Diagnostic Catheter Market Size by Product Type, Application, End-User, Region – Revenue Pool Analysis, Margin Structure Assessment, Capital Flow Trends, Competitive Benchmarking & Forecast to 2030
Overview
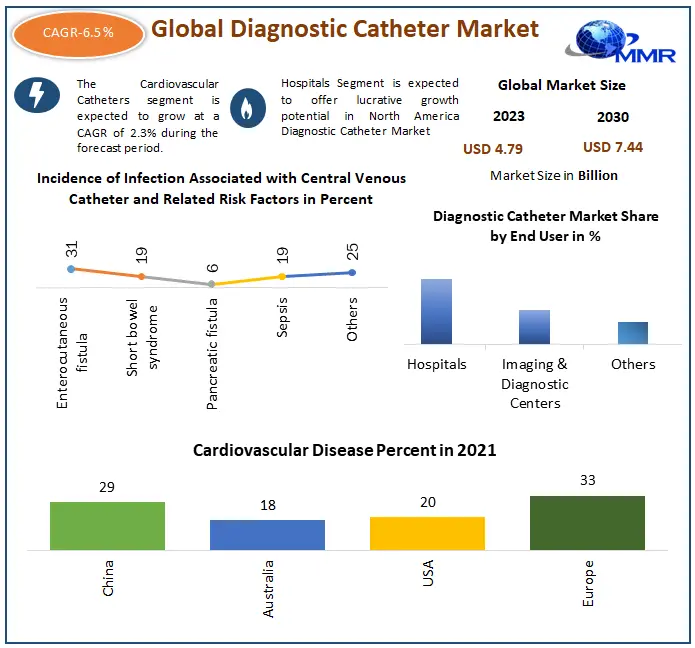
The Diagnostic Catheter market size is USD 4.79 Billion in 2023 to USD 7.44 Billion by 2030; it is estimated to grow at a CAGR of 6.5% from 2024 to 2030
Diagnostic Catheter Market Overview
A diagnostic catheter is a medical device used in diagnostic procedures to access various parts of the body for visualization, sampling, and measurement purposes. It typically consists of a thin, flexible tube made of materials such as plastic or rubber, with specialized features such as imaging capabilities, sensors, or channels for fluid delivery. The diagnostic catheter market covers various industrial applications, including cardiology, urology, gastroenterology, neurology, and peripheral vascular procedures. In cardiology, diagnostic catheters are extensively used for procedures such as angiography, electrophysiology studies, and intravascular ultrasound to diagnose and treat cardiovascular conditions. Similarly, in urology, catheters aid in the diagnosis and management of urinary tract disorders, such as kidney stones or urinary incontinence. Gastroenterology utilizes catheters for endoscopic procedures like endoscopic retrograde cholangiopancreatography (ERCP) or colonoscopy. Neurology procedures, such as cerebral angiography and neurointerventional radiology, rely on catheters for diagnosing and treating neurological disorders like strokes or aneurysms. In peripheral vascular procedures, diagnostic catheters are crucial in assessing blood flow and diagnosing peripheral artery disease.
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
The diagnostic catheter market scenario is characterized by significant growth driven by technological advancements, increasing prevalence of chronic diseases, and rising demand for minimally invasive procedures. Key players such as Boston Scientific Corporation, Medtronic plc, and Johnson & Johnson dominate the market, leveraging their innovative product portfolios and extensive distribution networks. For example, Boston Scientific's "OptiCross" diagnostic catheter offers enhanced maneuverability and visualization for precise lesion assessment during coronary angiography. Medtronic's "Launcher" coronary guiding catheter facilitates smooth navigation through complex coronary anatomy, enhancing procedural efficiency. Johnson & Johnson's subsidiary Cordis Corporation provides a End-User of diagnostic catheters for cardiology applications, including the "Cordis Emerge" diagnostic catheter market, known for its exceptional torque control and tip flexibility. These examples highlight how key players continually innovate to meet the evolving demands of healthcare professionals and improve patient outcomes in diagnostic procedures. For instance, January 9, 2024, Cook Medical introduces Slip-Cath Beacon Tip Hydrophilic Selective Catheter in the U.S. and Canada, catering to vascular and non-vascular procedures. Developed in collaboration with physicians, it offers enhanced features like visibility, usability, and torqueability. Packaged in protective foil for durability, it boasts a three-year shelf life, surpassing competitors. Designed for angiographic procedures alongside vascular access sheaths and wire guides, Slip-Cath complements Cook's extensive vascular product line. As part of its commitment to healthcare providers, Cook Medical continues to innovate, providing expanded treatment options and ultimately improving patient outcomes.
