Drug Device Combination Products Market Size by Product Type, Application, End user, Region, Industry-Wide Analysis, Competitive Landscape Assessment & Long-Term Forecast to 2032
Overview
The Drug Device Combination Products Market size was valued at USD 118.2 Billion in 2024 and the total Drug Device Combination Products Market size is expected to grow at a CAGR of 8.2% from 2025 to 2032, reaching nearly USD 222.04 Billion by 2032.
Drug Device Combination Products Market Overview:
Drug Device combination products represent an advancement in medical technology. These are specialized medical devices that incorporate both a device and active medicinal substances into a single entity through physical or chemical means. These innovative solutions offer precise medication delivery, localized administration, and tailored treatments, greatly enhancing the safe and efficient management of various chronic illnesses. Examples include transdermal delivery systems, inhalers, infusion pumps, drug-eluting stents, and prefilled syringes. To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
The adoption of pharmaceutical device combination products is driven by two key factors on a global scale. The increasing prevalence of chronic diseases has created a growing need for effective and innovative treatment methods. These products find applications in managing conditions such as hepatitis, diabetes, cancer, rheumatoid arthritis, multiple sclerosis, cerebral palsy, spinal cord injuries, and more. They offer multiple advantages, including reduced side effects, controlled drug distribution, improved patient compliance, and precise drug delivery.
This expanding market is also boosted by the growing demand for accurate and intelligent drug delivery systems that optimize therapeutic effectiveness while minimizing the required dosage. Industry players are integrating tracking and monitoring technologies into these combination products, enhancing their utility. They are introducing high-end products featuring advanced active ingredients, formulations, delivery methods, and dosage forms such as peptides, liposomes, and transdermal systems. A focus on developing new biologics, innovative formulations, and next-generation high-viscosity products is evident. The demand for Drug Device combination products is expected to rise as they offer patients treatment solutions that improve adherence to therapy, ultimately improving outcomes for those with chronic illnesses.
Drug Device Combination Products Market Dynamics
Drivers:
Rising Prevalence of Chronic Diseases Boosts the Market Growth
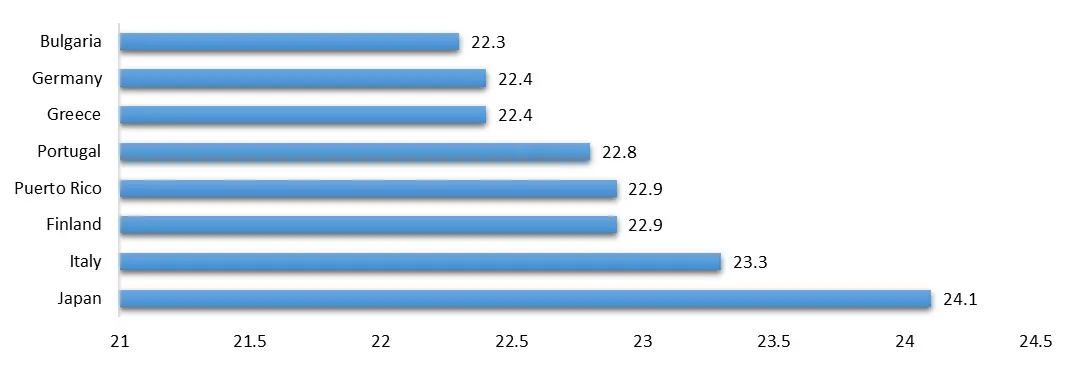
An increasing number of individuals grapple with persistent health conditions such as diabetes, heart disease, cancer, and respiratory disorders, necessitating continuous medical attention. The uptick in chronic diseases can be attributed to various factors. Population aging contributes significantly to the prevalence of these health issues, while unhealthy lifestyles, characterized by physical inactivity and poor dietary choices, exacerbate the problem. Excess weight is a specific concern, particularly concerning metabolic and cardiac conditions. Exposure to environmental pollutants, such as pollution, raises the risk of chronic health problems, including lung diseases and specific cancers. The prevalence of chronic diseases places substantial strain on healthcare systems, leading to elevated healthcare costs and heightened demand for medical services. Addressing this challenge necessitates a focus on preventive measures, early detection, and the development of novel therapeutic approaches. Promising solutions involve innovative treatments like Drug Device combination products, which provide more efficient and personalized care to those dealing with chronic diseases.
