Laboratory Proficiency Testing Market- Industry Structure Evaluation, Demand Drivers Analysis, Regional Growth Analysis and Identification, Competitive Positioning Review & Global Market Size Forecast to 2030
Overview
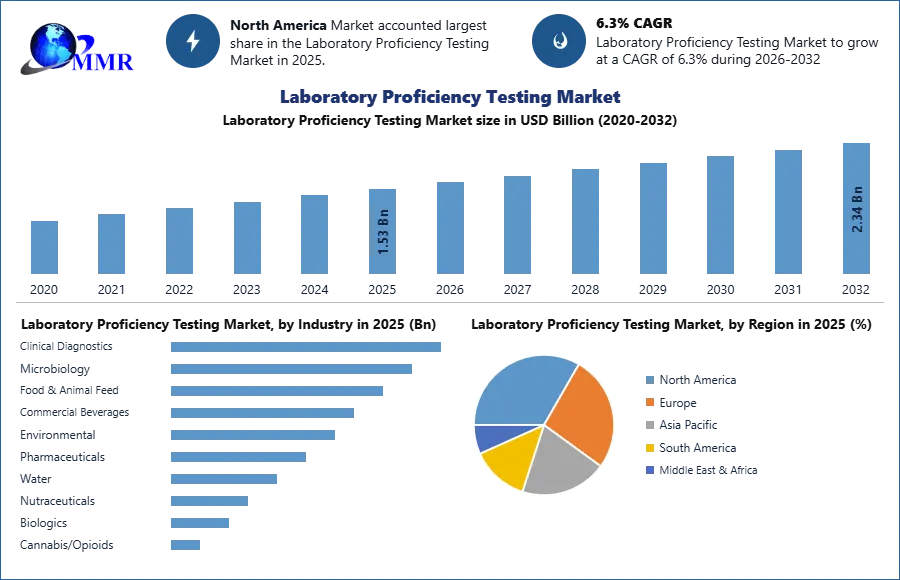
The Laboratory Proficiency Testing Market was valued at USD 1.53 Bn. in 2025. The Global Laboratory Proficiency Testing Market size is estimated to grow at a CAGR of 6.3% from 2026-2032, reaching USD 2.34 Bn by 2032.
Laboratory Proficiency Testing Market Overview:
The report explores the Laboratory Proficiency Testing market's segments (Industry and Technology). Data has been provided by market participants, and regions. This market report provides a thorough analysis of the rapid advances that are currently taking place across all industry sectors. Facts and figures, illustrations, and presentations are used to provide key data analysis for the historical period from 2020 to 2025. The report investigates the Laboratory Proficiency Testing market's drivers, limitations, prospects, and barriers. This MMR report includes investor recommendations based on a thorough examination of the Laboratory Proficiency Testing market's contemporary competitive scenario.
 To know about the Research Methodology:-Request Free Sample Report
To know about the Research Methodology:-Request Free Sample Report
Laboratory Proficiency Testing Market Dynamics:
Proficiency testing is an essential precondition for operational excellence
In some industries, reliable analytical trials are a vital prerequisite for cost-efficient production and quality control. Globally, several laboratories are targeting conducting PT programs to benchmark and demonstrate their technical capabilities. These PT programs prove the accuracy and reliability of a laboratory’s testing capabilities and can also be used to authenticate the entire testing process, including the competency of testing personnel. These programs can also help identify the areas of testing that are not performing as expected. This indicates subtle shifts and trends that, over time, would affect their analysis; reduce the cost of operations by addressing logistical issues that could render the measurements unusable or scientifically flawed; identify analytical issues with the sample matrix and analytes of interest; and aid in the identification of best practices and best laboratories for specific fields of work.
For this, numerous countries are signatories of international agreements that require PT. ILAC, APLAC, and NELAP are international organizations for accreditation bodies operating in agreement with ISO/IEC 17011 and involved in the authorization of conformity assessment bodies, including calibration laboratories (using ISO/IEC 17025), testing laboratories (using ISO/IEC 17025), medical testing laboratories (using ISO 15189), and inspection bodies (using ISO/IEC 17020). As PT is an essential precondition for operational excellence in various industries, the growth of the laboratory proficiency testing market is expected to grow in line with the growth of these industries.
