GI Stool Testing Market Size by Product Type, Test Type, Application, Distribution Channel, End-Use, Region – Segment-Level Market Assessment, Growth Opportunity Analysis, Competitive Mapping & Forecast to 2032
Overview
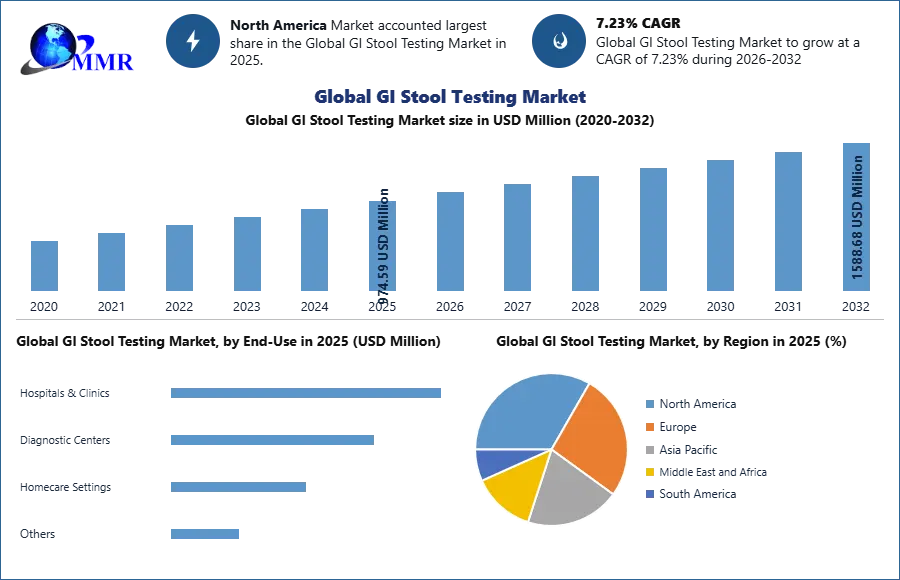
The Global GI Stool Testing Market size was valued at USD 974.59 Million in 2025 and the total GI Stool Testing Market revenue is expected to grow at a CAGR of 7.23 % from 2026 to 2032, reaching nearly USD 1588.68 Million.
Gastrointestinal (GI) stool testing refers to diagnostic tests that analyze stool samples for the detection of gastrointestinal diseases, including infections, bleeding, inflammation, and colorectal cancer. It includes various tests like fecal occult blood tests (FOBT), DNA-based stool tests, and other advanced methods aimed at detecting biomarkers indicative of diseases like colorectal cancer. The GI Stool Testing Market is driven by factors such as the rise in adoption of direct-to-consumer laboratory testing, increasing awareness and screening programs, and technological advancements in point-of-care tests. The most commonly used test for bowel cancer screening is the fecal occult blood test (FOBT), which is non-specific for colorectal cancer and has limited sensitivity.
The GI Stool Testing Market trend is witnessing a surge in non-invasive and at-home stool testing solutions, due to technological advancements and consumer preferences for convenient diagnostics. Key drivers include the development of innovative devices like Shineco Inc.'s patented fecal specimen sampling device, streamlining sample collection, and enhancing the user experience during self-collection. Additionally, the introduction of novel screening tests, such as ColoClear by Prenetics Group Limited and New Horizon Health, utilizing advanced DNA biomarkers to detect early colorectal cancer signs, further propels market growth and fosters increased adoption of non-invasive testing methods. These recent developments by GI Stool Testing Market players signify a shift towards user-friendly, non-invasive, and efficient stool-based screening options, contributing to the market's growth and providing individuals with accessible and effective means of gastrointestinal disease detection and early intervention.
GI Stool Testing Market Scope and Research Methodology:
The GI Stool Testing Market's scope involves a broad range of diagnostic tests conducted on stool samples to identify and monitor gastrointestinal disorders and diseases. It includes various testing approaches, including fecal occult blood tests (FOBT), stool DNA tests, and other stool-based examinations, contributing to the detection and management of gastrointestinal ailments. The market involves both traditional and innovative screening methods, catering to different disease detection needs within the gastrointestinal domain. The research methodology employed in analyzing the GI Stool Testing Market typically involves a comprehensive approach. It incorporates primary and secondary research techniques to gather relevant data and insights.
