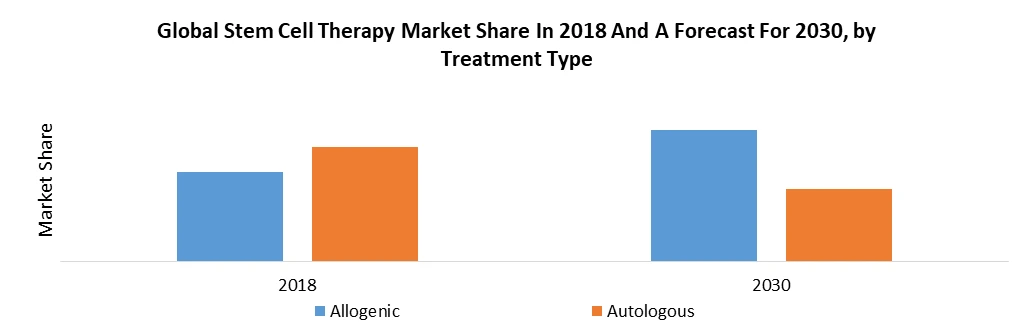Mesenchymal Stem Cells Market – Industry Structure Evaluation, Demand Drivers Analysis, Regional Growth Analysis and Identification, Competitive Positioning Review & Global Market Size Forecast to 2030
Overview
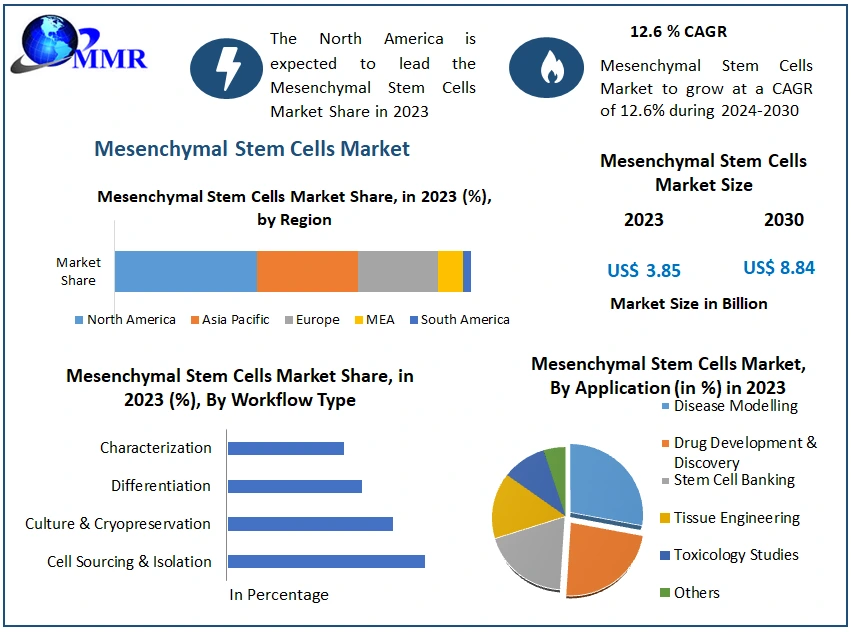
The Mesenchymal Stem Cells Market size was valued at USD 3.85 Billion in 2023 and the total Mesenchymal Stem Cells Market revenue is expected to grow at a CAGR of 12.6 % from 2024 to 2030, reaching nearly USD 8.84 Billion.
Mesenchymal Stem Cells (MSCs) are multipotent stromal cells that can differentiate into a variety of cell types, including osteoblasts (bone cells), chondrocytes (cartilage cells), adipocytes (fat cells), and myocytes (muscle cells). They are found in various tissues, including bone marrow, adipose tissue, umbilical cord tissue, and dental pulp. MSCs have garnered significant attention in regenerative medicine due to their potential therapeutic applications in tissue repair, immunomodulation, and inflammation regulation. These cells possess unique immunomodulatory properties, making them promising candidates for treating autoimmune diseases, graft-versus-host disease (GVHD), and inflammatory conditions. The Mesenchymal Stem Cells (MSCs) market is witnessing steady growth, driven by increasing research activities, the rising prevalence of chronic diseases, and growing investments in regenerative medicine. The market includes various segments, including cell sourcing, application, and end-user industries such as biotechnology and pharmaceuticals. North America currently leads the market, supported by advanced healthcare infrastructure and favorable regulatory policies. However, Asia Pacific is expected to emerge as a key growth driver, fueled by increasing healthcare expenditures, expanding research initiatives, and rising adoption of advanced therapies.
