Global Early Toxicity Testing Market by Technique (In vivo, In silico, In vitro), End User (Pharmaceuticals industry, Cosmetics industry, Chemicals industry, Food industry, Others) - Forecast to 2032
Overview
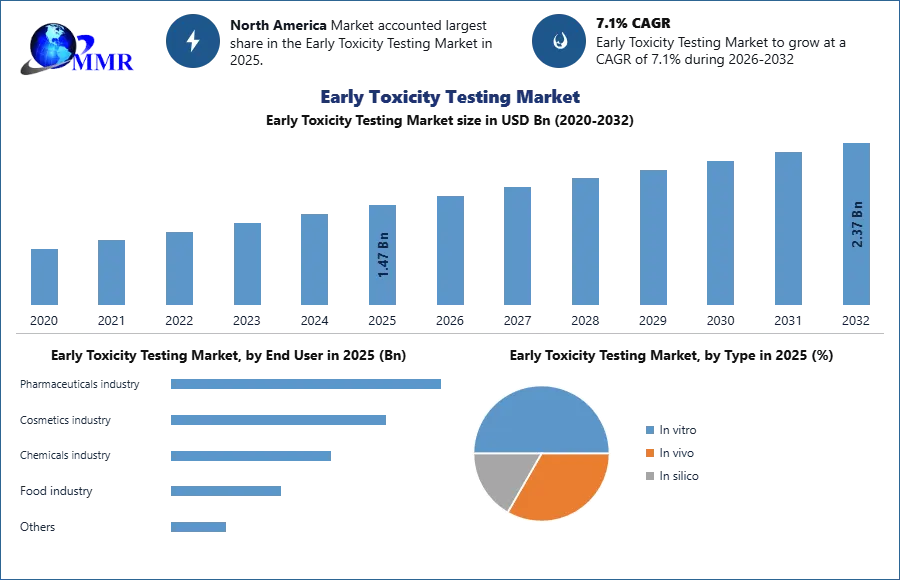
Early Toxicity Testing Market is expected to reach USD 2.37 Bn. at a CAGR of 7.1% during the forecast period 2032.
Toxic testing procedures provide more relevant and mechanistic insight into the toxic potential of a chemical the compound, which is introduced the first time on the subject.
Early Toxicity Testing Market Snapshot
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
Early toxicity testing is an important procedure, carried out during all stages of clinical trial phases, to determine Absorption Distribution Metabolism Excretion Toxicity (ADMET) properties of the drug molecule. Toxicity testing help clinicians to know about pharmacokinetic and pharmacodynamics of drug molecule, hence it helps to judge whether the drug is beneficial therapeutic or not for human beings.
The report covers detailed competitive landscape including company profiles of key players operating in the global market. Some key players in the early toxicity testing market include Covance, Thermo Fisher Scientific, Quest Diagnostics Corporation, Agilent Technologies. An in-depth view of the competitive outlook includes future capacities, key mergers & acquisitions, financial overview, partnerships, collaborations, new product launches, new product developments and other developments with information in terms of Drivers, Restrain, Opportunities and challenges.
Early Toxicity Testing Market Dynamics
Technological advancements and rising adoption of both in-vivo & in vitro techniques, driving the global market during the forecast period. Growing research & development activities across the healthcare sector and strict government regulations create the need for toxicity testing for newly invented therapeutic drugs. Advance technology adoption in toxicity testing carried out at newly developed and developing clinical research organizations, drive the global early toxicity testing market.
Funds released by governments to support research in the new drug Discovery and Development (NDDD) department is expected to favor the growth of this market. As the overall procedure of toxicity testing studies may not be able to give positive results every time, hence the success rate for new drug approvals remains low due to failures of efficacy and safety. This hampers the growth of the global toxicity testing market.However, certain limitations associated with preclinical testing is restraining the global market.
Early Toxicity Testing Market Segmentation
By Type: The in vitro segment dominated the early toxicity testing market in 2025 due to its cost-effectiveness, faster results, and reduced ethical concerns compared to in vivo testing. Growing adoption across pharmaceutical, chemical, and cosmetic industries is further driving its demand, while in vivo and in silico methods hold smaller but steady shares.