The World's Oldest Populations(%) in the year 2024
Desire for Innovative Treatments and Methods Drives Market Growth
There is a growing demand for more effective and precise healthcare treatments, driven by individuals facing complex and enduring health conditions, particularly chronic diseases. Patients aspire to therapies with minimal side effects and a high success rate while adhering to treatment plans. To meet this demand, the healthcare industry is consistently innovating, and leveraging technology, medications, and medical devices to create smarter and patient-centric treatment approaches. The drive for personalized and customized treatments is underpinned by the aim of providing to the distinct requirements of each patient.
As the healthcare landscape evolves, with a growing focus on personalized care and patient demands for treatments that not only target medical conditions but also elevate their overall well-being, the pharmaceutical industry maintains its unwavering dedication. This commitment is evident in the industry's continuous efforts in researching, developing, and introducing Drug Device combination products. These innovative solutions are thoughtfully designed to align with the continually changing healthcare needs of patients, making them an essential response to the dynamic shifts in healthcare delivery. This proactive approach by the pharmaceutical sector serves as a testament to its adaptability and its role in advancing healthcare by providing treatments that are not only effective but also attentive to the holistic well-being of patients.
Regulatory Hurdles and Safety Concerns Restrain the Market Growth
Drug Device combination product development and regulatory approval is a challenging and drawn-out procedure. Both the pharmaceutical and medical device industries have their own regulatory obligations, which businesses must manage. These regulations frequently involve many regulatory agencies and standards. Delays in product development and market entry may result from this. It is crucial to guarantee the security and efficiency of Drug Device combination solutions and pharma combination product drug-device combination products. Adverse incidents or device malfunctions are just two examples of product safety issues that may result in regulatory proceedings, recalls, or legal ramifications. To solve safety issues, manufacturers must make significant investments in clinical research and post-market monitoring. Protecting intellectual property in the Drug Device combination products market can be challenging due to the convergence of technologies from both the pharmaceutical and medical device sectors. Companies may face legal battles over patents, which can hinder innovation and market access.
Technological Innovation Creates Lucrative Growth Opportunities for Market Growth
Technological innovation in the Drug Device combination products market represents a dynamic force driving the development of novel and sophisticated medical solutions. This innovation encompasses several pivotal elements, including advanced drug delivery systems that harness microtechnology and nanotechnology to provide precise and targeted treatments, thereby ensuring optimal therapeutic outcomes. Connectivity and the Internet of Things (IoT) are pivotal in revolutionizing patient care. They enable real-time data collection and remote monitoring, enhancing patient engagement and treatment adherence. The application of biocompatible materials and advanced manufacturing techniques ensures the safety and durability of medical products. Artificial intelligence and machine learning are integral to data analysis and predictive modeling, supporting healthcare providers in making data-driven decisions. Moreover, personalized medicine, guided by genomics and diagnostics, allows for customized treatment approaches that provide to the convenience kit combination product. These innovations collectively drive the transformation of healthcare delivery, fostering improved patient outcomes and care quality.
The integration of telemedicine platforms and user-friendly interfaces ensures the seamless delivery of healthcare and empowers patients to actively manage their well-being. These technological advancements are indispensable for meeting the evolving demands of healthcare, elevating patient outcomes, and ultimately transforming the delivery of medical care. The FDA's guidance on Drug-Device combination products assumes a central role in ensuring the safety and efficacy of these innovative healthcare solutions.
Drug Device Combination Products Market Challenges
DDCPs are subject to both the US Food and Drug Administration's (FDA) and the Centers for Medicare and Medicaid Services (CMS) regulatory oversight. FDA guidance medical device combination products, it is difficult for producers as a result to comprehend the regulatory environment and introduce their FDA combination products. Because it necessitates an understanding of both drug and device development, DDCP development is expensive and time-consuming. The difficulty of DDCP reimbursement may be caused by the fact that payers are unfamiliar with these medications or may be reluctant to cover them given their high cost. On how to use DDCPs safely and correctly, patients need education. This can be challenging given how intricate DDCPs are.
Drug Device Combination Products Market Trends
Systems for regulated drug delivery that are intelligent and precise can give medication to particular bodily parts. This may assist to lessen negative effects while increasing the drug's effectiveness. The development of novel Drug Device combination products makes use of AI and ML. AI is being utilized, for instance, to create medicine delivery systems that can adjust to the specific requirements of each patient. Personalized medicine is a method of treating patients that considers their unique qualities, such as their genes, way of life, and environment. Products that combine drugs and devices are becoming more and more crucial to customized medicine because they give patients access to more precise and efficient treatment alternatives. In the Drug Device combination products market, co-packaged combination products offer convenience and efficiency in medication delivery.
