Proliferative Vitreoretinopathy Market Size – Industry Structure Evaluation, Demand Drivers Analysis, Regional Growth Analysis and Identification, Competitive Positioning Review & Global Market Size Forecast to 2032
Overview
Global Orthopedic Bone Cement and Casting Material Market size was valued at USD 760.92 Mn. in 2025, and the total Gas Turbine revenue is expected to grow at a CAGR of 5.30% from 2026 to 2032, reaching nearly USD 1092.29 Mn. by 2032.
Global Proliferative Vitreoretinopathy Market Definition
Proliferative vitreoretinopathy is a disease process that follows the proliferation of ectopic cell sheets in the vitreous and/or perirenal area, causing epiretinal membrane formation and traction, in patients with rhegmatogenous retinal detachments.
The report covers the current estimated and forecasted data for the Global Proliferative Vitreoretinopathy Market on a global and regional level. The report provides an in-depth analysis of the Global Proliferative Vitreoretinopathy Market for the period 2025 – 2032, wherein 2025 is the base year and the period from 2026 – 2032 is the forecast period.
The report provides qualitative and quantitative insights on the Proliferative Vitreoretinopathy industry trends and a detailed analysis of market size and growth rate of all segments in the market. The Global Proliferative Vitreoretinopathy Market is segmented by End-User and Region.
Global Proliferative Vitreoretinopathy Market Dynamics
According to the research conducted by the Retina Society Terminology committee, there is 5%-10% progress in proliferative vitreoretinopathy every year.
Consideration of pathobiological effects of PVR and identification of risk factors are increasing the successful operation rate of PVR. The adoption of endolaser applications in the PVR and standard surgical procedures are driving the global market. Tremendous advances in surgical techniques and technology in the ophthalmology field are propelling the global proliferative vitreoretinopathy market. The Increasing Prevalence of patients having retinoblastoma is driving the global market.
Global Proliferative Vitreoretinopathy Market Segment Analysis – by End-User
To know about the Research Methodology :- Request Free Sample Report
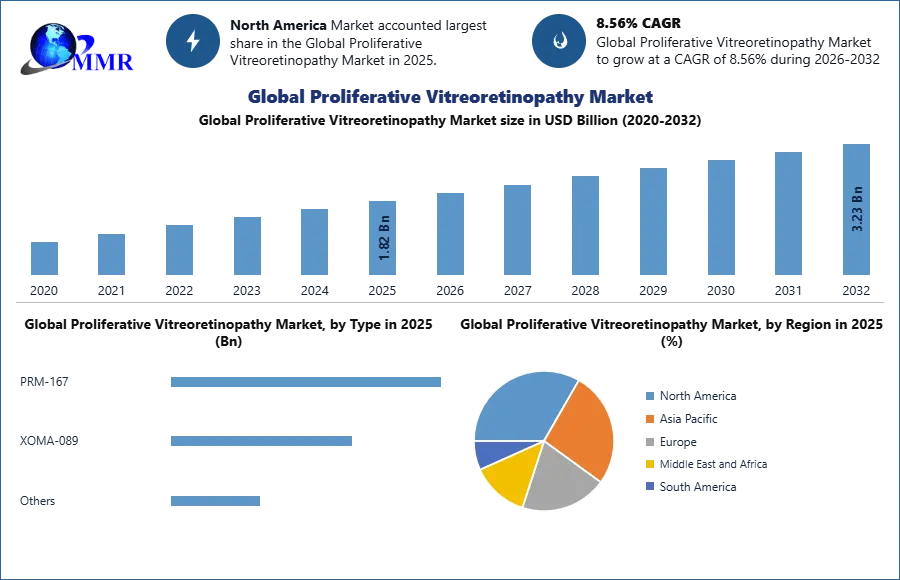
Hospitals commanded the largest market share of xx% in the global proliferative vitreoretinopathy market.
Testing of different drugs and drug-delivery systems for the vitreoretinopathy enhance the hospital treatment procedures of vitreoretinopathy. The occurrence of PVR in up to 50% of peoples who have had an open globe injury is boosting the global market. Innovation of new surgeries in retinal surgery and adoption of techniques in the operation procedures are driving the global vitreoretinopathy market.
Global Proliferative Vitreoretinopathy Market Regional Analysis
North America contributes the largest share for the growth of the global proliferative vitreoretinopathy market.
The ongoing research & development activities by the government and private research organizations are expected to project the global market in the forecast period. The growing interest of scientists in the findings of targeted therapeutics for vitreoretinopathy is resulting in the improvement of treatment and curation. Adoption of medical devices in the retinal operation procedure commended the largest market share of xx% in the global market.