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
The growing demand for regenerative therapies, increasing investments in stem cell research, and the rising prevalence of chronic diseases. Key market players are focusing on developing innovative therapies and expanding their product portfolios to capitalize on emerging opportunities. Regulatory approvals for MSC-based treatments, such as BioCardia's CardiALLO trial for ischemic heart failure, validate the safety and efficacy of these therapies. Additionally, collaborations between academic institutions and biotech firms accelerate product development and market growth. Advancements in cell culture techniques, increasing awareness of regenerative medicine, and the growing aging population. MSC-based therapies offer potential solutions to age-related health issues and degenerative conditions. Moreover, trends such as personalized medicine and the adoption of automation and bioreactor systems in cell therapy manufacturing are shaping the market landscape. Emerging applications of MSCs in orthopedics, neurology, and oncology further contribute to market growth, creating opportunities for market players to expand their presence and offerings. Companies are focusing on strategic collaborations, acquisitions, and product launches to strengthen their foothold in the market. For instance, Thermo Fisher Scientific and Axol Bioscience are leveraging MSC technology to support drug discovery for neurological diseases such as Alzheimer's and ALS. Additionally, advancements in genetic engineering and cell therapy manufacturing technologies are opening new avenues for developing more effective MSC-based therapies. The MSCs market is poised for significant growth, driven by evolving therapeutic applications, increasing investments, and favorable regulatory environments.
Market Dynamics:
Rising Prevalence of Chronic Diseases Drives MSC Therapy Demand:
The rising prevalence of chronic diseases, such as cardiovascular disorders, neurological conditions, and orthopedic injuries, drives the demand for mesenchymal stem cell (MSC) therapies. For example, BioCardia's CardiALLO™ trial addresses ischemic heart failure, a common outcome of vascular disease. Ongoing advancements in stem cell research, including the development of innovative differentiation techniques and an enhanced understanding of cell behavior, propel the growth of the MSC market. Research breakthroughs, such as the generation of high-quality cartilage from iPS cell-derived MSCs, offer promising avenues for tissue repair. Regulatory approvals for MSC-based therapies, like the FDA's approval of Omisirge for blood cancer patients, boost market growth by validating the safety and efficacy of these treatments.
Growing investments in biotechnology companies conducting MSC research and clinical trials fuel market growth. For instance, partnerships between academic institutions and biotech firms, such as Cynata Therapeutics' collaboration with LUMC for kidney transplant trials, drive innovation and market growth. The versatility of MSCs allows for their use in various therapeutic areas, including orthopedics, neurology, and oncology, broadening their market potential. Diverse applications, such as reducing the need for anti-rejection drugs in kidney transplant recipients, demonstrate the wide-ranging benefits of MSC therapies. Advances in cell therapy manufacturing technologies, such as automation and bioreactor systems, streamline production processes, improve scalability, and reduce costs, fostering market growth.
The aging population worldwide contributes to the demand for regenerative therapies to address age-related diseases and degenerative conditions. MSC-based treatments offer potential solutions for age-related health issues, driving market growth. The proliferation of clinical trials and research initiatives exploring the therapeutic potential of MSCs fuels market growth by generating valuable data and expanding treatment options. Initiatives like the Nereid Study investigating MSCs in renal transplant recipients contribute to the accumulation of evidence supporting their efficacy. Growing awareness among patients about the benefits of MSC therapies, coupled with increasing acceptance of regenerative medicine approaches, drives demand for these treatments. Patient advocacy and education initiatives contribute to market growth. Collaboration between industry players, academic institutions, and healthcare organizations on a global scale fosters innovation, accelerates clinical development, and expands market reach. Strategic partnerships facilitate access to diverse expertise, resources, and patient populations, driving the growth of the MSC market.
