Prenatal Genetic Testing Market Size by Tests, End Users, Technology, Region, Industry-Wide Analysis, Competitive Landscape Assessment & Long-Term Forecast to 2032
Overview
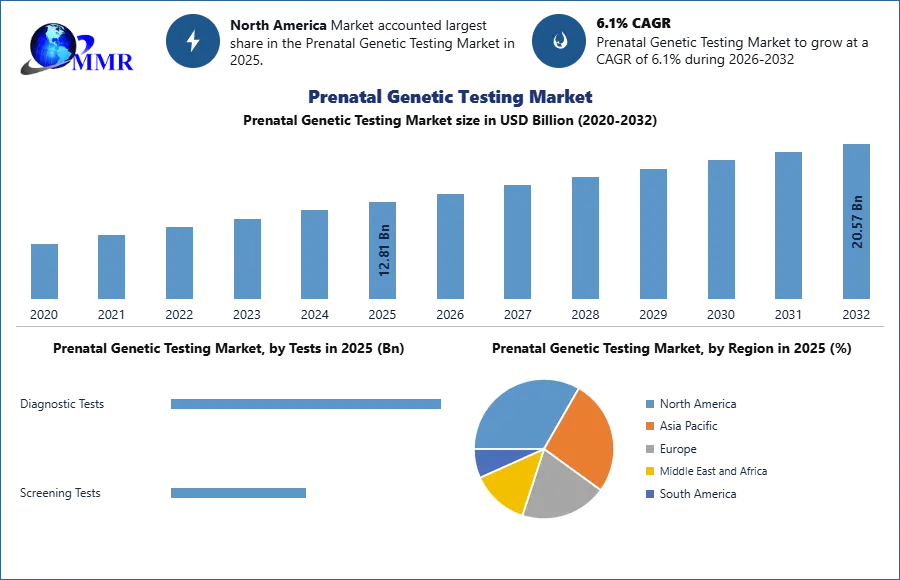
Global Prenatal Genetic Testing Market size was valued at USD 12.81 Bn. in 2025, and the total Prenatal Genetic Testing Market revenue is expected to grow by 6.1% from 2025 to 2032, reaching nearly USD 20.57 Bn.
Prenatal Genetic Testing Market Overview:
Prenatal genetic testing detects genetic abnormalities in the fetus and offers information regarding how inherited disorders and chromosomal abnormalities, such as Down Syndrome or Trisomy 18 and 13, can be characterized. Testing is provided for facilitating more timely and medically informed care in all hospitals, diagnostics labs, and fertility centres. Factors, such as the trend of childbearing at increased maternal age, greater informedness amongst expecting parents, and advancing non-invasive testing technologies, are all contributing to the increased demand for prenatal testing, companies such as Natera and Illumina have made available excellent accuracy, non-invasive prenatal testing (NIPT) platforms that leverage cell-free fetal DNA from maternal blood to provide estimates of risk for potential disorders while maintaining the fetus's safety. Demand for testing, in particular, continues to emanate from the biggest markets located primarily in North America and Asia Pacific, due to continued adoption of assisted reproductive technologies (ART), ever-improving access to healthcare infrastructure in target regions, and increasingly permissive insurance coverage for genetic testing.
Illumina, Thermo Fisher Scientific, Natera, Eurofins Scientific, and BGI Genomics are major contributors to the global prenatal genetic testing market, offering technologies from next-generation sequencing (NGS) to microarrays and biochemical screening platforms. The companies are investing intently in AI-based genetic analytics, improving test sensitivity features for rare disorders, and increasing access in underserved regions through population-specific panels. Positive tailwinds from regulatory support, such as insurance reimbursement in the U.S. and public screening in countries like Japan and China, are expected to enhance the global prenatal genetic testing market.
The Prenatal Genetic Testing Report also covered segment-wise analysis by test type, technology, end users, and regions; recent developments from leading companies; competitive analysis; market share (2024); and emerging trends, including increased adoption of AI, expanded carrier screening, and regional genomics adaptation, giving a holistic analysis of the global prenatal genetic testing ecosystem.