Drug Device Combination Products Market Segment Analysis:
Based on By Product Type, in 2024, the Drug-Eluting Stents (DES) segment dominates the Drug-Device Combination Products market because of the rising prevalence of cardiovascular diseases, increased adoption of minimally invasive procedures, and technological advancements in stent coatings. DES offer significant advantages over traditional stents, such as reduced restenosis rates and improved long-term patient outcomes. The growing geriatric population and lifestyle-related disorders, including diabetes and hypertension, further drive demand. Additionally, continuous innovation in polymer coatings and drug formulations enhances stent efficacy, making them the preferred choice for interventional cardiologists. Regulatory approvals for newer-generation stents with enhanced biocompatibility also contribute to market growth. Strong investments by key market players and increasing awareness about cardiovascular health further solidify the dominance of DES in 2024.
Regional Insight
North America region dominates the Drug Device Combination Products Market in the year 2024. This is attributed to the significant new product development efforts carried out by prominent companies in this region. The increasing burden of chronic diseases in North America is considered a major driver, emphasizing the pressing need for these products in clinical settings and contributing to the region's market growth.
On the other hand, Asia Pacific is expected to experience substantial growth over the forecast period. The increased healthcare spending in the region. Additionally, rising awareness among physicians regarding the advantages of these products, including lower dosage requirements, timed release of medications, direct prevention of device-related infections through the use of anti-infective medications in combination with devices, and reduced systemic exposure, is contributing to the regional market's expansion. The market is also being driven by substantial research and development investments by global market players and the increasing commercialization of these systems developed by key industry players.
Drug Device Combination Products Market Scope Table : Inquire Before Buying
| Global Drug Device Combination Products Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2024 | Forecast Period: | 2025-2032 |
| Historical Data: | 2019 to 2024 | Market Size in 2024: | USD 118.2 Bn |
| Forecast Period 2025 to 2032 CAGR: | 7.1% | Market Size in 2032: | USD 222.04 Bn. |
| Segments Covered: | by Product Type | Drug Eluting Stents Infusion Pumps Inhalers Orthopedic Combination Products Photodynamic Therapy Devices Transdermal Patches Antimicrobial Catheters Others |
|
| by Application | Cardiovascular Disorders Diabetes Respiratory Problems Cancer Treatment Others |
||
| by End user | Hospitals Ambulatory Surgical Centers Others |
||
Drug Device Combination Products Market by Region
North America (United States, Canada, and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria, and the Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan, and the Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria, and the Rest of ME&A)
South America (Brazil, Argentina Rest of South America)
Drug Device Combination Products Market Key Players
1. Mérieux Nutrisciences Corporation
2. Medtronic
3. Terumo Corporation
4. Operon Strategist
5. Boston Scientific Corporation
6. BD
7. BIOTRONIK SE
8. Abbott Laboratories
9. Bausch Health Companies Inc
10. Translumina Therapeutics
11. Baxter International Inc.
12. Kaleo, Inc.
13. Biosensors International Group Ltd.
14. Novartis AG
15. Teva Pharmaceutical Industries Ltd.
16. Teleflex Incorporated
17. Smiths Medical
18. Johnson & Johnson
19. Boehringer Ingelheim
20. Zimmer Biomet
21. Fresenius Kabi
22. Cook Medical
23. MicroPort Scientific Corporation
24. B. Braun Melsungen AG
25. Micrel Medical Devices
26. Candela Medical
27. IRIDEX Corporation
28. Theralase Technologies Inc.
29. Cipla limited
30. Chiesi Farmaceutici S.p.A.
31. Others Key Players
Frequently Asked Questions:
1] What segments are covered in the Global Drug Device Combination Products Market report?
Ans. The segments covered in the Drug Device Combination Products Market report are based on products and Regions.
2] Which region is expected to hold the highest share in the Global Drug Device Combination Products Market?
Ans. The North American region is expected to hold the highest share of the Drug Device Combination Products Market.
3] What was the market size of the Global Drug Device Combination Products Market by 2024?
Ans. The market size of the Drug Device Combination Products Market by 2024 is expected to reach USD 118.2 Bn.
4] What is the forecast period for the Global Drug Device Combination Products Market?
Ans. The forecast period for the Drug Device Combination Products Market is 2025-2032.
5] What is the market size of the Global Drug Device Combination Products Market in 2032?
Ans. The market size of the Drug Device Combination Products Market in 2032 is valued at USD 222.04 Bn.