Liver Cancer Diagnostics Market by Product Type, End-User, and Region – Global Market Size Estimation, Industry-Wide Analysis, Competitive Landscape Assessment & Long-Term Forecast to 2032
Overview
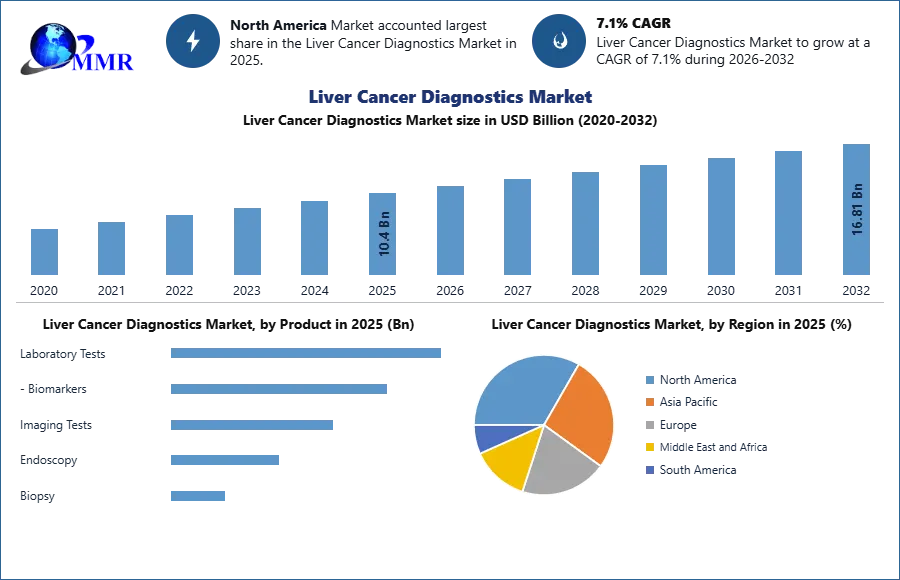
By 2032, the Liver Cancer Diagnostics Market is expected to reach USD 16.81 billion, thanks to growth in the industrial product segment. The report analyzes Liver Cancer Diagnostics Market dynamics by region and end-user industries.
Liver Cancer Diagnostics Market Overview:
The abnormal proliferation of cells in the body is referred to as cancer. Cancerous cells can develop in any part of the body and spread to other sections of the body. Cancer of the liver starts in the cells of the liver. The liver can develop a variety of cancers. Hepatocellular carcinoma is the most prevalent type of liver cancer, and it starts in the hepatocyte. Hepatoblastoma and intrahepatic cholangiocarcinoma are two further kinds of liver cancer. These are regarded as less prevalent.
Hepatocellular carcinomas account for over 80% of adult primary liver malignancies, according to the American Society of Clinical Oncology (ASCO). Techniques for detecting liver cancer are included in liver cancer diagnosis and confirmation.
To know about the Research Methodology :- Request Free Sample Report
Liver Cancer Diagnostics Market Dynamics:
Around the world, liver cancer is one of the top causes of cancer-related deaths. According to the American Cancer Society, the incidence of liver cancer has more than tripled in the last few decades, while death rates have more than doubled. According to the same source, roughly 42,230 new instances of liver cancer would be detected in the United States in 2025, resulting in around 30,230 deaths. Chronic viral hepatitis, Cirrhosis, high alcohol consumption, cigarette consumption, and obesity are all key risk factors for this condition. Although early detection and diagnosis of liver cancer are critical for reducing related mortality, there is a significant demand for novel diagnostic tools.
The Elecsys GALAD score is intended to assist doctors by giving an accurate and prompt diagnosis of hepatocellular cancer. EXACT Sciences Corporation earned FDA breakthrough designation for their blood-based test for early detection of liver cancer in November 2021. CancerSEEK, a liquid biopsy test developed by Johns Hopkins University researchers, was launched in June 2021. The test was created to detect cancers such as stomach, liver, ovarian, and esophageal cancers at an early stage, allowing for early treatment.
The global liver cancer diagnostics market is expected to grow due to an increase in the prevalence of liver cancer, high alcohol consumption, research and development activities such as the development of newer techniques for the diagnosis of liver cancer, the availability of effective cancer treatments, and the adoption of new techniques for the detection of liver cancer. The liver cancer diagnostics market is likely to be hampered by a lack of experienced specialists and a lack of awareness about technical improvements.
Further, to increase companies' market share, key firms are pursuing inorganic growth techniques such as mergers and acquisitions. Exact Sciences, for example, purchased Genomic Health for USD 2.8 billion in July 2025. Exact Sciences bolstered its position in the oncology diagnostics market with this acquisition.
Liver Cancer Diagnostics Market Segment Analysis:
The Industrial products segment is dominating the Test Type segment of the Liver Cancer Diagnostics Market:
The laboratory tests category dominated the market for liver cancer diagnostics in 2025, accounting for 39.5 percent of total revenue. During the forecast period, the segment is expected to increase at the fastest rate. The strong preference for laboratory tests due to their correctness and cost-efficiency can be linked to their big market and expansion. Laboratory tests are also used to assess high-risk patients, determine therapy options, determine disease stages, and determine how well the disease is responding to treatment.
The imaging testing segment had the second-largest revenue share, and it is expected to rise significantly over the research period. Imaging examinations can assist confirm the presence of a tumor in conjunction with laboratory and genetic tests. Computed Tomography (CT) scans, Magnetic Resonance Imaging (MRI) scans, X-rays, ultrasounds, and other radiographic examinations are examples of imaging testing. However, one of the drawbacks of imaging testing is that radiation exposure from the intake of fluorescent and radioactive elements employed in picture production can induce a variety of side effects.