To know about the Research Methodology :- Request Free Sample Report
Global Prenatal Genetic Testing Market Dynamics:
Rising Maternal Age to Drive Prenatal Genetic Testing Market Growth
Advanced maternal age refers to a pregnancy in which the individual giving birth is older than 35 years. Pregnant individuals in this age group face a higher risk of complications such as miscarriage, congenital disorders, and high blood pressure. Screening tests play a crucial role in detecting specific congenital disorders. This is increasing the demand for tests, which is contributing to the Prenatal Genetic Testing Market growth. While genetic screening tests are recommended for all pregnancies, they become particularly significant for individuals of advanced maternal age. These noninvasive screenings, conducted through blood tests or ultrasound, are optional and can identify if the baby is at a heightened risk for certain congenital conditions.
It's important to note that these tests are screenings, indicating they detect the possibility of a genetic condition. In cases where a screening test indicates a potential issue, invasive genetic testing methods may be employed. Chorionic villus sampling (CVS) is one such method, using cells from the placenta to diagnose genetic conditions. Amniocentesis is another invasive testing procedure, involving the insertion of a needle through the abdomen to extract amniotic fluid from the uterus. This fluid analysis aids in diagnosing certain genetic conditions.
Technological Advancement to Create Prenatal Genetic Testing Market Opportunity
The utilization of Next Generation Sequencing (NGS)-based methods has been predominant, but ongoing technological advancements, including qPCR, dPCR, and amplification technologies, aim to improve test quality and reduce costs. The commercial sector is expanding its focus to encompass sex chromosome aneuploidies, rare autosomal trisomies, and sub-microscopic copy-number variants. Automated in-lab workflows and remote patient monitoring during pregnancy are empowering providers to effectively manage pregnancies and associated risks.
A new generation of screening tests is emerging, promising to enhance pregnancy management by offering real-time information about placental development through trophoblast cells. This indicates a shift towards targeted management of pregnancies at risk, which is expected to drive the Prenatal Genetic Testing Market in the future. As technology evolves, discussions about expanding Non-Invasive Prenatal Testing (NIPT) beyond chromosomal abnormalities are expected to intensify. Therefore, the involvement of private clinics and biomedical professionals is crucial for facilitating access, particularly in underpenetrated regions. Validation studies serve as a central strategy to attract customers.
While establishing clear guidelines and services to address potential medical and ethical issues is imperative for the responsible future use of NIPT, leveraging complementary informatics technologies to enhance data analysis and reporting capabilities will be vital for expediting new product development. Additionally, expanding the core intellectual property portfolio and refining sample preparation technology are essential aspects of staying at the forefront of NIPT advancements.
Legal and Regulatory Frameworks to Create Prenatal Genetic Testing Market Challenge
Prenatal genetic testing is subject to many legal and regulatory frameworks across the world. Harmonizing these regulations to ensure ethical standards, patient privacy, and consistent practices poses a challenge for the Global Prenatal Genetic Testing Market. For example, the Centers for Medicare and Medicaid Services (CMS) enforces regulations to govern the analytical validity of clinical genetic tests, while federal oversight of the clinical validity of most genetic tests is currently absent. Recognizing this gap, the FDA has put forth new policies aimed at strengthening the regulation of analytical validity and broadening oversight to encompass the clinical validity of genetic tests.
CMS oversees clinical laboratories, including those engaged in clinical genetic testing, through the Clinical Laboratory Improvement Amendments (CLIA) program. Enacted in 1988, CLIA mandates a certification process that laboratories must undergo to lawfully perform clinical testing. The primary goal of CLIA is to assess and ensure the quality of clinical testing, encompassing the validation of procedures and the qualifications of the technicians administering the tests. Additionally, the program includes proficiency testing for certain types of tests.
Global Prenatal Genetic Testing Market Segment Analysis:
Based on Tests, the market is segmented into Diagnostic Tests (sub-segments: Amniocentesis, Chorion villus biopsy, Ultrasonography, and Maternal serum markers) and Screening Tests (sub-segments: Carrier Screening, Sequential Screening, and Maternal Serum Quad Screening). The Diagnostic Tests segment is expected to grow at a high rate during the forecast period, and among which the Non-invasive prenatal testing (NIPT) held the largest share. This is attributed to NIPT's non-invasive nature, lower risk, and wider range of conditions, which is contributing to its dominant share. The Screening Tests segment held the largest Prenatal Genetic Testing Market share in 2025, among which, the Carrier Screening segment held the largest share. It is attributed to its ability to identify potential genetic disease carriers before conception or early in pregnancy, promoting informed reproductive choices. This majorly contributes to its market lead.