Primary research involves conducting surveys, interviews, and interactions with industry experts, healthcare professionals, and key stakeholders. It helps in acquiring firsthand information and market dynamics, offering valuable insights into market trends, challenges, and opportunities. The analysis often includes qualitative and quantitative assessments to evaluate market size, growth prospects, and competitive scenarios. This involves studying market trends, market share analysis, growth rates, market segmentation, and forecasting future market trends. Additionally, the research methodology involves examining geographical trends, regulatory factors, technological advancements, and key market players' strategies to comprehend the market's dynamics comprehensively.
To know about the Research Methodology :- Request Free Sample Report
GI Stool Testing Market Dynamics:
Geriatric Population and Regulatory Support Drive Growth in the Stool-Based Testing Market:
The increasing adoption of direct-to-consumer laboratory testing, such as fecal and occult blood tests like Hemoccult, is expected to drive market growth. This trend allows individuals to conveniently access and perform stool-based tests at home, leading to market growth. Ongoing advancements in testing technologies, such as the development of improved fecal and occult blood tests like Hemoccult II SENSA, contribute to market growth by enhancing test accuracy and clinical sensitivity, thereby driving demand for these products. Growing emphasis on early detection of gastrointestinal diseases, including colorectal cancer, through stool-based tests like Cologuard, is propelling market growth. These tests enable the identification of conditions in their early stages, leading to higher demand and market growth. The convenience and noninvasiveness of stool-based tests, such as Cologuard, which can be performed at home without special preparation, changes in diet, or medication adjustments, are driving the GI Stool Testing Market growth. This ease of use encourages more individuals to undergo screening, contributing to the GI Stool Testing Market growth. Technological innovations in sample collection, such as the Stool Sample Quantitative Collection and Extraction Device (SQED), are streamlining the collection process and improving the accuracy of stool specimen extraction. This innovation is driving market growth by enhancing the efficiency and reliability of stool-based tests.
Rising awareness about the importance of gastrointestinal screening and the implementation of screening programs, such as for colorectal cancer using tests like Cologuard, is driving the GI Stool Testing Market growth. These initiatives lead to higher test uptake and market growth. Robust clinical research collaborations, as seen with Cologuard's development in collaboration with Mayo Clinic, contribute to market growth by validating the effectiveness and accuracy of stool-based tests. This scientific validation drives market growth by increasing confidence in these tests.
The growing geriatric population, who are at higher risk of gastrointestinal diseases, is driving market growth for stool-based tests. The increased prevalence of these conditions among the elderly leads to higher demand for screening, contributing to GI Stool Testing Market growth. The expansion of automated immunoassay systems, facilitated by innovations like the SQED, is driving market growth by enabling direct use of extracted stool samples for automated testing. This streamlines the testing process and contributes to market growth. Supportive regulatory frameworks and reimbursement policies for stool-based tests, such as Cologuard, are driving market growth. These policies encourage healthcare providers and individuals to adopt these tests, leading to market growth. These market growth drivers collectively contribute to the expansion of the GI stool testing market, fostering innovation, accessibility, and effectiveness in the diagnosis and monitoring of gastrointestinal diseases.
Technological Innovations in Stool DNA Testing Propelled Market Expansion:
The development of advanced point-of-care fecal occult blood tests (FOBT), such as Hemoccult, with improved sensitivity and specificity, presents a significant growth opportunity. These tests offer convenient and rapid screening for gastrointestinal bleeding, driving market growth through increased adoption and demand. The increasing adoption of direct-to-consumer laboratory testing for fecal occult blood, including at-home screening tests like ColoClear, presents a growth opportunity. This trend allows individuals to conveniently access and perform stool-based tests at home, contributing to market growth. The GI stool testing market is expected to witness substantial growth in developing countries, such as Brazil, South Africa, Mexico, and others. This presents an opportunity for market growth through increased demand and adoption of stool-based tests in these regions. Ongoing technological innovations in stool DNA testing, such as the development of multitarget fecal DNA and RNA assays, present a significant growth opportunity. These tests, which detect key mutations and epigenetic changes, offer enhanced diagnostic sensitivity and specificity, driving GI Stool Testing Market growth due to increased demand for advanced screening methods.