Personalized Medicine Trend Spurs MSC Therapy Demand:
Mesenchymal stem cells (MSCs) show promise in treating various diseases beyond their current indications. For example, Axol Bioscience explores MSCs' potential in Alzheimer's and ALS therapies, expanding the market's scope. Advances in cell culture techniques and genetic engineering enable the development of more effective MSC-based therapies. For instance, Cytori Therapeutics focuses on enhancing MSCs' immunomodulatory properties to improve treatment outcomes for various conditions, driving market growth. Regulatory approvals of new MSC-based therapies pave the way for market growth. For example, BioCardia's CardiALLO trial, aimed at treating ischemic heart failure with allogeneic MSCs, demonstrated regulatory recognition and market potential. Collaborations between research institutions and industry players facilitate knowledge sharing and accelerate product development. StemCell Applications and StemoniX collaborate to advance life science research, leveraging MSCs' therapeutic potential and fostering market growth.
The trend toward personalized medicine drives demand for MSC-based therapies tailored to individual patient needs. Companies like Cyagen Biosciences offer customized MSC products, catering to the personalized medicine trend and fueling market growth. Increasing public awareness of regenerative medicine and stem cell therapies boosts demand for MSC-based treatments. Companies like PromoCell GmbH engage in educational initiatives to raise awareness about the therapeutic potential of MSCs, driving market growth. Continued investment in research and development drives innovation in MSC-based therapies, expanding the market. Companies like Lonza AG allocate resources to R&D to develop novel MSC-based products, driving market growth through innovation. Growth opportunities exist in emerging markets with increasing healthcare expenditure and demand for advanced therapies. For example, Asia-Pacific countries like China and Japan witness rising adoption of MSC-based treatments, presenting lucrative opportunities for market growth.
Regional Regulatory Variations in the Mesenchymal Stem Cell Market:
The exact mechanisms of action of mesenchymal stem cells (MSCs) are not fully understood, hindering the development of optimized treatments. For example, while MSCs show promise in treating neurological disorders like Alzheimer's disease, their precise mode of action in the central nervous system remains elusive, impeding the translation of preclinical findings into effective clinical therapies. Standardizing MSC production and characterization processes poses a significant challenge due to variations in cell sourcing, isolation techniques, and culture conditions. This lack of standardization lead to variability in therapeutic outcomes and regulatory hurdles. For instance, differences in manufacturing protocols result in variability in the potency and safety profiles of MSC-based products, impacting their regulatory approval and market adoption. MSCs still elicit immune responses in recipients, leading to safety concerns and potential adverse reactions. For example, studies have reported instances of immune rejection and tumor formation associated with MSC therapy, raising safety concerns and regulatory scrutiny. Addressing these safety issues is crucial for gaining regulatory approval and ensuring market acceptance of MSC-based therapies. The mesenchymal stem cells market faces challenges due to the complex regulatory landscape governing cell therapy products. Regulatory requirements vary across regions, leading to inconsistencies in approval processes and market access. For instance, differences in regulatory frameworks between countries result in delays in product commercialization and increased development costs for MSC-based therapies, posing barriers to market growth.
Mesenchymal Stem Cells Market Segment Analysis:
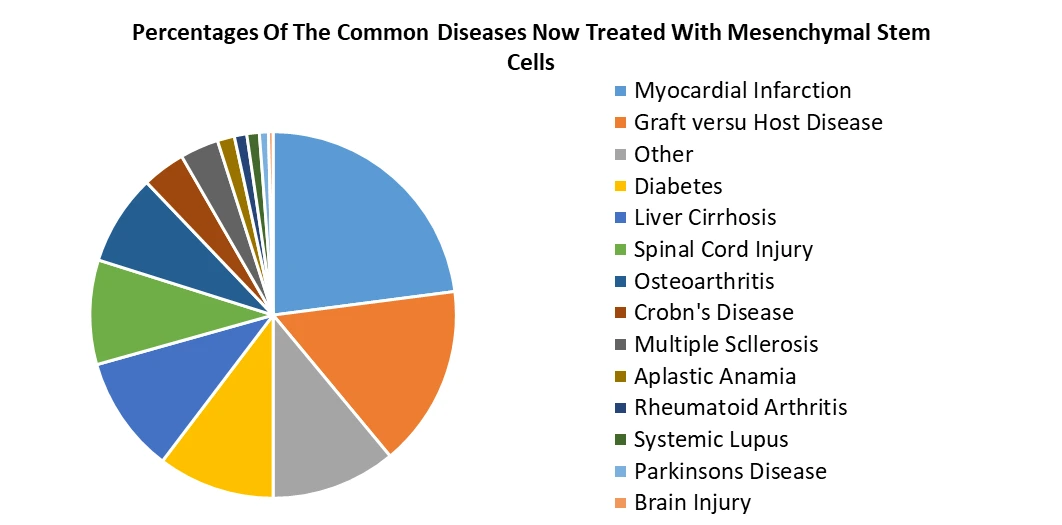