Technological advancements in the testing industry led to the creation of many opportunities:
The growing focus on decreasing lead time, sample utilization, cost of testing, and drawbacks associated with several technologies have resulted in the development of new technologies, such as advanced spectrophotometry and chromatography techniques. The wide-scale implementation of these technologies provides an opportunity for medium- and small-scale laboratories to expand their service offerings and compete with large players in the industry, as these technologies offer higher sensitivity, accurate results, reliability, and multi-contaminant and non-targeted screening with low turnaround time, among other benefits.
This is favoring the growth of the laboratory proficiency testing market as major players are offering new, fast, and more precise technologies, such as LC (liquid chromatography), HPLC (high-performance liquid chromatography), and ICP-MS (inductively coupled plasma mass spectrometry) for testing the safety and quality of products across diverse industry verticals. These are some of the factors that are leading to the growth of the opportunities in the Laboratory Proficiency Testing Market through the forecast period.
The requirement of high-capital investments for accurate and sensitive testing is limiting the growth:
With the rise in the use of proficiency testing by authorization bodies as a tool to ensure the credibility of laboratories participating in accreditation programs, the cost pressure on the participating laboratories is rising. The time and resources compulsory for proficiency testing are generally associated with the overhead costs of operating a laboratory. Investments need to be made at many stages of PT programs, such as while preparing the program of study, while running various samples, handling and shipping the samples, and during the interpretation and reporting phase. In the case of poor results, high costs are involved in repeated measurements to correct the poor data. These tests are not cheap, and when staff time is figured in, proficiency testing can become an expensive burden for small laboratories. Also, the need for extensive sample preparation requires laboratory analysts to use advanced testing technologies.
Advanced technologies, such as liquid chromatography (LC), high-performance liquid chromatography (HPLC), and spectrometry, are sensitive, accurate, and efficient. However, these technologies have disadvantages such as high costs, extensive sample preparation time, and calibration issues, which add to the investments required to conduct proficiency testing. Additionally, most analytical methods are suitable only for certain types of samples, while more innovative methods are required for the testing of samples such as pharmaceuticals and opioids. Though, the inclusion of these advanced methods increases the overall cost of testing, which in turn results in higher capital investments for laboratories.
The need for proficiency testing scheme harmonization is a major challenge:
The main driver of proficiency testing is to compare or evaluate the harmonization of laboratories involved in a common field of testing. A precise and valuable comparison to other laboratories is an expected and critical outcome, whether as an element of the laboratory authorization process or as part of an internal quality assurance program. Still, proficiency testing schemes (PTS) are not universally coordinated. For occurrence, schemes developed for clinical laboratories differ from those developed for food testing laboratories. Similarly, environmental PTS varies from those used for industrial hygiene testing. Moreover, even within an industry, such as environmental proficiency testing, there are regional, national, and international differences that culminate in a lack of organization.
The lack of constancy between different PTS is negatively affecting the ability of laboratories to achieve their expected goals. This can be attributed to the technical dissimilarity among the markets and the analytical procedures being assessed. Also, certain financial and regulatory impacts to drive programmatic differences may affect the entire program. Thus, an understanding of the reasons for the differences in PTS and exploring opportunities for increasing harmonization between schemes are critical for achieving the desired results.
Laboratory Proficiency Testing Market Trends:
Laboratory Proficiency Testing Market Segment Analysis:
Based on industry, the Laboratory Proficiency Testing Market was segmented into Clinical Diagnostics, Microbiology, Food & Animal Feed, Commercial Beverages, Environmental, Pharmaceuticals, Water, Nutraceuticals, Biologics, Cannabis/Opioids, Dietary Supplements, and Cosmetics. Among these, the clinical diagnostics segment dominated the market in 2025 due to the increasing development of complex diagnostic tests and the growing need for accurate and early disease detection. Clinical laboratories increasingly relied on proficiency testing programs to maintain quality assurance, validate laboratory performance, and comply with regulatory standards. The rising demand for advanced diagnostic testing and strict quality control requirements across healthcare laboratories further supported the dominance of this segment.
Based on technology, the market was segmented into Cell Culture, PCR, Immunoassays, Chromatography, Spectrophotometry, and Others. Among these, cell culture technology dominated the market in 2025 as it plays a critical role in the testing and validation of clinical diagnostic samples, microbiological analysis, and biopharmaceutical development. The growing adoption of cell culture methods in disease research, vaccine development, and laboratory testing supported the expansion of this segment.
