Minimal Residual Disease Testing Market Size by Technology, Application, End-User, Region, Industry-Wide Analysis, Competitive Landscape Assessment & Long-Term Forecast to 2032
Overview
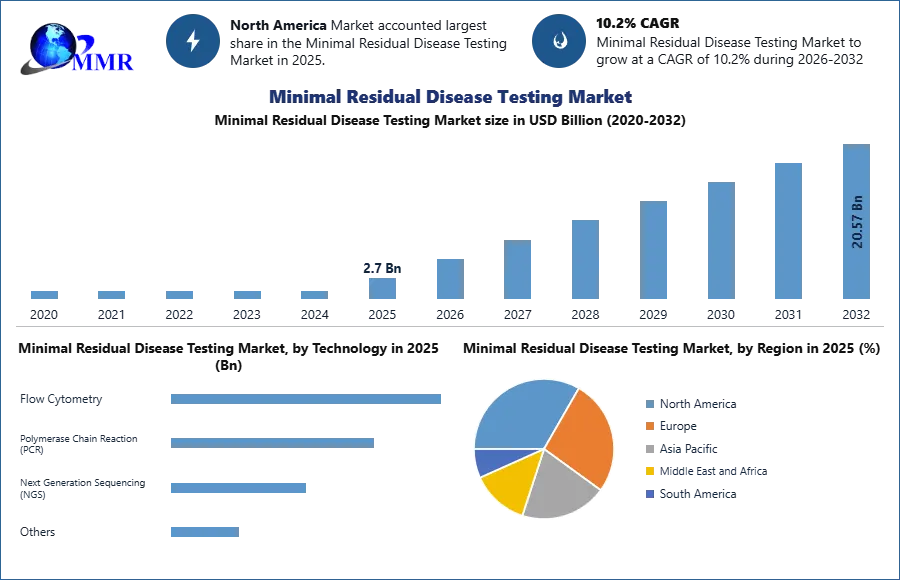
The Minimal Residual Disease Testing Market size was valued at USD 2.7 Billion in 2025 and the total Minimal Residual Disease Testing Market revenue is expected to grow at a CAGR of 10.2% from 2026 to 2032, reaching nearly USD 20.57 Billion by 2032.
Hematologic malignancies include a diverse range of tumors within the hematopoietic and lymphoid tissues, including leukaemia, Hodgkin lymphoma (HL), non-Hodgkin lymphoma (NHL), and multiple myeloma. These cancers arise from uncontrolled cell proliferation and collectively constitute approximately 6.5% of all cancer cases. They rank as the fourth most common type of cancer in both men and women in economically developed countries like the United States, Germany, India, China, etc. In 2020, there are expected to be 85,720 new cases of lymphoma diagnosed in the United States (8,480 cases of HL, 77,240 cases of NHL).
The increasing prevalence of cancer is one of the key factors driving market growth over the forecast period. Globally, cancer is the second-leading cause of death. In 2025, the incidences of cancer reached 20.12 million, and the number of cancer-related deaths was 9.96 million. International Agency for Research on Cancer, cancer incidence is expected to reach 19.45 million by 2027, and 22.6 million by 2030. The rising number of cancer cases is expected to require governments to initiate new programs for cancer screening of the population. Increasing investments in the field of MRD testing programs are driving the market growth. Minimal residual disease testing increasing research in the field of MRD diagnostics and monitoring which is further driving the demand for the minimal residual disease testing market.
Some of the major challenges associated with the market are the high cost associated with testing and the complex regulatory framework associated with the testing. For drug approval, FDA considers both intended use and associated risks, and clinical & analytical information supporting regulatory submission. The average cost associated with the test is over USD 3,500-4000.
To know about the Research Methodology :- Request Free Sample Report
Minimal Residual Disease Testing Market Trends:
Advances Technologies in Testing Influencing the Minimal Residual Disease Testing Market
The advancements in flow cytometry since its inception, the basic principles have remained essentially unchanged for over 60 years, and the technique has primarily focused on fluorescence-based analysis. Flow cytometry traditionally relies on detecting fluorescence signals emitted by fluorochrome-labeled examinations to analyze cellular properties and identify specific cell populations. The fluorescence-based approach has been fundamental in characterizing various biomolecules, including antibodies, nucleic acids, and intracellular markers, providing valuable insights into cellular function, differentiation, and disease states.