The expansion of automated immunoassay systems, facilitated by innovations like the in-situ fecal specimen sampling device by Shineco, Inc., presents a growth opportunity. These advancements streamline the collection and testing process, contributing to market growth through improved efficiency and accessibility. Growing awareness of gastrointestinal diseases and the implementation of screening programs present a growth opportunity. This trend leads to higher test uptake and market growth through increased demand for stool-based tests, especially in asymptomatic individuals at average risk of colorectal cancer. The GI stool testing market is dominated by North America and Europe, with the U.S. being a major contributor. This presents an opportunity for market growth due to continued technological advancements and increased adoption of point-of-care testing products in these regions.
The focus on non-invasive testing methods, such as the development of novel at-home screening tests like ColoClear, presents a growth opportunity. These tests offer a non-invasive and convenient option for individuals, driving market growth by increasing screening compliance and demand. Efforts to reduce the costs of stool DNA tests and implement them as point-of-care diagnostics present a significant growth opportunity. This initiative aims to make advanced DNA tests more accessible, driving GI Stool Testing Market growth due to increased adoption and demand for these tests. Regulatory support and patent grants for innovative stool sample collection devices, such as the in-situ fecal specimen sampling device, present a growth opportunity. These developments encourage innovation and market growth through the protection of novel technologies and devices. These growth opportunities collectively contribute to the expansion of the GI stool testing market, fostering innovation, accessibility, and effectiveness in the diagnosis and monitoring of gastrointestinal diseases.
Table 1. Components of the Fecal Dysbiosis Marker Analysis
| Markers | Analytes |
| Digestion | Triglycerides |
| Chymotrypsin | |
| Iso-butyrate, iso-valerate, and n-valerate | |
| Meat and vegetable fibers | |
| Absorption | Long-chain fatty acids |
| Cholesterol | |
| Total fecal fat | |
| Total short-chain fatty acids | |
| Microbiology | Levels of Lactobacilli, bifidobacteria, Escherichiacoli, and other “potential pathogens,” including Aeromonas, Bacillus cereus, Campylobacter, Citrobacter, Klebsiella, Proteus, Pseudomonas, Salmonella, Shigella, Staphylococcus aureus, and Vibrio |
| Identification and quantitation of fecal yeast (including Candida albicans, Candida tropicalis, Rhodotorula, and Geotrichum) (optional viral and/or parasitology components) | |
| Metabolic | N-butyrate (considered a key energy source for colonic epithelial cells) |
| β-glucuronidase | |
| pH | |
| Short-chain fatty acid distribution (adequate amounts and proportions of the different short-chain fatty acids reflect the basic status of intestinal metabolism) | |
| Immunology | Fecal secretory immunoglobulin A (as a measure of luminal immunologic function) |
| Calprotectina |
The Invasive Nature of Total Colonoscopy Hindering Comprehensive Screening Programs:
The non-specific nature of the fecal occult Blood Test (FOBT) poses a challenge to the GI stool testing market. As it primarily detects blood in the stool, it lacks specificity for colorectal cancer, leading to potential false-positive results and the need for additional confirmatory tests, hindering its effectiveness. The low acceptance of invasive methods, such as total colonoscopy, presents a restraint on the market. Despite being the most reliable investigation for colorectal cancer diagnosis, its invasive nature and associated costs limit its widespread acceptance, creating a challenge for comprehensive screening programs.
