1. Acquired Haemophilia Therapeutics Market Introduction
1.1. Study Assumption and Market Definition
1.2. Scope of the Study
1.3. Executive Summary
2. Global Acquired Haemophilia Therapeutics Market: Competitive Landscape
2.1. MMR Competition Matrix
2.2. Competitive Landscape
2.3. Key Players Benchmarking
2.3.1. Company Name
2.3.2. Business Segment
2.3.3. End-user Segment
2.3.4. Revenue (2025)
2.3.5. Company Locations
2.4. Leading Acquired Haemophilia Therapeutics Market Companies, by market capitalization
2.5. Market Structure
2.5.1. Market Leaders
2.5.2. Market Followers
2.5.3. Emerging Players
2.6. Mergers and Acquisitions Details
3. Acquired Haemophilia Therapeutics Market: Dynamics
3.1. Acquired Haemophilia Therapeutics Market Trends by Region
3.1.1. North America Acquired Haemophilia Therapeutics Market Trends
3.1.2. Europe Acquired Haemophilia Therapeutics Market Trends
3.1.3. Asia Pacific Acquired Haemophilia Therapeutics Market Trends
3.1.4. Middle East and Africa Acquired Haemophilia Therapeutics Market Trends
3.1.5. South America Acquired Haemophilia Therapeutics Market Trends
3.2. Acquired Haemophilia Therapeutics Market Dynamics by Region
3.2.1. North America
3.2.1.1. North America Acquired Haemophilia Therapeutics Market Drivers
3.2.1.2. North America Acquired Haemophilia Therapeutics Market Restraints
3.2.1.3. North America Acquired Haemophilia Therapeutics Market Opportunities
3.2.1.4. North America Acquired Haemophilia Therapeutics Market Challenges
3.2.2. Europe
3.2.2.1. Europe Acquired Haemophilia Therapeutics Market Drivers
3.2.2.2. Europe Acquired Haemophilia Therapeutics Market Restraints
3.2.2.3. Europe Acquired Haemophilia Therapeutics Market Opportunities
3.2.2.4. Europe Acquired Haemophilia Therapeutics Market Challenges
3.2.3. Asia Pacific
3.2.3.1. Asia Pacific Acquired Haemophilia Therapeutics Market Drivers
3.2.3.2. Asia Pacific Acquired Haemophilia Therapeutics Market Restraints
3.2.3.3. Asia Pacific Acquired Haemophilia Therapeutics Market Opportunities
3.2.3.4. Asia Pacific Acquired Haemophilia Therapeutics Market Challenges
3.2.4. Middle East and Africa
3.2.4.1. Middle East and Africa Acquired Haemophilia Therapeutics Market Drivers
3.2.4.2. Middle East and Africa Acquired Haemophilia Therapeutics Market Restraints
3.2.4.3. Middle East and Africa Acquired Haemophilia Therapeutics Market Opportunities
3.2.4.4. Middle East and Africa Acquired Haemophilia Therapeutics Market Challenges
3.2.5. South America
3.2.5.1. South America Acquired Haemophilia Therapeutics Market Drivers
3.2.5.2. South America Acquired Haemophilia Therapeutics Market Restraints
3.2.5.3. South America Acquired Haemophilia Therapeutics Market Opportunities
3.2.5.4. South America Acquired Haemophilia Therapeutics Market Challenges
3.3. PORTER's Five Forces Analysis
3.4. PESTLE Analysis
3.5. Technology Roadmap
3.6. Regulatory Landscape by Region
3.6.1. North America
3.6.2. Europe
3.6.3. Asia Pacific
3.6.4. Middle East and Africa
3.6.5. South America
3.7. Key Opinion Leader Analysis For Acquired Haemophilia Therapeutics Industry
3.8. Analysis of Government Schemes and Initiatives For Acquired Haemophilia Therapeutics Industry
3.9. Acquired Haemophilia Therapeutics Market Trade Analysis
3.10. The Global Pandemic Impact on Acquired Haemophilia Therapeutics Market
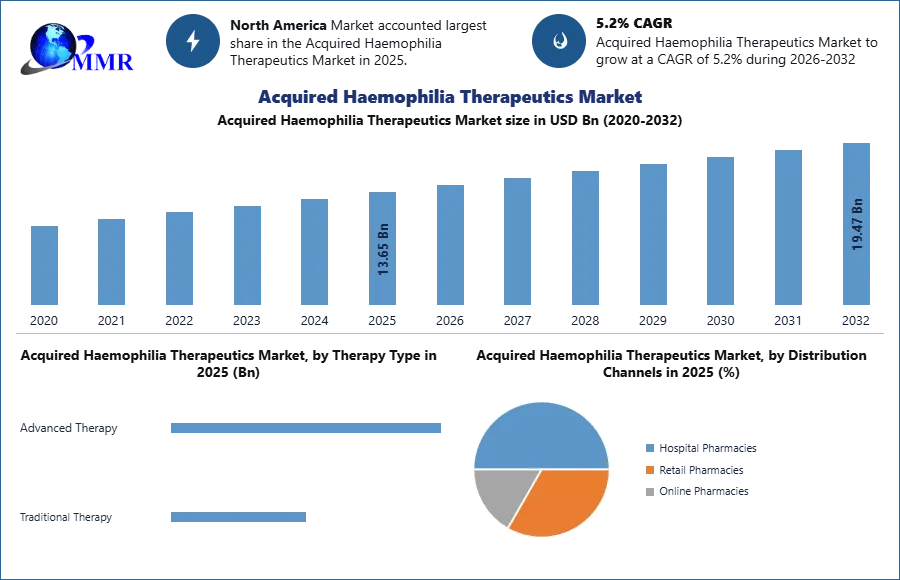
4. Acquired Haemophilia Therapeutics Market: Global Market Size and Forecast by Segmentation (in USD Bn) 2025-2032
4.1. Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
4.1.1. Bypassing Agents
4.1.2. Recombinant Factor VIIa
4.1.3. Activated Prothrombin Complex Concentrates (aPCC)
4.1.4. Immunosuppressive Drugs
4.1.5. Others
4.2. Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
4.2.1. Hospital Pharmacies
4.2.2. Retail Pharmacies
4.2.3. Online Pharmacies
4.3. Acquired Haemophilia Therapeutics Market Size and Forecast, by Region (2025-2032)
4.3.1. North America
4.3.2. Europe
4.3.3. Asia Pacific
4.3.4. Middle East and Africa
4.3.5. South America
5. North America Acquired Haemophilia Therapeutics Market Size and Forecast by Segmentation (in USD Bn) 2025-2032
5.1. North America Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