Based on Source of Isolation, Bone marrow-derived MSCs will dominate the market in 2023, owing to their long-standing use in clinical applications and established protocols for isolation. These cells find extensive applications in orthopedics, cardiology, and autoimmune disorders due to their regenerative potential and immunomodulatory properties. However, cord blood-derived MSCs are rapidly gaining prominence, driven by advancements in cord blood banking and the availability of a non-invasive and abundant cell source. Cord blood-derived MSCs hold promise in regenerative medicine, particularly in treating neurological disorders and hematological malignancies. Peripheral blood-derived MSCs, although less prevalent, are gaining traction in research and clinical trials due to their accessibility and minimally invasive isolation procedures. As research continues to elucidate the therapeutic potential of each MSC source, cord blood-derived MSCs are expected to witness increased adoption, potentially rivaling bone marrow-derived MSCs in certain applications, while peripheral blood-derived MSCs are anticipated to see greater utilization in niche therapeutic areas, such as wound healing and tissue engineering.
Mesenchymal Stem Cells Market Regional Insights:
North America dominates the Mesenchymal Stem Cells market and is expected to grow during the forecast period, with the region benefiting from advanced healthcare infrastructure, extensive research activities, and supportive regulatory frameworks. North America's dominance is further fueled by the presence of key market players, such as Thermo Fisher Scientific and Cytori Therapeutics Inc. Additionally, the region's focus on regenerative medicine and substantial investments in stem cell research contributes to its leading position. Asia Pacific is poised for significant growth in the forecast period, driven by increasing healthcare expenditures, rising adoption of advanced therapies, and expanding research initiatives. Countries like China and Japan are witnessing rapid growth in the MSCs market, propelled by supportive government initiatives and a large patient population. For instance, Asia Pacific's mesenchymal stem cells market is projected to grow at a rapid pace during the forecast period, owing to its large population base and increasing demand for regenerative therapies. The region's growing emphasis on personalized medicine and advancements in stem cell technologies further contribute to its burgeoning market. Additionally, collaborations between academic institutions and industry players drive innovation and market expansion in the Asia Pacific. While North America continues to dominate the MSCs market, Asia Pacific is expected to emerge as a key growth driver in the foreseeable future, presenting lucrative opportunities for market players and stakeholders.
Competitive Landscape
The recent advancements in cell therapy, such as BioCardia's CardiALLO trial, FDA's approval of Omisirge, and Cynata Therapeutics' partnership with LUMC, signify a significant push towards innovative treatments. These developments address critical medical needs, including ischemic heart failure, blood cancers, and post-kidney transplant complications. As these therapies demonstrate promising efficacy and safety profiles, they are poised to capture a substantial share of the growing regenerative medicine market. With increased acceptance and adoption by healthcare providers and patients alike, these breakthroughs are expected to drive substantial market growth, offering new hope for patients and bolstering investor confidence in the biotechnology sector.
On December 19, 2023, BioCardia initiated its CardiALLO™ Phase I/II trial, treating ischemic heart failure patients with BCDA-03 Allogeneic Mesenchymal Stem Cells. These "off-the-shelf" MSCs have immunomodulatory properties, potentially impacting heart failure's inflammatory processes and enhancing microvascular function. The trial, led by Dr. Carl Pepine and Dr. David Anderson, aims to improve outcomes for patients with reduced ejection fraction. BioCardia's VP of Clinical, Debby Holmes Higgin, highlights the therapy's potential for one-time treatment, offering immune modulation and paracrine signaling benefits to enhance microvascular function and improve patient outcomes.
On September 27, 2023, the FDA committee rejected NurOwn as an ALS therapy due to concerns about efficacy, manufacturing, and data consistency. BrainStorm Cell Therapeutics' stromal cell therapy, NurOwn, failed to demonstrate sufficient efficacy for mild to moderate ALS treatment. The committee vote of 17-1-1 highlighted doubts about product efficacy and manufacturing. FDA presentation revealed a lack of efficacy compared to the placebo in the phase 3 study, questioning subgroup analyses' reliability.