The high costs associated with advanced stool DNA tests, such as multitarget fecal DNA and RNA assays, pose a challenge to market growth. These tests, while promising in their sensitivity and specificity, are currently limited by high costs, potentially restricting their widespread adoption and accessibility. The GI stool testing market faces challenges in developing countries, where limited resources and infrastructure hinder the adoption of advanced screening methods. This presents a restraint on market growth, particularly in regions with low healthcare infrastructure and accessibility. The need for further confirmatory tests, such as colonoscopy, for patients identified by screening tools like FOBT and stool DNA tests, poses a challenge. This requirement adds complexity and cost to the screening process, potentially impacting patient compliance and hindering market growth.
The sensitivity of traditional FOBTs to numerous interfering substances and dietary factors presents a challenge. This leads to false-positive results, necessitating additional confirmatory tests and potentially impacting the efficiency of colorectal cancer detection. Regulatory challenges and reimbursement policies for advanced stool testing methods hinder market growth. The need for regulatory approvals and reimbursement support impacts the accessibility and adoption of innovative stool testing technologies, posing a challenge to market growth.
Technological barriers to developing advanced point-of-care tests for stool-based screening present a challenge. While there is a rise in the adoption of direct-to-consumer laboratory testing, the development of cost-effective and accurate point-of-care tests remains a challenge for market growth. Patient acceptance and compliance with stool-based screening methods pose a challenge to market growth. The acceptance of at-home screening tests and the willingness of individuals to undergo stool-based testing impact the overall effectiveness of colorectal cancer screening programs. The high treatment costs associated with gastrointestinal disorders and colorectal cancer pose a challenge to the stool testing market. In price-sensitive markets, such as developing and emerging economies, the high costs associated with devices and treatment negatively impact market growth. These challenges highlight the complexities and barriers that impact the GI stool testing market, influencing the adoption, accessibility, and effectiveness of stool-based screening methods for colorectal cancer and gastrointestinal diseases.
GI Stool Testing Market Segment Analysis:
Based on Product Type, The GI Stool Testing Market presents diverse test types catering to various applications, each witnessing varying degrees of adoption and relevance within gastrointestinal diagnostics. Occult Blood Tests, including Guaiac FOB, Immuno FOB Agglutination, Lateral Flow Immuno FOB, and Immuno FOB ELISA, primarily focus on detecting microscopic blood in stool, aiding in colorectal cancer screening. These tests, especially Immuno FOB ELISA due to its enhanced sensitivity and specificity, exhibit high adoption rates for colorectal cancer screening among asymptomatic individuals and those at average risk.
Ova and Parasite Tests and Bacteria Tests are predominantly applied for diagnosing gastrointestinal infections caused by pathogens or parasites, finding considerable adoption in clinical settings for identifying infectious agents causing diseases like gastroenteritis. The Fecal Biomarkers Test segment, encompassing Tumor M2-PK Stool Tests and Transferrin Assays, stands out for its utilization in detecting biomarkers associated with gastrointestinal disorders, including colorectal cancer. Tumor M2-PK Stool Tests exhibit promising adoption for their role in identifying early-stage colorectal cancer and monitoring disease progression. Transferrin Assays demonstrate significant application in detecting gastrointestinal bleeding. Despite their efficacy, these biomarker-based tests encounter moderate adoption due to cost considerations and technological complexities. Occult blood tests like Immuno FOB ELISA dominate colorectal cancer screening. Biomarker tests exhibit promise but face challenges in extensive adoption due to cost constraints and technological complexities.
GI Stool Testing Market Regional Insights:
North America is dominating the GI Stool Testing market due to factors such as the rising burden of colon cancer and gastroesophageal reflux disease (GERD) among the population, increasing product launches, and the presence of major players like Abbott Laboratories, Genova Diagnostics, bioMérieux SA, Cardinal Health, and Danaher Corporation. The region is expected to hold the largest market share, with the United States being a major contributor to the market's growth. The market is witnessing steady growth, driven by continuous technological advancements, growing environmental awareness, and the rising need for streamlined operations. Additionally, the FDA's stringent regulatory standards ensure the validation and approval of innovative stool testing technologies, fostering market growth and adoption.