The hospitals and diagnostic laboratories segment are leading the End-User segment of the Liver Cancer Diagnostics Market:
In 2025, the hospitals and diagnostic laboratories segment dominated the market for liver cancer diagnostics, accounting for 82.4 % of revenue. Hospitals are primary care sites for medical disorders such as hepatocellular carcinoma diagnosis and treatment. For disease diagnosis, management, and treatment, the bulk of the population relies on these long-term hospitals. In addition, diagnostic laboratories deliver quick, cost-effective, and highly sensitive results. As a result, they are highly preferred in both established and emerging nations.
Liver Cancer Diagnostics Market Regional Insights:
The Asia Pacific is estimated to dominate the growth of the Liver Cancer Diagnostics Market.
North America dominated the liver cancer diagnostics market in 2025, accounting for 40.6% of revenue, and is expected to continue to do so throughout the forecast period. This region's dominance is due to its well-established healthcare infrastructure, effective health-insurance policies, and widespread use of modern diagnostic technologies. Furthermore, the regional expansion is aided by the presence of major companies on the ground and their ongoing strategic activities. Since of the higher prevalence of hepatocellular carcinoma and strong knowledge about early detection, the United States controls the market share in North America. Further, the high disposable income of individuals and rise in health care investment is estimated to lift the growth of the market in near future.
Recent Industry Developments (2025–2026)
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 19 February 2026 | Pilatus Biosciences Inc. | The FDA granted Fast Track designation to PLT012, an anti-CD36 monoclonal antibody for treating patients with hepatocellular carcinoma (HCC). | This designation accelerates the development of novel targeted therapies for solid tumors that do not respond to existing immunotherapies. |
| 15 December 2025 | Helio Genomics | Helio Genomics formed a strategic partnership with LiverRight to integrate the LiverTrace blood-based test into a nationwide virtual hepatology network. | This collaboration expands patient access to early detection tools and immediate specialist interpretation across all 50 U.S. states. |
| 11 November 2025 | Exact Sciences Corporation | The company revealed key clinical validation outcomes from the ALTUS Study, evaluating multi-biomarker blood tests for liver cancer screening. | Positive validation data strengthens the commercial viability of non-invasive liquid biopsy tests as alternatives to traditional ultrasound. |
| 15 August 2025 | BiBBInstruments AB | Henry Ford St. John Hospital tracked the primary U.S. case range utilizing the EndoDrill GI for endoscopic ultrasound-guided liver biopsies. | This clinical milestone marks the U.S. commercial entry of advanced electric biopsy sampling technology for more accurate tissue acquisition. |
| 14 May 2025 | Roche Diagnostics | The Elecsys PRO-C3 assay was utilized with the ADAPT formula to estimate liver fibrosis severity in clinical settings. | Enhanced diagnostic accuracy for fibrosis allows for earlier identification of high-risk patients before the progression to hepatocellular carcinoma. |
The objective of the report is to present a comprehensive analysis of the Liver Cancer Diagnostics Market to the stakeholders in the industry. The past and current status of the industry with the forecasted market size and trends are presented in the report with the analysis of complicated data in simple language. The report covers all the aspects of the industry with a dedicated study of key players that include market leaders, followers, and new entrants.
PORTER, SWOT, PESTEL analysis with the potential impact of micro-economic factors of the market have been presented in the report. External as well as internal factors that are supposed to affect the business positively or negatively have been analyzed, which will give a clear futuristic view of the industry to the decision-makers.
The report also helps in understanding the Liver Cancer Diagnostics Market dynamics, structure by analyzing the market segments and projecting the Liver Cancer Diagnostics Market size. Clear representation of competitive analysis of key players by Grade, price, financial position, Grade portfolio, growth strategies, and regional presence in the Liver Cancer Diagnostics Market make the report investor’s guide.
Liver Cancer Diagnostics Market Scope: Inquire before buying
| Liver Cancer Diagnostics Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 10.4 USD Billion |
| Forecast Period 2026-2032 CAGR: | 7.1% | Market Size in 2032: | 16.81 USD Billion |
| Segments Covered: | by Product | Laboratory Tests - Biomarkers Imaging Tests Endoscopy Biopsy |
|
| by Biomakers | Oncofetal and Glycoprotein Antigens Enzymes and Isoenzymes Growth Factors and Receptors Molecular Markers Pathological Biomarkers |
||
| by End User | Hospitals & Diagnostic Laboratories Academic & Research Institutes Pharmaceutical & CRO Laboratories Poultry and Aquaculture |
||
Liver Cancer Diagnostics Market, by Region:
• North America
• Europe
• Asia Pacific
• Middle East and Africa
• South America
Key Players / Competitors Profiles Covered in Brief in Global Liver Cancer Diagnostics Market Report in Strategic Perspective:
- Abbott Laboratories
- Thermo Fisher Scientific Inc.
- F. Hoffmann-La Roche Ltd
- Qiagen N.V.
- Siemens Healthineers
- Becton, Dickinson and Company
- Illumina, Inc.
- Epigenomics AG
- Koninklijke Philips N.V.
- Fujifilm Medical Systems U.S.A., Inc.
- GE HealthCare
- BioMérieux S.A.
- Foundation Medicine, Inc.
- Perspectum
- Helio Genomics
- Agilent Technologies, Inc.
- Sysmex Corporation
- Canon Medical Systems Corporation
- Hologic, Inc.
- Exact Sciences Corporation
- Guardant Health
- Fujirebio
- Tosoh Corporation
- Boditech Med Inc.
- Diazyme Laboratories, Inc.