Based on End Users, the Prenatal Genetic Testing Market is segmented into Hospitals, Specialty clinics and Diagnostic centers. Hospitals segment dominated the prenatal genetic testing market in 2025 and is expected to hold largest market share over forecast period. Hospitals commonly have a considerable share of the market because they are full-service providers for prenatal care that offer both screening and diagnostic options in one place. Most hospitals can maintain high-end technology, as well as have genetic counselors and specialists, making them the preeminent and safest choice for high-risk pregnancy management. Hospitals represent the easiest and most likely choice for patients (compared to private medical clinics or practitioners) because many hospitals offer broader insurance coverage and are often paid through public health programs. Hospitals are also capable of handling the greater volumes of patients and incidents as needed. Collectively, these elements have a strong likelihood of positioning hospitals as the preferred primary provider for prenatal genetic testing.
Prenatal Genetic Testing Market Regional Insights
North American Prenatal Genetic Testing Market dominated the global market with the largest share in 2025. This is attributed to the high research and development in the healthcare sector. The increase in incidence of all hypertensive disorders of pregnancy is the driving factor for global prenatal genetic testing in North America. Prenatal genetic screenings are generally done in the countries of North America for patients who would benefit from genetic counselling. Doctors and clinicians recommend prenatal genetic testing as a precaution to avoid unnecessary health risks to both mother and fetus.
Asia Pacific Prenatal Genetic Testing Market is expected to grow at a high rate during the forecast period. This is attributed to the increasing disposable income, rising awareness of genetic disorders, and expanding healthcare facilities. Several countries in the region have initiated government-supported programs to promote prenatal care and genetic screening. These initiatives aim to improve awareness, accessibility, and affordability of prenatal genetic testing services. In the region, China is the largest market for Prenatal Genetic Testing. The country is experiencing a growth in prenatal genetic testing, which is driven by a growing middle class, advanced healthcare infrastructure, and rising awareness.
Recent Industry Developments (2025–2026)
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 15 August 2025 | Natera, Inc. | Launched Fetal Focus, a non-invasive prenatal test (NIPT) designed specifically for single-gene disorders when paternal samples are unavailable. | The launch expands clinical utility for mothers in complex family scenarios where biological father testing is not possible. |
| 12 June 2025 | Galatea Bio | Partnered with Fabric Genomics and Broad Clinical Labs to integrate polygenic risk scoring (PRS) into standard genetic testing workflows. | This collaboration enhances clinical depth by providing a more comprehensive view of genetic susceptibility to common diseases. |
| 22 May 2025 | Gene Solutions | Formed a strategic partnership with NEWCL Biomedical Laboratory to establish a state-of-the-art NGS facility in Taiwan. | The expansion strengthens regional infrastructure in Asia-Pacific, increasing the local capacity for advanced prenatal screening. |
Prenatal Genetic Testing Market Scope: Inquire before buying
| Prenatal Genetic Testing Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 12.81 USD Billion |
| Forecast Period 2026-2032 CAGR: | 6.1% | Market Size in 2032: | 20.57 USD Billion |
| Segments Covered: | by Tests | Diagnostic Tests Screening Tests |
|
| by Technology | Non-Invasive Prenatal Testing Invasive Testing Technology Biochemical Screening |
||
| by End Users | Hospitals Specialty clinics Diagnostic centers |
||
Prenatal Genetic Testing Market by Region:
North America (United States, Canada, and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria, and the Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, ASEAN, and the Rest of APAC)
Middle East & Africa (South Africa, GCC, Nigeria, Rest of ME&A)
South America (Brazil, Argentina, Rest of South America)
Key Players / Competitors Profiles Covered in Brief in Global Prenatal Genetic Testing Market Report in Strategic Perspective:
- Illumina, Inc.
- Thermo Fisher Scientific Inc.
- Natera Inc.
- Eurofins Scientific
- BGI Genomics Co., Ltd.
- F. Hoffmann-La Roche Ltd
- Laboratory Corporation of America Holdings
- Quest Diagnostics Incorporated
- Myriad Genetics, Inc.
- Agilent Technologies Inc.
- Berry Genomics Co., Ltd.
- BillionToOne, Inc.
- BioReference Laboratories, Inc.
- Igenomix S.L.U.
- Lilac Insights Pvt. Ltd.
- MedGenome Labs Ltd.
- Sequenom Laboratories
- Ariosa Diagnostics
- LifeCodexx AG
- Annoroad Gene Technology
- Invitae Corporation
- Baylor Genetics
- Neuberg Diagnostics
- Gene Solutions
- Fulgent Genetics, Inc.