Early Toxicity Testing Market Regional Analysis:
North America dominated the early toxicity testing market in 2025 driven by the presence of leading pharmaceutical and biotechnology companies, stringent regulatory requirements, and advanced laboratory infrastructure. The region’s focus on drug safety, growing R&D investments, and increasing adoption of in vitro and in silico testing methods are fueling market growth, making it the largest contributor to global revenue.
The objective of the report is to present a comprehensive analysis of the Global Healthcare Associated Infections Treatment Market including all the stakeholders of the industry. The past and current status of the industry with forecasted market size and trends are presented in the report with the analysis of complicated data in simple language.
The objective of the report is to present a comprehensive analysis of the Global Healthcare Associated Infections Treatment Market including all the stakeholders of the industry. The past and current status of the industry with forecasted market size and trends are presented in the report with the analysis of complicated data in simple language.
The report covers all the aspects of the industry with a dedicated study of key players that includes market leaders, followers and new entrants by region. PORTER, SVOR, PESTEL analysis with the potential impact of micro-economic factors by region on the market has been presented in the report. External as well as internal factors that are supposed to affect the business positively or negatively have been analysed, which will give a clear futuristic view of the industry to the decision-makers. The report also helps in understanding Global Laryngeal Mirrors Market dynamics, structure by analysing the market segments and projects the Global Laryngeal Mirrors Market size.
Recent Industry Developments (2025–2026)
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 28 January 2025 | MIMETAS | The company launched OrganoPlate UniFlow (UF), the first commercial pumpless flow system for drug discovery, at SLAS 2025. | The system supports up to 512 chips to replicate blood circulation in liver and kidney models, enhancing early toxicity screening accuracy. |
| 04 February 2025 | Xellar Biosystems | The company entered a sponsored research collaboration with Sanofi to develop Organ-on-a-Chip (OOC) models. | The partnership focuses on vascular toxicity assessment, aiming to identify potential safety issues in the early preclinical phase. |
| 06 March 2025 | Insilico Medicine | The AI-designed drug Rentosertib achieved Phase IIa validation, meeting primary endpoints for safety and tolerability. | The successful safety profiling via the Pharma.AI platform demonstrates the power of generative AI in predicting early-stage drug toxicity. |
| 03 June 2025 | Evotec | The company joined the NURTuRE-AKI consortium to utilize multi-omics for understanding acute kidney injury. | This collaboration enhances nephrotoxicity testing capabilities by identifying molecular biomarkers for early safety assessment. |
| 01 October 2025 | CN Bio | The company launched PhysioMimix Core, an all-in-one multi-organ-on-chip system for high-throughput testing. | The system enables validated performance across complex physiological models, reducing reliance on animal testing for ADME-Tox studies. |
| 18 February 2026 | Charles River Laboratories | The company filed its annual report detailing a strategy to prioritize New Approach Methodologies (NAMs) and digital tools. | By integrating AI and in vitro models, the firm aims to optimize its safety assessment portfolio to meet rising demand for non-animal testing. |
Scope of the Global Early Toxicity Testing Market: Inquire before buying
| Early Toxicity Testing Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 1.47 USD Bn |
| Forecast Period 2026-2032 CAGR: | 7.1% | Market Size in 2032: | 2.37 USD Bn |
| Segments Covered: | by Type | In vitro In vivo In silico |
|
| by Product | Instruments Kits Consumables |
||
| by End User | Pharmaceuticals industry Cosmetics industry Chemicals industry Food industry Others |
||
Early Toxicity Testing Market, by Region
North America (United States, Canada and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria, Turkey, Russia and Rest of Europe)
Asia Pacific (China, India, Japan, South Korea, Australia, ASEAN and Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A)
South America (Brazil, Argentina, Columbia and Rest of South America)
Key players/competitors profile covered in Early Toxicity Testing Market report in strategic perspective
Covance
Thermo Fisher Scientific
Quest Diagnostics Corporation
Agilent Technologies
Sigma Aldrich
BD bioscience
Bio-rad
Gene Logic
Accelrys
Charles River Laboratories
Ocimum Biosolutions
Eurofins Scientific
Merck KGaA
Evotec AG
Enzo Biochem Inc.
Bioanalytical Systems
General Electric Company
Bruker Corporation
The Jackson Laboratory
Invitrogen
Danaher Corporation
IDBS and PerkinElmer Inc.