In Europe's GI Stool Testing Market, countries like Germany, France, and the UK boast well-established healthcare systems and early adoption of innovative medical technologies. The European population's increasing awareness of gastrointestinal diseases and government initiatives promoting colorectal cancer screening programs contribute to market growth. Moreover, the emphasis on preventive healthcare measures and comprehensive screening programs, alongside favorable reimbursement policies, boosts the uptake of stool-based tests. Collaborations between research institutions and market players facilitate advancements in testing methodologies, enhancing market dominance.
The Asia-Pacific region, particularly countries like China, Japan, and India, displays burgeoning growth potential in the GI stool testing market. Factors such as the rising prevalence of gastrointestinal disorders, a growing geriatric population, and increasing healthcare expenditure contribute to market expansion. Improving healthcare infrastructure, coupled with a surge in healthcare awareness and the adoption of preventive healthcare measures, fuels the demand for stool-based tests. Furthermore, increasing government initiatives to address colorectal cancer and gastrointestinal diseases through awareness campaigns and screening programs are propelling market growth in the region.
Table 2. The Following CPT Codes Are Used To Identify Individual Components Of Fecal Analysis:
| Code | Description |
| 82239 | Bile acids; total |
| 82271 | Blood, occult, by peroxidase activity (eg, guaiac), qualitative; other sources |
| 82272 | Blood, occult, by peroxidase activity (eg, guaiac), qualitative, feces, 1-3 simultaneous determinations, performed for other than colorectal neoplasm screening |
| 82271 | Blood, occult, by peroxidase activity (eg, guaiac), qualitative; other sources |
| 82274 | Blood, occult, by fecal hemoglobin determination by immunoassay, qualitative, feces, 1-3 simultaneous determinations |
| 82542 | Column chromatography, includes mass spectrometry, if performed (eg, HPLC, LC, LC/MS, LC/MS-MS, GC, GC/MS-MS, GC/MS, HPLC/MS), non-drug analyte(s) not elsewhere specified, qualitative or quantitative, each specimen |
| 82656 | Elastase, pancreatic (EL-1), fecal, qualitative or semi-quantitative |
| 82710 | Fat or lipids, feces; quantitative |
| 82715 | Fat differential, feces, quantitative |
| 82725 | Fatty acids, nonesterified |
| 82784 | Gammaglobulin (immunoglobulin); IgA, IgD, IgG, IgM, each |
| 83520 | Immunoassay for analyte other than infectious agent antibody or infectious agent antigen; quantitative, not otherwise specified |
| 83630 | Lactoferrin, fecal; qualitative |
| 83986 | pH; body fluid, not otherwise specified |
| 83993 | Calprotectin, fecal |
| 84311 | Spectrophotometry, analyte not elsewhere specified |
| 87045 | Culture, bacterial; stool, aerobic, with isolation and preliminary examination (eg, KIA, LIA), Salmonella and Shigella species |
| 87177 | Ova and parasites, direct smears, concentration, and identification |
| 87209 | Smear, a primary source with interpretation; complex special stain (eg, trichrome, iron hemotoxylin) for ova and parasites |
| 89160 | Meat fibers, feces |
Competitive Landscape
Key Players of the GI Stool Testing Market profiled in the report are AdvaCare Pharma, Biomrieux Inc., Bio-Rad Laboratory, Cardinal Health, Cenogenics Corporation, CTK Biotech Inc., Danaher Corp., Diasorin S P A, Eiken Chemical Co. Ltd., Epitope Diagnostics Inc., Exact Sciences Corp., F. Hoffmann-La Roche Ltd., Genova Diagnostics, Helena Laboratories Corp., Hologic, Inc., McKesson Corp., Meridian Bioscience Inc. This provides huge opportunities to serve many End-users and customers and expand the GI Stool Testing Market.
In December 2022, Shineco, Inc. attained FDA approval and secured a USPTO patent safeguarding its innovative in-situ fecal specimen sampling device. This device integrates a massager-like fingertip swab, a collection tube, and a lid featuring a unidirectional sampling inlet, enhancing stool sample collection efficacy and convenience.
