Conjugate Vaccine Market Size – Industry Structure Evaluation, Demand Drivers Analysis, Regional Growth Analysis and Identification, Competitive Positioning Review & Global Market Size Forecast to 2032
Overview
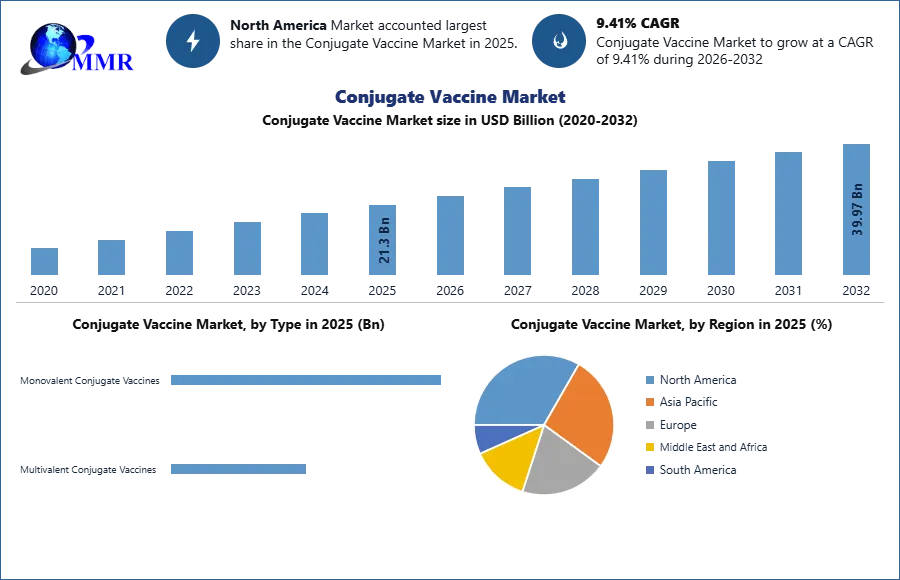
Conjugate Vaccine Market is expected to reach USD 39.97 Bn. at a CAGR of 9.41% during the forecast period 2032.
To know about the Research Methodology :- Request Free Sample Report
Conjugate Vaccine Market Definition:
A conjugate vaccine is composed of polysaccharide antigen conjugated with a protein carrier to enhance immunogenicity and the effectiveness of the vaccine. For example, Pneumococcal conjugate vaccine used to immunize young children and infants against disease like a pneumococcal caused by Streptococcus pneumonia.
The report study has analyzed revenue impact of covid-19 pandemic on the sales revenue of market leaders, market followers and disrupters in the report and same is reflected in our analysis.
Conjugate Vaccine Market Dynamics:
The report comprises of worldwide Conjugate Vaccine Market presentation in terms of revenue from various segments and detailed evaluation of the trends and factors that are playing a substantial role in the market. These factors involve the drivers, restraints, opportunities and challenges for the key companies and stakeholders in the industry.
The global conjugate vaccine market is flourishing because of increasing cases of infectious diseases such as pneumococcal, typhoid and others with the growing demand of meningococcal vaccines and pneumococcal vaccines. For instance, as per the WHO Community-acquired pneumonia (CAP) is a major respiratory disease inclining due to variable severity and clinical heterogeneity which results in frequent death due to infection, all-around almost one million children worldwide die from pneumonia annually which led to an increase in demand for safe and effective vaccines for the relief and death prevention. As per the U.S., the highest mortality cases informed were due to vaccine-preventable diseases.
However, Conjugate Vaccine also have some limitations. Like, people in rural or Distant Areas have Low Accessibility of Vaccines because of lack of transportation and storage problems anticipated to hamper and restrain the market growth during the forecast period.
In addition, growing R&D activities for the development of innovative products and advancement in technology. Further, adoption of variant strategic business activities such as acquisitions, mergers, etc. are estimated to create productive opportunities for the global market over the forecast period.
