Artificial Urinary Sphincter Implantation Device Market by Gender, and Region – Global Market Size Estimation, Industry-Wide Analysis, Competitive Landscape Assessment & Long-Term Forecast to 2032
Overview
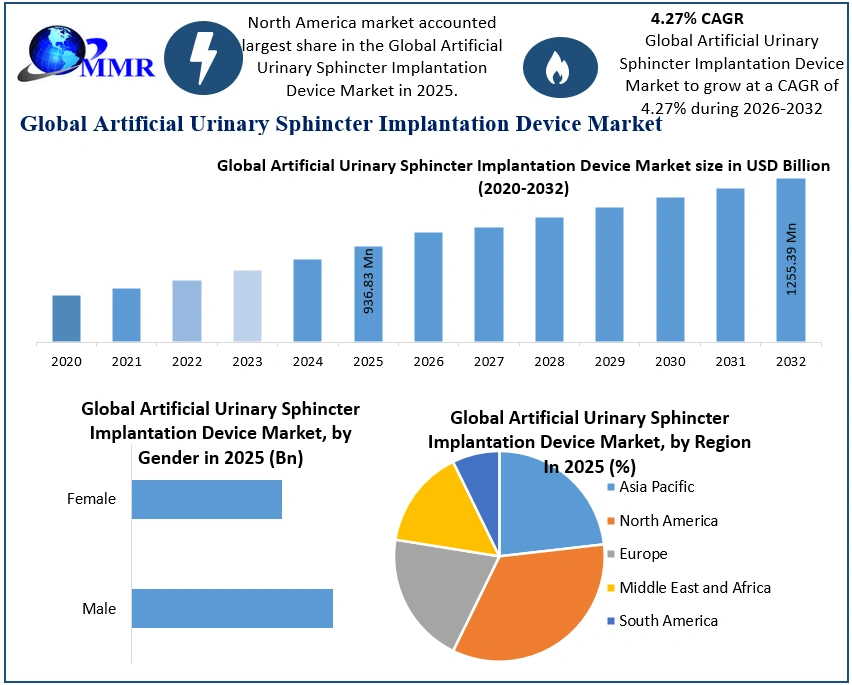
The Artificial Urinary Sphincter Implantation Device Market size was valued at USD 936.83 Million in 2025 and the total Artificial Urinary Sphincter Implantation Device revenue is expected to grow at a CAGR of 4.27% from 2025 to 2032, reaching nearly USD 1255.39 Million by 2032.
An artificial urinary sphincter (AUS) is an implanted device, treats individuals with moderate to severe stress urinary incontinence most commonly in men. The AUS is designed to supplement the function of the natural urinary sphincter that restricts urine flow out of the bladder.In the USA approximately 50% of all nursing home residents and 15%-30% of women over the age of 65 years are suffering from Urge Incontinence. Novel and ingenious technical design in AUS devices has been used to restore the quality of life for many men and women plagued by severe stress urinary incontinence.
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
The report covers detailed competitive landscape including company profiles of key players operating in the global market. Some key players in the artificial urinary sphincter implantation device market include Albyn Medical Ltd., ANDROMEDA medizinische Systeme GmbH, Cook Urological Inc., CooperSurgicalInc. An in-depth view of the competitive outlook includes future capacities, key mergers & acquisitions, financial overview, partnerships, collaborations, new product launches, new product developments and other developments with information in terms of Drivers, Restrain, Opportunities and challenges.
Artificial Urinary Sphincter Implantation Device Market Dynamics
Novel techniques and materials will emerge to meet the ongoing need for alternative, minimally invasive, and adjustable options for management of patients with low-to-moderate volume incontinence. This help to widden the opportunities of the global artificial urinary sphincter Implantation Device Market. Increasing investment in the research and development and rising health care expenditure in adoption of new urinary implantation devices, drive the global market by 2025-2032.Problems with any part of the AUS, which requires surgery to take it out or replace it. This may be the restraining factor for the global market growth.0
InVance Male Suburethral Sling segment is expected to command largest market share of xx% by 2032.
The InVance male suburethral sling represents an important treatment option for male stress urinary incontinence. This is an alternative minimally invasive surgical choice for men with mild to moderate stress urinary incontinence (SUI).Prevalence of prostate cancer with need for ongoing treatments, will likely remain a significant need ofInVance Male Suburethral sling treatment SUI for the foreseeable future. It is observed that there will be ongoing improvements to the InVance male suburethral sling to increase longevity, reduce infectivity, and to better cater to patients with limited manual/mental capability.
It is similarly expected that new variations and improvements to the existing InVance male slings will continue to be developed, with additional and longer-term follow-up on previously installed slings, helps in growth of this segment during forecast period.
AMS 800 segment is expected to command second largest market share of xx% by 2032.
The AMS 800 Urinary Control System is intended for use in the treatment of both female &male urinary incontinence. The patients who are determined by their doctor as permanent blockage of the lower urinary tract or who have uncontrollable contraction of the bladder use AMS 800 artificial urinary sphincter implantation device.
Stress Incontinence segment is expected to command largest market share of xx% by 2032.