The objective of the report is to present a comprehensive analysis of the global proliferative vitreoretinopathy market including all the stakeholders of the industry. The past and current status of the industry with forecasted market size and trends are presented in the report with the analysis of complicated data in simple language.
The report covers all the aspects of the industry with a dedicated study of key players that include market leaders, followers, and new entrants. PORTER, SVOR, PESTEL analysis with the potential impact of micro-economic factors of the market have been presented in the report. External as well as internal factors that are supposed to affect the business positively or negatively have been analyzed, which will give a clear futuristic view of the industry to the decision-makers.
The report also helps in understanding the global proliferative vitreoretinopathy market dynamics, structure by analyzing the market segments and project the global proliferative vitreoretinopathy market clear representation of competitive analysis of key players by price, financial position, by detection and equipment portfolio, growth strategies, and regional presence in the global proliferative vitreoretinopathy market make the report investor’s guide.
Global Aeromonads Diagnostic Testing Market Recent Developments:
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 17 March 2026 | Aldeyra Therapeutics | Aldeyra reported its intent to shift focus toward ADX-2191 (intravitreal methotrexate) for rare retinal diseases following a Complete Response Letter (CRL) for its dry eye candidate. | This strategic reallocation of R&D resources aims to accelerate the development of ADX-2191 as a potential first-in-class treatment for preventing proliferative vitreoretinopathy. |
| 25 February 2026 | Veeva Clinical Trials | A new clinical trial was registered to evaluate the safety and efficacy of intravitreal topotecan specifically for patients with rhegmatogenous retinal detachment due to PVR. | The study investigates topotecan’s anti-proliferative activity as a possible pharmacological adjuvant to vitrectomy, targeting a reduction in post-surgical recurrence rates. |
| 19 February 2026 | Apellis Pharmaceuticals | Apellis presented five-year safety data from the GALE extension study, monitoring patients for adverse events including retinal detachment and PVR following complement inhibition. | Long-term safety profiles for intravitreal therapies are vital for establishing risk-benefit ratios in chronic retinal management where PVR remains a severe complication. |
| 08 February 2026 | ResearchGate / Int Ophthalmol | A comprehensive review identified methotrexate and infliximab as the most promising candidates for pharmacological modulation of PVR in high-risk populations. | The analysis provides a standardized protocol framework for future randomized trials, emphasizing the transition from purely surgical management to adjunctive molecular therapy. |
| 28 August 2025 | Aldeyra Therapeutics | Aldeyra received Orphan Designation from the European Medicines Agency (EMA) for ADX-2191 for treating primary large B-cell lymphomas and related retinal diseases. | The designation grants ten years of market exclusivity in the EU, significantly boosting the commercial valuation of the vitreous-compatible methotrexate formulation. |
| 19 August 2025 | Aldeyra Therapeutics | The U.S. FDA granted Fast Track Designation to ADX-2191 to expedite the review of treatments for rare genetic retinal diseases. | This regulatory milestone facilitates frequent communication with the FDA, potentially shortening the timeline for bringing PVR-prophylactic agents to the clinical market. |
Global Aeromonads Diagnostic Testing Market Report Scope: Inquire before buying
| Global Proliferative Vitreoretinopathy Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 1.82 USD Billion |
| Forecast Period 2026-2032 CAGR: | 8.56% | Market Size in 2032: | 3.23 USD Billion |
| Segments Covered: | by Type | PRM‑167 XOMA‑089 Others |
|
| by End‑User | Hospital Clinic Ambulatory Surgery Centers (ASCs) Others |
||
Global Proliferative Vitreoretinopathy Market, by Region
• North America
• Europe
• Asia Pacific
• Middle East and Africa
• South America
Global Proliferative Vitreoretinopathy Market Key Players
- Novartis AG
- Regeneron Pharmaceuticals, Inc.
- Genentech, Inc.
- Pfizer, Inc.
- Allergan plc (AbbVie Inc.)
- Bausch + Lomb Corporation
- Promedior Inc.
- RXi Pharmaceuticals Corp
- Clearside Biomedical, Inc.
- Oxurion NV
- Adverum Biotechnologies, Inc.
- Aldeyra Therapeutics, Inc.
- Carl Zeiss Meditec AG
- HOYA Corporation
- Essilor International S.A.
- HAAG-Streit Holding AG
- Johnson & Johnson Vision
- Nidek Co. Ltd.
- Topcon Corporation
- Ziemer Ophthalmic Systems AG
- Hoffmann-La Roche Ltd
- Bayer AG
- Santen Pharmaceutical Co., Ltd.
- Alcon (Novartis/Independent)
- Intra-Cellular Therapies
- Regulus Therapeutics
- Kodiak Sciences
- Iveric Bio
- Aerpio Pharmaceuticals
- Apellis Pharmaceuticals
Others