Conjugate Vaccine Market Market Segment Analysis:
The report covers competitive analysis of the Conjugate Vaccine Market in each of the geographical segments thereby providing insight into a market share at the macro as well as micro levels.
Based on product type, the multivalent conjugate vaccine segment is estimated to have prominent growth for global conjugate vaccine market, resulting in increasing clinical trials for the development of multivalent conjugate vaccines. For example, on March 2019, Initiation by Pfizer for the three Phase III clinical trials to evaluate its 20-valent pneumococcal conjugate vaccine (20vPnC) to deliberate the safety and immunogenicity of 20vPnC vaccine in a candidate at and above 18 years and older.
In terms of age group, Pedriatic vaccines are estimated to have the highest share in market prominently because of the not fully developed immune system in children and increasing pediatric vaccination dose. As per WHO, in 2018 around 86% of infants worldwide received 3 doses of DTP-3 that protect against diphtheria-tetanus-pertussis. Furthermore, initiatives by government, like Vaccination Program for Children and vaccination provides to children or who lack health insurance and cannot afford the cost of vaccination, which is approved by the Office of Management & Budget, further funded by the Medicaid Services (CMS) to the Centers for Disease Control (CDC).
Conjugate Vaccine Market Regional Insights:
North America’s Conjugate Vaccine Market is estimated to expand at a value of US$ 385.26 Mn by 2032, with a CAGR of 3.2%. North America is the largest market and U.S is accounted to have major revenue, because of the increased diagnosis & treatment, and rising in demand for meningococcal, pneumococcal vaccines and other conjugate vaccines anticipate to fuel the Conjugate Vaccine Market. Also, increasing awareness about vaccination, existence of well-known pharmaceutical companies, and advancement in technology and research and development activities. In addition, government initiatives for vaccination program and funding are anticipating the market growth. Furthermore, as proclaimed by center of disease control, vaccination is the most prominent and effective evidence to prevent disease and eradicate various life threating disease.
However, Asia is expected to grow at the highest CAGR during the forecast period thanks to the growing focus of major players on emerging market such as Singapore, China, India. In addition increasing geriatric population and initiatives by governmental & organizations such as WHO and GAVI in increasing immunization and thus, drives the market.
Europe is estimated to have substantial growth as to initiatives taken by key players in development of Conjugate Vaccine. For example, in March Pfizer (US) received approval from European commission to use prevenar 13 for prevention and vaccination of pneumonia.
Conjugate Vaccine Market Competitive Analysis:
The report conjugate vaccines market analysis includes information of detailed analysis leading manufacturers and upcoming trends & challenges that will influence market growth. Also, companies strategize implementation such as acquisition, partnership, collaborations and Product launches on forthcoming growth opportunities and to lessen the competition in the competitive landscape.
The objective of the report is to present a comprehensive analysis of the Global Conjugate Vaccine Market including all the stakeholders of the industry. The past and current status of the industry with forecasted market size and trends are presented in the report with the analysis of complicated data in simple language. The report covers all the aspects of the industry with a dedicated study of key players that includes market leaders, followers, and new entrants. PORTER, SWOT, PESTEL analysis with the potential impact of micro-economic factors of the market has been presented in the report. External as well as internal factors that are supposed to affect the business positively or negatively have been analyzed, which will give a clear futuristic view of the industry to the decision-makers.