Artificial urinary sphincter device continues to restore the quality of life for many men and women plagued by severe stress urinary incontinence. Male slings are an alternative minimally invasive surgical choice for men’s with mild to moderate stress urinary incontinence (SUI). Stress incontinence is the major reason behind the patients with artificial urinary sphincter device. The InVance male suburethral sling procedure makes it possible to treat stress urinary incontinence after prostate surgery with satisfactory and lasting short-term results. AMS 800 device as a highly durable, effective and safe treatment option for stress urinary incontinence. Urinary Implantation device with stress-release balloon to increase occlusion pressure during periods of stress. The Pro-ACT device was originally conceived and developed for the treatment of female stress Incontinence. Hence global artificial urinary sphincter implantation device market boost by growing research & development in these devices and growing technological adoption.
Male segment is expected to command largest market share of xx% by 2032.
Male stress urinary incontinence (SUI) affects a percentage of men undergoing urologic procedures with a significant impact on quality of life. Widden treatment availability on male stress urinary incontinence drive the global market globally. Males with other complications such as infection, erosion, retention, explintation, and transient pain, rising the global artificial urinary sphincter implantation device market. An increasing number of options continue to be developed for the management of male urinary incontinence, which in turn impel the market growth during forecast period.
North America is expected to dominate the global artificial urinary sphincter implantation device market during forecast period.
Health system strengthening, human resource development, capacity building and regulation in public health from North America drive the global artificial urinary sphincter implantation device market during forecast period. One fundamental reason for upsurge functioning of health systems and healthy indications among individuals is the high investment on healthcare sector by government in this region. Improving quality of healthcare professionals especially urologist from North America, helps to cure patients suffering from urinary bladder disorders. Public health concerned with disease prevention and control at the population level through organized efforts and informed choices of society, organizations, public and private communities and individuals, foster the global market growth from North America. A good system of regulation in this region is fundamental to successful public health outcomes as it reduces exposure to disease through enforcement of sanitary codes, which in turn drive the global market by reducing side effects caused by artificial urinary sphincter device.
The objective of the report is to present a comprehensive analysis of the Global Artificial Urinary Sphincter Implantation Device Market to the stakeholders in the industry. The past and current status of the industry with forecasted market size and trends are presented in the report with the analysis of complicated data in simple language. The report covers all the aspects of the industry with a dedicated study of key players that includes market leaders, followers, and new entrants. PORTER, SVOR, PESTEL analysis with the potential impact of micro-economic factors of the market have been presented in the report. External as well as internal factors that are supposed to affect the business positively or negatively have been analyzed, which will give a clear futuristic view of the industry to the decision-makers.
The report also helps in understanding Global Artificial Urinary Sphincter Implantation Device Market dynamics, structure by analyzing the market segments and project the Global Artificial Urinary Sphincter Implantation Device Market size. Clear representation of competitive analysis of key players by product, price, financial position, product portfolio, growth strategies, and regional presence in the Global Artificial Urinary Sphincter Implantation Device Market make the report investor’s guide.
Artificial Urinary Sphincter Implantation Device Market Scope: Inquire before buying
| Artificial Urinary Sphincter Implantation Device Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | USD 936.83 Mn. |
| Forecast Period 2026 to 2032 CAGR: | 4.27% | Market Size in 2032: | USD 1255.39 Mn. |
| Segments Covered: | by Gender | Male Female |
|
Artificial Urinary Sphincter Implantation Device Market, by Region
North America (United States, Canada and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan and Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A)
South America (Brazil, Argentina Rest of South America)
Artificial Urinary Sphincter Implantation Device Market, Key Players:
1.Albyn Medical, Ltd.
2.ANDROMEDA medizinischeSysteme GmbH
3.Cook Urological Inc.
4.CooperSurgical Inc.
5.Laborie Medical Technologies, Inc.
6.Boston Scientific Corporation
7.ZEPHYR Surgical Implants
8.RBM-Med
9. Silimed
10. GT Urological
11. Others
Frequently Asked Questions:
1. Which region has the largest share in Global Artificial Urinary Sphincter Implantation Device Market?
Ans: North America region held the highest share in 2025.
2. What is the growth rate of Global Artificial Urinary Sphincter Implantation Device Market?
Ans: The Global Artificial Urinary Sphincter Implantation Device Market is growing at a CAGR of 4.27% during forecasting period 2026-2032.
3. What is scope of the Global Market report?
Ans: Global Market report helps with the PESTEL, PORTER, COVID-19 Impact analysis, Recommendations for Investors & Leaders, and market estimation of the forecast period.
4. Who are the key players in Global Market?
Ans: The important key players in the Global Market are – Albyn Medical, Ltd., ANDROMEDA medizinischeSysteme GmbH, Cook Urological Inc., CooperSurgicalInc., Laborie Medical Technologies, Inc., Boston Scientific Corporation, ZEPHYR Surgical Implants, RBM-Med, Silimed, GT Urological, Others, and
5. What is the study period of this Market?
Ans: The Global Market is studied from 2025 to 2032.