In June 2022, Prenetics Group Limited, in collaboration with New Horizon Health, unveiled ColoClear by Circle. This pioneering at-home screening test employs sophisticated technology to identify multiple DNA biomarkers and blood cells in human stool, enabling non-invasive early detection of colorectal cancer.
These advancements reflect a growing trend towards non-invasive and convenient stool testing methods, aligning with the increasing demand for streamlined operations and the rising need for early detection of gastrointestinal diseases, including colorectal cancer. The GI stool testing market is projected to witness substantial growth, driven by factors such as the high burden of gastrointestinal diseases, technological advancements, and the increasing demand for advanced screening methods. The rising prevalence of GI diseases due to factors such as junk food consumption, hectic lifestyles, and an aging population is further propelling the growth of the GI stool testing market. These recent developments are anticipated to contribute to the market's expansion by offering more accurate, convenient, and non-invasive screening options, thereby addressing the growing need for effective diagnostic tools in the field of gastrointestinal health.
GI Stool Testing Market Scope: Inquiry Before Buying
| Global GI Stool Testing Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 974.59 USD Million |
| Forecast Period 2026-2032 CAGR: | 7.23% | Market Size in 2032: | 1588.68 USD Million |
| Segments Covered: | by Product Type | Consumables Analyzers |
|
| by Test Type | Occult Blood Test
Ova and Parasites Test
Others |
||
| by Application | Infection Inflammatory Bowel Disease Gastroesophageal Reflux Disease (GERD) Cancer Others |
||
| by Distribution Channel | Hospital & clinic pharmacies Brick & Mortar E-commerce |
||
| by End-Use | Hospitals & Clinics Diagnostic Centers Homecare Settings Others |
||
GI Stool Testing Market, by region
North America (United States, Canada and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria, Turkey, Russia and Rest of Europe)
Asia Pacific (China, India, Japan, South Korea, Australia, ASEAN and Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A)
South America (Brazil, Argentina, Columbia and Rest of South America)
Key Players/Competitors Profiles covered in the GI Stool Testing Market in Strategic Perspective
1. AdvaCare Pharma
2. Biomrieux Inc
3. Bio-Rad Laboratory
4. Cardinal Health
5. Cenogenics Corporation
6. CTK Biotech Inc.
7. Danaher Corp.
8. Diasorin S P A
9. Eiken Chemical Co. Ltd.
10. Epitope Diagnostics Inc.
11. Exact Sciences Corp.
12. F. Hoffmann-La Roche Ltd.
13. Genova Diagnostics
14. Helena Laboratories Corp.
15. Hologic, Inc.
16. McKesson Corp.
17. Meridian Bioscience Inc
18. Pinnacle BioLabs
19. Prenetics Group Limited
20. QIAGEN
21. Quest Diagnostics Incorporated
22. Quidelortho Corp.
23. ScheBo Biotech AG
24. Shineco, Inc.
25. Siemens Healthineers AG
FAQs:
1. What are the growth drivers for the GI Stool Testing Market?
Ans. Geriatric Population and Regulatory Support Drive Growth in the Stool-Based Testing Market and are expected to be the major driver for the GI Stool Testing Market.
2. What is the major opportunity for the GI Stool Testing Market growth?
Ans. Technological Innovations in Stool DNA Testing Propelled Market Expansion and is expected to be a major Opportunity in the GI Stool Testing Market.
3. Which country is expected to lead the global GI Stool Testing Market during the forecast period?
Ans. North America is expected to lead the GI Stool Testing Market during the forecast period.
4. What is the projected market size and growth rate of the GI Stool Testing Market?
Ans. The GI Stool Testing Market size was valued at USD 974.59 Million in 2025 and the total GI Stool Testing Market revenue is expected to grow at a CAGR of 7.23 % from 2026 to 2032, reaching nearly USD 1588.68 Million.
5. What segments are covered in the GI Stool Testing Market report?
Ans. The segments covered in the GI Stool Testing Market report are by Product Type, Test Type, Application, Distribution Channel, End-Use, and Region.