5.1.1. Bypassing Agents
5.1.2. Recombinant Factor VIIa
5.1.3. Activated Prothrombin Complex Concentrates (aPCC)
5.1.4. Immunosuppressive Drugs
5.1.5. Others
5.2. North America Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
5.2.1. Hospital Pharmacies
5.2.2. Retail Pharmacies
5.2.3. Online Pharmacies
5.3. North America Acquired Haemophilia Therapeutics Market Size and Forecast, by Country (2025-2032)
5.3.1. United States
5.3.1.1. United States Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
5.3.1.1.1. Bypassing Agents
5.3.1.1.2. Recombinant Factor VIIa
5.3.1.1.3. Activated Prothrombin Complex Concentrates (aPCC)
5.3.1.1.4. Immunosuppressive Drugs
5.3.1.1.5. Others
5.3.1.2. United States Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
5.3.1.2.1. Hospital Pharmacies
5.3.1.2.2. Retail Pharmacies
5.3.1.2.3. Online Pharmacies
5.3.2. Canada
5.3.2.1. Canada Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
5.3.2.1.1. Bypassing Agents
5.3.2.1.2. Recombinant Factor VIIa
5.3.2.1.3. Activated Prothrombin Complex Concentrates (aPCC)
5.3.2.1.4. Immunosuppressive Drugs
5.3.2.1.5. Others
5.3.2.2. Canada Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
5.3.2.2.1. Hospital Pharmacies
5.3.2.2.2. Retail Pharmacies
5.3.2.2.3. Online Pharmacies
5.3.3. Mexico
5.3.3.1. Mexico Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
5.3.3.1.1. Bypassing Agents
5.3.3.1.2. Recombinant Factor VIIa
5.3.3.1.3. Activated Prothrombin Complex Concentrates (aPCC)
5.3.3.1.4. Immunosuppressive Drugs
5.3.3.1.5. Others
5.3.3.2. Mexico Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
5.3.3.2.1. Hospital Pharmacies
5.3.3.2.2. Retail Pharmacies
5.3.3.2.3. Online Pharmacies
6. Europe Acquired Haemophilia Therapeutics Market Size and Forecast by Segmentation (in USD Bn) 2025-2032
6.1. Europe Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
6.2. Europe Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
6.3. Europe Acquired Haemophilia Therapeutics Market Size and Forecast, by Country (2025-2032)
6.3.1. United Kingdom
6.3.1.1. United Kingdom Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
6.3.1.2. United Kingdom Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
6.3.2. France
6.3.2.1. France Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
6.3.2.2. France Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
6.3.3. Germany
6.3.3.1. Germany Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
6.3.3.2. Germany Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
6.3.4. Italy
6.3.4.1. Italy Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
6.3.4.2. Italy Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
6.3.5. Spain
6.3.5.1. Spain Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
6.3.5.2. Spain Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
6.3.6. Sweden
6.3.6.1. Sweden Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
6.3.6.2. Sweden Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
6.3.7. Austria
6.3.7.1. Austria Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
6.3.7.2. Austria Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
6.3.8. Rest of Europe
6.3.8.1. Rest of Europe Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
6.3.8.2. Rest of Europe Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
7. Asia Pacific Acquired Haemophilia Therapeutics Market Size and Forecast by Segmentation (in USD Bn) 2025-2032
7.1. Asia Pacific Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
7.2. Asia Pacific Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
7.3. Asia Pacific Acquired Haemophilia Therapeutics Market Size and Forecast, by Country (2025-2032)
7.3.1. China
7.3.1.1. China Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
7.3.1.2. China Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
7.3.2. S Korea
7.3.2.1. S Korea Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
7.3.2.2. S Korea Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
7.3.3. Japan
7.3.3.1. Japan Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
7.3.3.2. Japan Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
7.3.4. India
7.3.4.1. India Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