Fluorescence-based detection has been the cornerstone of flow cytometry, and recent developments have grown with the technique’s capabilities. For instance, the introduction of spectral flow cytometry has enabled the simultaneous detection of a broader range of fluorochromes, addressing the limitations imposed by the spectral overlap. In addition, the development of mass spectrometry technology has brought about the emergence of mass cytometry, also known as CyTOF (cytometry by Time-Of-Flight). The minimal residual disease testing market growth is influenced by significant R&D investments in biotechnology and life science research, in recent A National Science Foundation (NSF) award was granted for the acquisition of a flow cytometer, supporting advancements in single-cell technologies and revamped teaching modules in immunology and high-throughput discovery.
Minimal Residual Disease Testing Market Dynamics:
The Rising Incidence of Hematologic Malignancies
The rising incidence of hematologic malignancies, including leukemia, lymphoma, and myeloma, globally underscores the pressing need for more precise and sensitive methods like Minimal Residual Disease (MRD) testing for disease monitoring and management. As the frequency of these blood cancers increases, the demand for effective monitoring tools like MRD testing is expected to surge. In the United States, the statistics are compelling. Every 3 minutes, one person is diagnosed with a blood cancer.
MRD testing plays a critical role in managing hematologic malignancies by providing insights into disease burden and treatment response at a molecular level. By detecting residual cancer cells that may not be identified through traditional diagnostic methods, MRD testing allows clinicians to make informed decisions regarding treatment strategies, including the initiation of early interventions to prevent disease relapse. The importance of MRD testing has become increasingly recognized in clinical practice, and the minimal residual disease testing market is poised for significant growth. Companies specializing in molecular diagnostics and oncology are at the forefront of developing and commercializing MRD testing technologies.
For instance, The Exact Sciences’ MRD solution is being designed to utilize a tumor-informed (bespoke) approach with whole genome sequencing. This approach identifies somatic genetic alterations in DNA extracted from the patient tumor tissue and detects a subset of these mutations in ctDNA present in the patient blood. The MRD test that Exact Sciences is developing is intended for patients diagnosed with solid tumor malignancies to detect ctDNA before, during, and after cancer treatment.
Regulatory Hurdles Challenges In The Minimal Residual Disease (MRD) Testing Market
The widespread adoption of MRD testing across different healthcare settings and regions like North America, and Europe are posing a significant challenge. Educating healthcare professionals about the importance and benefits of MRD testing and integrating it into existing clinical workflows are key hurdles to overcome. Regulatory approval processes for MRD tests are firm and time-consuming. Finding authorization from regulatory agencies such as the FDA (Food and Drug Administration) or the EMA (European Medicines Agency) requires extensive clinical validation data, which delays minimal residual disease testing market entry and increases development costs.
Minimal residual disease testing as a general measure of tumor burden has multiple potential regulatory and clinical uses as a biomarker. Depending upon the clinical setting, minimal residual disease testing is used to reflect a patient’s response to treatment or as a prognostic tool to assess a patient’s risk of future relapse. MRD is used to enrich clinical trial populations or guide allocation into specific treatment arms in clinical trials by minimal residual disease testing market key players. There are challenges within each context of use that need to be addressed, such as the underlying disease, patient heterogeneity, therapeutic context, target of therapy, or a combination of disease parameters, to allow effective use of minimal residual disease testing in regulatory decision-making.
Minimal Residual Disease Testing Market Regional Insights:
The preventive measures undertaken by governments to stop the progression of diseases. In North America, the U.S. and Canada are the two major markets for cancer screening due to high awareness of the benefits associated with screening among patients & physicians. North America dominated the minimal residual disease testing market in 2025 with 42.45% of the market share. Moreover, several initiatives are being taken to provide MRD testing in the region. For instance, in November 2022, Natera received a National MRD testing contract with the U.S. Department of Veterans Affairs (VA) National Precision Oncology Program for providing MRD and monitoring services with the company’s Signetera MRD test. For instance, in February 2022, the Foundation for the National Institutes of Health (FNIH) Biomarkers Consortium (BC) launched a project for validating new methods for quantifying and detecting MRD. The strong MRD reimbursement framework is further enhancing the adoption of testing in the region.