Diagnostic Catheter Market Dynamics:
Technological Advancements is Driving the Diagnostic Catheter Market
Technological advancements play a pivotal role in expanding market demand. Innovations such as high-definition mapping catheters, exemplified by the OPTRELL™ Mapping Catheter with TRUEref™ Technology, offer enhanced accuracy and efficiency in treating complex cardiac arrhythmias. Moreover, the aging population contributes significantly to the market's expansion, as elderly individuals are more susceptible to cardiovascular and vascular diseases that necessitate diagnostic interventions. With conditions like atrial fibrillation affecting millions globally, there is an increasing need for advanced diagnostic tools to address the growing prevalence of chronic diseases. Additionally, rising awareness of the importance of early disease detection and screening programs further fuels market growth, encouraging the adoption of diagnostic catheters for timely interventions.
Favorable reimbursement policies catalyze the Diagnostic Catheter Market trend, incentivizing healthcare providers to invest in advanced catheter technologies. Collaborations between medical device companies and healthcare institutions drive innovation and Diagnostic Catheter Market demand by leveraging shared expertise and resources. Additionally, the increasing healthcare expenditure, particularly in developed countries, supports the adoption of advanced diagnostic technologies like high-definition mapping catheters. As the demand for personalized medicine and targeted therapies continues to grow, there is an escalating need for precise diagnostic tools such as high-resolution mapping catheters to tailor treatments to individual patient needs effectively. These factors collectively contribute to the dynamic growth of the diagnostic catheter market, promising continued advancements in cardiovascular and vascular healthcare delivery.
On July 24, 2023, Biosense Webster, part of Johnson & Johnson MedTech, launched the OPTRELL Mapping Catheter in the U.S., powered by CARTO 3 System. It offers high-definition mapping for complex cardiac arrhythmias like AFib, affecting 40 million worldwide. With 48 electrodes, tight spacing, and small size, it provides superior signal resolution. This aids in the precise localization of arrhythmias, crucial for effective ablation therapy. AFib, if untreated, can lead to severe complications like heart disease and stroke. The catheter's advanced technology enhances clinicians' ability to map and treat abnormal heart rhythms, potentially improving outcomes for millions of patients.
High Cost of Development and Maintenance Hamper the Diagnostic Catheter Market Growth
The stringent regulatory requirements serve as a significant barrier to market entry and growth. Compliance with regulatory standards, such as FDA clearance in the United States, demands extensive clinical trials and meticulous documentation, resulting in prolonged approval processes and increased costs for manufacturers. These hurdles often dissuade smaller companies from entering the market or developing innovative catheter technologies, stifling competition and limiting Diagnostic Catheter Market diversity. Moreover, the high cost of development and maintenance further exacerbates accessibility issues, as research, manufacturing, and infrastructure investments incur substantial financial burdens. Specialized catheters equipped with advanced imaging capabilities, such as intravascular ultrasound (IVUS) or optical coherence tomography (OCT), require sophisticated technology and expertise, driving up production costs and pricing out smaller players from the market.
The technological challenges and innovation barriers present ongoing obstacles to market growth and evolution. The rapid pace of technological advancement necessitates continuous research and development efforts to keep pace with industry standards and consumer demands. Integrating advanced imaging modalities into diagnostic catheters, such as IVUS or OCT, demands substantial R&D investment and expertise, deterring smaller manufacturers from competing with established Diagnostic Catheter Market leaders. Additionally, competition from alternative diagnostic modalities like MRI and CT scans, which offer non-invasive imaging solutions, further complicates market dynamics. Patients may prefer non-invasive procedures due to concerns about discomfort and procedural risks associated with catheterization, diverting demand away from diagnostic catheters. To address these challenges, stakeholders must prioritize innovation, streamline regulatory processes, and invest in workforce training to foster Diagnostic Catheter Market growth and meet the evolving needs of healthcare providers and patients.
