance the effectiveness of existing treatments and develop novel therapeutic strategies to improve patient outcomes. This is majorly challenging the growth of the myelodysplastic syndrome treatment market.
The lack of significant improvement in survival rates despite the widespread use of certain treatments, such as azacitidine, reflects a persistent gap in addressing the evolving needs of myelodysplastic syndrome patients. This stagnant progress underscores the necessity for continued research and innovation in the field of MDS treatment to achieve meaningful advancements in patient care. As a significant consequence, many MDS patients develop anemia, necessitating regular red blood cell transfusions. The research identified 742 studies, of which 17 met the eligibility criteria. The total annual medical expenses per patient vary, ranging from US$ 9,840 to US$ 19,811 for transient ischemic attack (TI) conditions and from US$ 29,608 to US$ 51,066 for Myelodysplastic Syndrome (MDS).
These challenges signify the complexities and shortcomings present in the current landscape of the Myelodysplastic Syndrome Treatment Market. Addressing these issues requires collaborative efforts from pharmaceutical companies, research institutions, and healthcare providers to develop novel therapies, enhance treatment efficacy, and ultimately improve outcomes for patients with MDS.
Myelodysplastic Syndrome Treatment Market Regional Insights:
The Myelodysplastic Syndrome (MDS) Treatment Market is witnessing significant growth, primarily driven by rising healthcare costs and increasing end-user demand for effective treatments. North America is expected to dominate the market, with a projected market share of 39.97% in 2023, by heightened awareness of MDS among Americans. Initiatives by major players and non-profit organizations, aimed at launching new initiatives and public awareness campaigns, further bolster myelodysplastic syndrome treatment market growth. Novartis and Gilead Sciences have made noteworthy advancements in MDS treatment, with FDA approvals for sabatolimab and magrolimab, respectively, indicating a promising future for innovative therapies in the myelodysplastic syndrome treatment market.
In Europe, governmental initiatives for disease diagnosis and treatment are contributing to significant market expansion, with an anticipated market share of 35.87% in 2023. Initiatives like the European LeukemiaNet WP8 program, which focuses on creating evidence-based guidelines for MDS diagnosis and treatment, demonstrate concerted efforts to enhance patient care and treatment outcomes driven by advancements in therapeutic interventions, increasing awareness, and strategic initiatives by key stakeholders. Asia Pacific is predicted to experience strong revenue market growth throughout the forecast period with the rising number of patients suffering from fungal diseases and infections, major manufacturers increasing R&D activities, hospital-related infections, and government support.
Myelodysplastic Syndrome Treatment Market Scope : Inquire Before Buying
| Global Myelodysplastic Syndrome Treatment Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2023 | Forecast Period: | 2024-2030 |
| Historical Data: | 2018 to 2023 | Market Size in 2023: | US $ 3.08 Mn. |
| Forecast Period 2024 to 2030 CAGR: | 6.3% | Market Size in 2030: | US $ 4.72 Mn. |
| Segments Covered: | by Type | Myelodysplastic syndrome, unclassified (MDS-U) Myelodysplastic syndrome associated with isolated del(5q) Refractory cytopenia Refractory anemia |
|
| by Treatment | Chemotherapy Immunotherapy Stem cell/bone marrow transplantation Growth Factors |
||
| by Patient Group | Above 50 years Below 50 years |
||
Myelodysplastic Syndrome Treatment Market, by Region:
1. North America (United States, Canada, and Mexico)
2. Europe (UK, France, Germany, Italy, Spain, Sweden, Russia, and the Rest of Europe)
3. Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan, and the Rest of APAC)
4. Middle East and Africa (South Africa, GCC, and the Rest of MEA)
5. South America (Brazil, Argentina, Rest of South America)
Key Players in the Myelodysplastic Syndrome Treatment Market:
1. Cleveland Clinic (United States)
2. Mylan N.V. (United States)
3. Pfizer, Inc. (United States)
4. Amgen Inc. (United States)
5. Astex Pharmaceuticals, Inc. (United States)
6. Teva Pharmaceutical Industries Ltd. (Israel)
7. AbbVie (United States)
8. Bristol-Myers Squibb (United States)
9. Sandoz, Inc. (Switzerland)
10. Novartis AG (Switzerland)
11. Accord Healthcare Ltd. (London)
12. Dr. Reddy's Laboratories, Inc. (Hyderabad)
13. Sun Pharmaceutical Industries Limited (Mumbai)
14. LUPIN (Mumbai)
15. Ono Pharmaceutical Co., Ltd. (Japan.)
16. Takeda Pharmaceutical Company Limited (Japan.)
17. Otsuka Holdings Co., Ltd. (Japan.)
FAQs:
1. Which treatment is driving the Market?
Ans. Chemotherapy Treatment drives the Myelodysplastic Syndrome Treatment Market.
2. Which region is expected to lead the global Market during the forecast period?
Ans. North America is expected to lead the global Myelodysplastic Syndrome Treatment Market during the forecast period.
3. What is the projected market size and growth rate of the Market?
Ans. The Myelodysplastic Syndrome Treatment Market size was valued at 3.08 Million in 2023 and the total Myelodysplastic Syndrome Treatment Market revenue is expected to grow at a CAGR of 6.3% from 2024 to 2030, reaching nearly USD 4.72 Million by 2030.
4. What segments are covered in the Myelodysplastic Syndrome Treatment Market report?
Ans. The segments covered in the Myelodysplastic Syndrome Treatment Market report are Type, Treatment, Patients Group, and region.