The European minimal residual disease testing market accounted for the second-largest market share the growing geriatric population create greater opportunities for the Europe market during the forecast period. The Germany and France minimal residual disease testing market is expected to be the fastest-growing market in the Europe region. The Asia-Pacific market is the fastest growing region from 2026 to 2032, with the growing incidence of Hematological cancer, increased geriatric population, and the strategic formation to meet the demand of customers boosts Asia-Pacific region growth. For instance, the China market accounted for the largest market share in 2025, and the India market is expected fastest-growing market in the Asia-Pacific region.
Recent Industry Developments (2025–2026)
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 14 January 2025 | NeoGenomics / Adaptive Biotechnologies | The companies launched an exclusive multi-year strategic commercial collaboration to advance MRD monitoring for blood cancers. | The partnership integrates clonoSEQ testing into comprehensive diagnostic assessment services, enhancing patient management in the U.S. |
| 22 January 2025 | Guardant Health | Medicare confirmed a $2,007 reimbursement rate for the Guardant Reveal liquid biopsy MRD test. | This financial support broadens patient access to non-invasive recurrence monitoring for colorectal cancer. |
| 11 February 2025 | Myriad Genetics | The U.S. Patent and Trademark Office granted a foundational patent for a tumor-informed, high-definition MRD assay. | The patent secures proprietary sequencing techniques that improve the sensitivity of circulating tumor DNA (ctDNA) detection. |
| 15 April 2025 | Labcorp | Labcorp announced the expansion of its precision oncology portfolio with the release of the "Plasma Detect" MRD solution. | The launch strengthens competitive positioning in Stage III colon cancer recurrence assessment. |
| 18 June 2025 | QIAGEN | The company expanded its MRD portfolio through new strategic partnerships with Tracer Biotechnologies and Foresight Diagnostics. | These collaborations aim to accelerate the development of companion diagnostics for both solid tumors and blood cancers. |
| 20 November 2025 | Abbott / Exact Sciences | Abbott announced a definitive agreement to acquire Exact Sciences for a deal including $1.8 billion of net debt. | The acquisition, expected to close in 2026, consolidates market power in the high-growth cancer screening and MRD segments. |
Minimal Residual Disease Testing Market Competitive Landscape:
Guardant Health's update on the COBRA study underscores its commitment to advancing MRD testing in cancer management. As a leading player in precision oncology, Guardant Health faces competition from other companies developing MRD tests and related technologies. Competitors may include established diagnostic companies, emerging biotechnology firms, and academic institutions conducting research in the field of MRD testing. Guardant Health's continuous innovation and ongoing studies demonstrate its efforts to maintain a competitive edge in the rapidly evolving landscape of MRD testing and precision oncology.
Minimal Residual Disease Testing Market Scope: Inquiry Before Buying
| Minimal Residual Disease Testing Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 2.7 USD Billion |
| Forecast Period 2026-2032 CAGR: | 10.2% | Market Size in 2032: | 20.57 USD Billion |
| Segments Covered: | by Technology | Flow Cytometry Polymerase Chain Reaction (PCR) Next Generation Sequencing (NGS) Others |
|
| by Application | Hematological Malignancy Lymphoma Leukaemia Solid Tumors Others |
||
| by End User | Hospitals and Specialty Clinics Diagnostic Laboratories Academic and Research Institutes Others |
||
Minimal Residual Disease Testing Market by Region:
North America (United States, Canada, and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Russia, and the Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan, and the Rest of APAC)
Middle East and Africa (South Africa, GCC, and the Rest of MEA)
South America (Brazil, Argentina, Rest of South America)
Key Players / Competitors Profiles Covered in Brief in Global Minimal Residual Disease Testing Market Report in Strategic Perspective:
- F. Hoffmann-La Roche Ltd
- Labcorp Inc.
- Guardant Health
- Sysmex Corporation
- NeoGenomics Laboratories, Inc.
- Adaptive Biotechnologies Corporation
- MolecularMD (ICON plc)
- ArcherDX (Invitae Corporation)
- Bio-Rad Laboratories, Inc.
- Natera, Inc.
- OPKO Health, Inc.
- Genetron Health
- Quest Diagnostics, Inc.
- Asuragen, Inc.
- Invivoscribe, Inc.
- ARUP Laboratories, Inc.
- Mission Bio, Inc.
- Cergentis B.V.
- SAGA Diagnostics AB
- Twist Bioscience
- Illumina, Inc.
- Foundation Medicine
- QIAGEN
- Exact Sciences Corporation
- Thermo Fisher Scientific