Intravascular Ultrasound (IVUS) and Optical Coherence Tomography (OCT) Transforming Coronary Imaging Boost Diagnostic Catheter Market
The rising prevalence of cardiovascular diseases worldwide underscores the need for advanced diagnostic interventions, propelling the demand for diagnostic catheters. With cardiovascular disorders affecting millions globally and accounting for a significant portion of healthcare expenditures, diagnostic catheters play a crucial role in accurately diagnosing and managing these conditions. Moreover, technological advancements in catheter design and functionality, such as improved imaging capabilities and navigational tools, further enhance the diagnostic accuracy and efficacy of catheter-based procedures. Innovations like intravascular ultrasound (IVUS) and optical coherence tomography (OCT) revolutionize coronary imaging, enabling precise visualization of vessel structures and pathology, thus driving the adoption of diagnostic catheters across various medical specialties, including cardiology and radiology.
The growing preference for minimally invasive procedures and the expanding geriatric population contribute to the Diagnostic Catheter Market growth. As patients seek less invasive alternatives to traditional diagnostic methods, the demand for catheter-based procedures continues to rise. Minimally invasive techniques offer benefits such as shorter recovery times, reduced risk of complications, and improved patient outcomes, driving the adoption of diagnostic catheters across medical specialties. Additionally, the aging population worldwide is prone to age-related cardiovascular conditions, necessitating frequent diagnostic evaluations. As the number of elderly individuals increases globally, the demand for diagnostic catheters is expected to surge, providing significant growth opportunities for manufacturers and suppliers in the market.
On February 8, 2024, BIOTRONIK introduced the Micro Rx™ catheter, a new rapid exchange microcatheter designed to enhance guidewire support during percutaneous coronary interventions (PCI) with ease. Exclusively distributed by BIOTRONIK and manufactured by IMDS (Interventional Medical Device Solutions), this cutting-edge device joins BIOTRONIK's portfolio alongside NHancer Rx, TrapIT, and ReCross catheters. The Micro Rx catheter boasts a reinforced distal shaft with a core wire between two layers of braids for optimal push transfer, along with a tapered tip featuring the market's smallest leading edge for enhanced penetration force. Additionally, it includes a Proximal-End-Stop that seamlessly integrates with a guide extension catheter for added support. The Micro Rx catheter is now available in the United States and will launch in Canada in March 2024.
Diagnostic Catheter Market Segment Analysis:
Based on Product Type, the market is divided into Cardiovascular Catheters, Neurovascular Catheters, Urological Catheters, Specialty Catheters, Intravenous Catheters, and Other Types. Among these Cardiovascular Catheters sub-segment witnessed the highest market share in 2023. Cardiac catheterization, known as a crucial invasive imaging procedure in the medical industry, serves as a pivotal tool for assessing heart function. By inserting a catheter, a minute tube, into a blood vessel located in either the arm or groin and subsequently navigating it into the coronary arteries, healthcare providers can conduct comprehensive evaluations of cardiac health. This procedure is instrumental in identifying various cardiac abnormalities and conditions, facilitating prompt diagnosis and treatment planning. Moreover, cardiac catheterization plays a vital role in the advancement of minimally invasive interventions within the medical industry. One of the primary significance lies in its ability to diagnose and address congenital heart defects effectively. By leveraging cardiac catheterization, healthcare providers can precisely locate and evaluate structural abnormalities in the heart, allowing for timely interventions to correct these defects. Furthermore, the procedure enables healthcare professionals to perform corrective measures, such as repairing heart flaws or replacing malfunctioning heart valves, without resorting to extensive surgical procedures. This aspect not only reduces the risks associated with traditional surgery but also enhances patient outcomes by minimizing recovery times and postoperative complications.
Cardiac catheterization contributes significantly to the evolution of minimally invasive techniques in cardiovascular interventions. By utilizing catheters to access the heart and its surrounding vessels, healthcare providers can perform a wide End-User of therapeutic procedures with remarkable precision and efficiency. These procedures include but are not limited to angioplasty, stent placement, and transcatheter valve replacement, all of which are instrumental in managing various cardiovascular conditions. The advent of minimally invasive interventions not only improves patient comfort and recovery but also reduces healthcare costs and hospitalization durations, thus revolutionizing the treatment landscape in the cardiology sector which drives the Diagnostic Catheter Market.
Competitive Landscape
On February 23, 2022, Teleflex extended FDA clearance to expand the use of its specialty catheters and coronary guidewires for chronic total occlusion percutaneous coronary interventions (CTO PCI). CTOs, which affect around 20% of patients undergoing diagnostic coronary angiography, pose significant health risks and limit symptoms. The clearance covers devices like the GuideLiner V3 Catheter and Spectre Guidewire. Results from the CTO-PCI study, involving 150 patients, demonstrate high technical (93.3%) and procedural (over 75% free of major adverse cardiovascular events) success rates. Over 57,000 CTO interventions are performed annually in the US.