Based on end-user, the market was segmented into Hospitals and Clinical Laboratories, Pharmaceutical and Biotechnology Companies, Food and Beverage Testing Laboratories, Environmental Testing Laboratories, Academic and Research Institutions, and Others. Among these, hospitals and clinical laboratories dominated the market in 2025 due to the increasing volume of diagnostic testing and the need for standardized laboratory performance evaluation. The use of proficiency testing programs in these facilities ensures accuracy, reliability, and regulatory compliance in clinical testing procedures.
Laboratory Proficiency Testing Market Regional Insights:
The objective of the report is to present a comprehensive analysis of the global Laboratory Proficiency Testing Market to the stakeholders in the industry. The past and current status of the industry with the forecasted market size and trends are presented in the report with the analysis of complicated data in simple language. The report covers all the aspects of the industry with a dedicated study of key players that include market leaders, followers, and new entrants.
PORTER, PESTEL analysis with the potential impact of micro-economic factors of the market have been presented in the report. External as well as internal factors that are supposed to affect the business positively or negatively have been analyzed, which will give a clear, futuristic view of the industry to the decision-makers.
The reports also help in understanding the Laboratory Proficiency Testing Market dynamic and structure by analyzing the market segments and projecting the Laboratory Proficiency Testing Market size. Clear representation of competitive analysis of key players by Design, price, financial position, product portfolio, growth strategies, and regional presence in the Laboratory Proficiency Testing Market make the report investor’s guide.
Laboratory Proficiency Testing Industry Ecosystem
Laboratory Proficiency Testing Market Recent Developments (2025–2026):
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 26 February 2026 | LGC Group | Acquired ChromaDex’s Reference Standards business to enhance its global portfolio of high-purity materials for pharmaceutical and food testing. | This acquisition strengthens LGC’s ability to support laboratories with ISO-accredited standards, bolstering its position in the proficiency testing supply chain. |
| 02 February 2026 | Randox Laboratories | Received FDA 510(k) clearance for its newest clinical drug testing panel, designed for use on automated diagnostic platforms. | The approval facilitates expanded toxicology screening and necessitates the rollout of corresponding proficiency testing schemes to ensure multi-site diagnostic accuracy. |
| 19 November 2025 | Bio-Rad Laboratories | Launched a specialized Proficiency Testing program for Autoantibodies to address diagnostic variability in autoimmune disease detection. | The program provides clinical labs with external quality assessment (EQA) tools to standardize complex serology testing and improve patient diagnostic outcomes. |
Laboratory Proficiency Testing Market Scope: Inquire before buying
| Laboratory Proficiency Testing Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 1.53 USD Billion |
| Forecast Period 2026-2032 CAGR: | 6.3% | Market Size in 2032: | 2.34 USD Billion |
| Segments Covered: | by Industry | Clinical Diagnostics Microbiology Food & Animal Feed Commercial Beverages Environmental Pharmaceuticals Water Nutraceuticals Biologics Cannabis/Opioids Dietary Supplements Cosmetics |
|
| by Technology | Cell Culture PCR Immunoassays Chromatography Spectrophotometry Others |
||
| by End-User | Hospitals and Clinical Laboratories Pharmaceutical and Biotechnology Companies Food and Beverage Testing Laboratories Environmental Testing Laboratories Academic and Research Institutions Other |
||
Laboratory Proficiency Testing Market, by Region:
North America (United States, Canada and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan and Rest of APAC)
South America (Brazil, Argentina, and rest of South America)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A)
Laboratory Proficiency Testing Market, Key Players:
- LGC Limited
- Bio-Rad Laboratories, Inc.
- College of American Pathologists (CAP)
- Merck KGaA
- American Proficiency Institute
- Randox Laboratories Ltd.
- Waters Corporation
- Fera Science Limited (Fapas)
- AOAC INTERNATIONAL
- QACS International
- Intertek Group plc
- Eurofins Scientific
- SGS SA
- Bureau Veritas SA
- Trilogy Analytical Laboratory
- Bipea
- Phenova Inc.
- NSI Lab Solutions
- Absolute Standards Inc.
- Advanced Analytical Solutions, LLC
- Weqas
- SPEX CertiPrep
- Global Proficiency Ltd.
- Envirocare Labs
- Matrix Sciences