Recent Industry Developments (2025–2026):
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 19 January 2026 | AIM Vaccine | The company received clinical approval for its upgraded 20-valent Pneumococcal Conjugate Vaccine (PCV20) in China. | This development enhances domestic competition against global pharmaceutical giants in the high-demand Chinese vaccine market. |
| 14 January 2026 | Merck & Co. Inc. | Merck completed the strategic acquisition of Cidara Therapeutics to bolster its conjugate vaccine pipeline. | The acquisition expands Merck's capabilities in targeting resistant pneumococcal strains, strengthening its long-term market position. |
| 03 August 2025 | Pfizer Inc. | Pfizer officially launched its next-generation 20-valent Pneumococcal Polysaccharide Conjugate Vaccine (PCV20) for adults in India. | This launch addresses the high disease burden in South Asia by providing broader serotype coverage in a single dose. |
| 15 April 2025 | GSK plc | The CDC’s ACIP voted to endorse Penmenvy, a five-in-one meningococcal conjugate vaccine, for adolescent schedules. | This endorsement simplifies immunization programs by reducing the number of injections required for comprehensive protection. |
| 11 February 2025 | Bavarian Nordic A/S | The European Commission granted marketing authorization for VIMKUNYA, a recombinant chikungunya vaccine for individuals aged 12 and older. | It represents a major expansion into recombinant/conjugate technologies for emerging viral threats in the European region. |
Key Trends & Developments in the Market:
• In March 2019, Pfizer introduced three phases III trials on its new 20-valent pneumococcal conjugate vaccine.
• In November 2018, Haryana Government proclaimed to launch a specially designed vaccine new Pneumococcal Conjugate Vaccine to safeguard the children from pneumonia. ANMOL has also launched an application to keep all their data online. The main aim was to provide a cost-effective vaccine for children suffering from pneumonia.
• In September 2018, the first public-sector typhoid conjugate vaccine campaign was launched by Navi Mumbai Municipal Corporation. The main aim was to vaccinate around 320,000 children between the aged of 9 months to 15 years. The NMMC received 100000 doses from the Bharat Biotech International Limited. This campaign was done in two phases and their main motive is to reduce the burden of typhoid.
The report also helps in understanding Global Conjugate Vaccine Market dynamics, structure by analyzing the market segments and projects the Global Conjugate Vaccine Market size. Clear representation of competitive analysis of key players By Pathogen Type, price, financial position, Product portfolio, growth strategies, and regional presence in the Global Conjugate Vaccine Market make the report investor’s guide
Conjugate Vaccine Market Scope: Inquire before buying
| Conjugate Vaccine Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 21.3 USD Billion |
| Forecast Period 2026-2032 CAGR: | 9.41% | Market Size in 2032: | 39.97 USD Billion |
| Segments Covered: | by Type | Monovalent Conjugate Vaccines Multivalent Conjugate Vaccines |
|
| by Disease Indication | Pneumococcal Influenza DTP Meningococcal Others |
||
| by Pathogen Type | Bacterial Conjugate Vaccine Viral Conjugate Vaccine Combination (Viral and Bacterial) Conjugate Vaccine |
||
| by Patient Age | Pediatrics Conjugate Vaccine Adults Conjugate Vaccine |
||
Conjugate Vaccine Market, by Region:
• North America (United States, Canada and Mexico)
• Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe)
• Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan and Rest of APAC)
• Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A)
• South America (Brazil, Argentina Rest of South America)
Key Players / Competitors Profiles Covered in Brief in Global Conjugate Vaccine Market Report in Strategic Perspective:
- Sanofi S.A.
- Pfizer Inc.
- Merck & Co. Inc.
- GlaxoSmithKline plc
- Bharat Biotech International Limited
- Serum Institute of India Private Limited
- Biological E. Limited
- Bavarian Nordic A/S
- CSL Limited
- Novartis AG
- GreenSignal Bio Pharma Limited
- Taj Pharmaceuticals Limited
- Vaxcyte Inc.
- Neuron Biotech Co. Ltd.
- Bio-Med Pvt. Ltd.
- Sinovac Biotech Ltd.
- Takeda Pharmaceutical Company Limited
- Valneva SE
- Dynavax Technologies Corporation
- Astellas Pharma Inc.
- Hualan Biological Engineering Inc.
- Panacea Biotec Ltd.
- Protein Sciences Corporation
- Daiichi Sankyo Company Limited
- Mitsubishi Tanabe Pharma Corporation