7.3.4.2. India Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
7.3.5. Australia
7.3.5.1. Australia Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
7.3.5.2. Australia Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
7.3.6. Indonesia
7.3.6.1. Indonesia Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
7.3.6.2. Indonesia Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
7.3.7. Malaysia
7.3.7.1. Malaysia Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
7.3.7.2. Malaysia Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
7.3.8. Vietnam
7.3.8.1. Vietnam Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
7.3.8.2. Vietnam Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
7.3.9. Taiwan
7.3.9.1. Taiwan Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
7.3.9.2. Taiwan Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
7.3.10. Rest of Asia Pacific
7.3.10.1. Rest of Asia Pacific Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
7.3.10.2. Rest of Asia Pacific Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
8. Middle East and Africa Acquired Haemophilia Therapeutics Market Size and Forecast by Segmentation (in USD Bn) 2025-2032
8.1. Middle East and Africa Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
8.2. Middle East and Africa Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
8.3. Middle East and Africa Acquired Haemophilia Therapeutics Market Size and Forecast, by Country (2025-2032)
8.3.1. South Africa
8.3.1.1. South Africa Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
8.3.1.2. South Africa Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
8.3.2. GCC
8.3.2.1. GCC Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
8.3.2.2. GCC Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
8.3.3. Nigeria
8.3.3.1. Nigeria Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
8.3.3.2. Nigeria Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
8.3.4. Rest of ME&A
8.3.4.1. Rest of ME&A Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
8.3.4.2. Rest of ME&A Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
9. South America Acquired Haemophilia Therapeutics Market Size and Forecast by Segmentation (in USD Bn) 2025-2032
9.1. South America Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
9.2. South America Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
9.3. South America Acquired Haemophilia Therapeutics Market Size and Forecast, by Country (2025-2032)
9.3.1. Brazil
9.3.1.1. Brazil Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
9.3.1.2. Brazil Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
9.3.2. Argentina
9.3.2.1. Argentina Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
9.3.2.2. Argentina Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
9.3.3. Rest Of South America
9.3.3.1. Rest Of South America Acquired Haemophilia Therapeutics Market Size and Forecast, by Drug Class (2025-2032)
9.3.3.2. Rest Of South America Acquired Haemophilia Therapeutics Market Size and Forecast, by Distribution Channels (2025-2032)
10. Company Profile: Key Players
10.1. Takeda Pharmaceutical Company Limited
10.1.1. Company Overview
10.1.2. Business Portfolio
10.1.3. Financial Overview
10.1.4. SWOT Analysis
10.1.5. Strategic Analysis
10.1.6. Scale of Operation (small, medium, and large)
10.1.7. Details on Partnership
10.1.8. Regulatory Accreditations and Certifications Received by Them
10.1.9. Awards Received by the Firm
10.1.10. Recent Developments
10.2. Novo Nordisk A/S
10.3. Sanofi
10.4. F. Hoffmann-La Roche Ltd
10.5. Pfizer Inc.
10.6. Bayer AG
10.7. Octapharma AG
10.8. Grifols S.A.
10.9. CSL Behring
10.10. Biogen Inc.
10.11. Sobi (Swedish Orphan Biovitrum AB)
10.12. LFB S.A.
10.13. Kedrion S.p.A
10.14. Genentech Inc.
10.15. Emergent BioSolutions Inc.
10.16. Aptevo Therapeutics
10.17. BioMarin Pharmaceutical Inc.
10.18. Spark Therapeutics Inc.
10.19. Sangamo Therapeutics Inc.
10.20. uniQure N.V.
10.21. Novo Nordisk Health Care AG
10.22. Baxter International Inc.
10.23. Bristol-Myers Squibb Company
11. Key Findings
12. Industry Recommendations
13. Acquired Haemophilia Therapeutics Market: Research Methodology
14. Terms and Glossary
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report