Table: Diagnostics Catheter Key Players
| Company | Description |
| Boston Scientific Corporation | Produces a wide End-User of medical devices, including diagnostic catheters for various medical procedures. |
| Medtronic | A major player in the medical device industry, offering diagnostic catheters among its extensive portfolio of medical devices and technologies. |
| Abbott Laboratories | A global healthcare company that produces various medical devices, including diagnostic catheters used in cardiovascular procedures. |
| Johnson & Johnson | Develops and manufactures diagnostic catheters for cardiac and vascular interventions through its subsidiary Cordis. |
| Terumo Corporation | A Japanese multinational medical devices and equipment company known for its diagnostic catheters used in cardiology and radiology. |
| B. Braun Melsungen AG | A German medical and pharmaceutical company that manufactures diagnostic catheters along with a wide End-User of medical products. |
| Merit Medical Systems, Inc. | A leading manufacturer and distributor of medical devices, including diagnostic and interventional catheters. |
| Cook Medical | A family-owned medical device company that produces diagnostic catheters for various medical specialties, including cardiology and radiology. |
Diagnostic Catheter Market Scope: Inquiry Before Buying
| Diagnostic Catheter Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2023 | Forecast Period: | 2024-2030 |
| Historical Data: | 2018 to 2023 | Market Size in 2023: | US $ 4.79 Bn |
| Forecast Period 2024 to 2030 CAGR: | 6.5 % | Market Size in 2030: | US $ 7.44 Bn. |
| Segments Covered: | by Product Type | Cardiovascular Catheters Neurovascular Catheters Urological Catheters Specialty Catheters Intravenous Catheters Other Types |
|
| by Application | Gastroenterology Cardiology Urology Neurology Others |
||
| by End-User | Hospitals Imaging & Diagnostic Centers |
||
Diagnostic Catheter Market, by Region
North America (United States, Canada and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan and Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A)
South America (Brazil, Argentina Rest of South America)
Diagnostic Catheter Market Key Players:
Major Contributors in the Diagnostic Catheter Market in North America:
1. Boston Scientific Corporation (Massachusetts, USA)
2. Johnson & Johnson (New Jersey, USA)
3. Abbott Laboratories (Illinois, USA)
4. Becton, Dickinson and Company (New Jersey, USA)
5. Cardinal Health, Inc.(Ohio, USA)
6. Edwards Lifesciences Corporation (California, USA)
7. Cook Medical LLC (Indiana, USA)
8. Teleflex Incorporated (Pennsylvania, USA)
9. Merit Medical Systems, Inc. (Utah, USA)
10. AngioDynamics, Inc. (New York, USA)
11. Integer Holdings Corporation (Texas, USA)
12. Smiths Medical (Minnesota, USA)
13. Penumbra, Inc. (California, USA)
Major Leading Player in the Diagnostic Catheter Market in Europe:
1. BTG International Ltd. (London, United Kingdom)
2. Medtronic plc (Dublin, Ireland)
3. B. Braun Melsungen AG (Melsungen, Germany)
4. Siemens Healthineers AG (Erlangen, Germany)
Major Prominent Players in the Diagnostic Catheter Market in Asia Pacific:
1. Asahi Intecc Co., Ltd. (Nagoya, Japan)
2. Terumo Corporation (Tokyo, Japan)
3. Olympus Corporation (Tokyo, Japan)
FAQ:
1] What segments are covered in the Global Diagnostic Catheter Market report?
Ans. The segments covered in the Diagnostic Catheter Market report are based on Product Type, Application, End-User, and Region.
2] Which region is expected to hold the highest share in the Global Diagnostic Catheter Market?
Ans. North America region is expected to hold the highest market share in the Diagnostic Catheter market.
3] What is the market size of the Global Diagnostic Catheter Market by 2030?
Ans. The market size of the Diagnostic Catheter Market by 2030 is expected to reach USD 7.44 Billion.
4] What is the forecast period for the Global Diagnostic Catheter Market?
Ans. The forecast period for the Diagnostic Catheter Market is 2024-2030.
5] What was the market size of the Global Diagnostic Catheter Market in 2023?
Ans. The market size of the Diagnostic Catheter Market in 2023 was valued at USD 4.79 Billion.