On April 17, 2023, FDA greenlights Omisirge (omidubicel-onlv), an allogeneic cord blood-based cell therapy, to accelerate neutrophil recovery and lower infection risk in adults and pediatric patients 12 years and older with blood cancers undergoing umbilical cord blood transplantation after myeloablative conditioning. Director Peter Marks hails the approval as a milestone in blood cancer treatment, emphasizing its potential to mitigate infection risks post-transplant, underscoring FDA's dedication to innovative cancer therapies.
On November 14, 2022, Cynata Therapeutics partners with Leiden University Medical Center (LUMC) to investigate mesenchymal stem cells' potential for reducing anti-rejection drug dependency post-kidney transplant. LUMC funds the Nereid Study, exploring the Safety and Efficacy of iPSC-derived Mesenchymal Stromal Cell Therapy in Renal Transplant Recipients. This collaboration aims to utilize Cynata's Cymerus™ MSCs to potentially mitigate renal graft rejection and decrease the need for anti-rejection drugs.
Mesenchymal Stem Cells Market Scope: Inquire before buying
| Global Mesenchymal Stem Cells Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2023 | Forecast Period: | 2024-2030 |
| Historical Data: | 2018 to 2023 | Market Size in 2023: | US $ 3.85 Bn. |
| Forecast Period 2024 to 2030 CAGR: | 12.6% | Market Size in 2030: | US $ 8.84 Bn. |
| Segments Covered: | by Workflow Type | Cell Sourcing & Isolation Culture & Cryopreservation Differentiation Characterization |
|
| by Type | Autologous Allogeneic |
||
| by Product Type | Cells & Cell Lines Kits, Media, & Reagents Others |
||
| by Indication | Bone And Cartilage Repair Cardiovascular Disease Inflammatory And Immunological Diseases Liver Diseases Cancer GvHD Others |
||
| by Source of Isolation | Bone Marrow Cord Blood Peripheral Blood Fallopian Tube Fetal Liver Lung Adipose Tissue |
||
| by Application | Disease Modelling Drug Development & Discovery Stem Cell Banking Tissue Engineering Toxicology Studies Others |
||
Mesenchymal Stem Cells Market, by Region
North America (United States, Canada and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan and Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest)
South America (Brazil, Argetina and Rest of South America)
Mesenchymal Stem Cells Market Key Players:
Key Players in North America:
1. Thermo Fisher Scientific, Inc. (United States)
2. Cytori Therapeutics Inc (United States)
3. STEMCELL Technologies Inc (Canada)
4. Celprogen Inc (United States)
5. BrainStorm Cell Therapeutics Inc. (United States)
6. Merck KGaA (MilliporeSigma) (United States)
Key Players in Europe:
7. Merck KGaA (MilliporeSigma) (Germany)
8. PromoCell GmbH (Germany)
9. Axol Biosciences Ltd. (United Kingdom)
Key Players in Asia Pacific:
10. Cyagen Biosciences Inc. (China)
11. Stemedica Cell Technologies Inc (Japan)
FAQs:
1. What are the growth drivers for the Mesenchymal Stem Cells Market?
Ans. Rising Prevalence of Chronic Diseases Drives MSC Therapy Demand is expected to be the major driver for the Mesenchymal Stem Cells Market.
2. What are the major Opportunities for the Mesenchymal Stem Cells Market growth?
Ans. Personalized Medicine Trend Spurs MSC Therapy Demand is the major opportunity for the Mesenchymal Stem Cells market.
3. Which country is expected to lead the global Mesenchymal Stem Cells Market during the forecast period?
Ans. Asia Pacific is expected to lead the Mesenchymal Stem Cells Market during the forecast period.
4. What is the projected market size and growth rate of the Mesenchymal Stem Cells Market?
Ans. The Mesenchymal Stem Cells Market size was valued at USD 3.85 Billion in 2023 and the total Mesenchymal Stem Cells Market revenue is expected to grow at a CAGR of 12.6 % from 2023 to 2030, reaching nearly USD 8.84 Billion.
5. What segments are covered in the Mesenchymal Stem Cells Market report?
Ans. The segments covered in the Mesenchymal Stem Cells Market report are by Workflow Type, Type, Grade, Product Type, Indication, Source of Isolation, Application, and Region.