Cardiac Biomarkers Market Size by Type, Application, End Use, Region – Segment-Level Market Assessment, Growth Opportunity Analysis, Competitive Mapping & Forecast to 2032
Overview
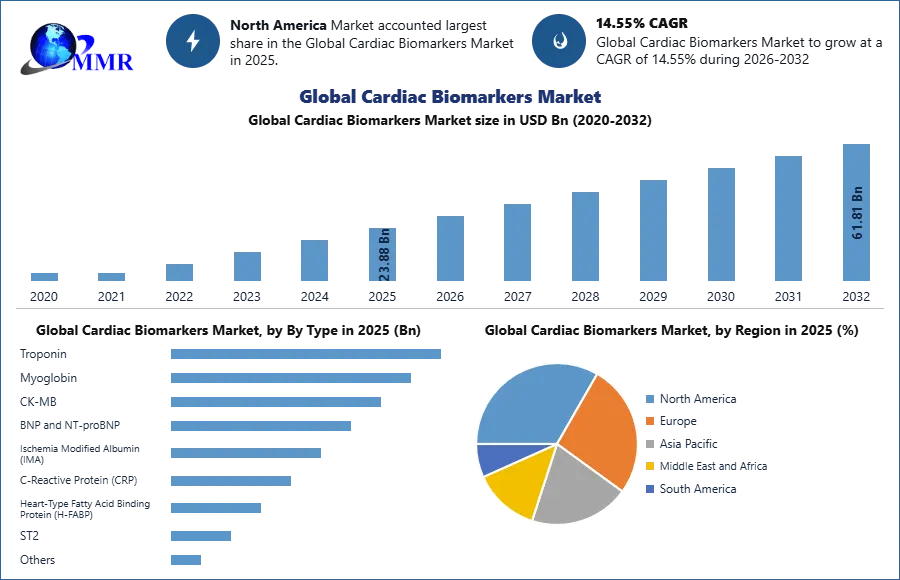
Global Cardiac Biomarkers Market was valued at USD 23.88 Bn in 2025 and is expected to reach USD 61.81 Bn by 2032, growing at a CAGR of 14.55% during the forecast period.
Cardiac Biomarkers Market Overview
Cardiac biomarkers are substances released into the bloodstream when the heart muscle is damaged or stressed. These biomarkers include enzymes, hormones, and proteins. Measuring these biomarkers help diagnose, assess risk, and manage conditions like acute coronary syndrome (ACS) and cardiac ischemia. In the United States, they're used to diagnose, assess risk, and manage acute coronary syndrome (ACS), a potentially life-threatening condition. ACS is a leading cause of death in the US, accounting for 625,000 hospital discharges annually.
 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
Examples of cardiac biomarkers include:
1. Creatine kinase (CK)
Also known as creatine phosphokinase (CPK), this muscle enzyme increases 3–4 hours after a heart attack and remains elevated for 3–4 days. This makes it useful for detecting re-infarction within 4–10 days of the initial attack.
2. Myoglobin
This small oxygen-carrying protein found in the heart rises rapidly after a heart attack, making it a valuable biomarker for differential diagnosis.
3. Glycogen phosphorylase isoenzyme BB (GPBB)
This enzyme is converted into a soluble form and released into the blood during ischemia. Levels of GPBB rise rapidly in myocardial infarction and unstable angina and is elevated 1–3 hours after ischemia.
Cardiac Biomarkers is a market with a global footprint, transcending geographical boundaries. Consumers from various continents display interest in these supplements, rendering the market a widespread presence. The Cardiac Biomarkers industry report provides a comprehensive and detailed overview of the market, incorporating statistical data alongside current trends. This report delineates the Cardiac Biomarkers market landscape, encompassing dynamics, market segments, regional analysis, and the market position of top competitors.
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 11 March 2026 | Grand View Research / bioMérieux | The acquisition of SpinChip Diagnostics ASA by bioMérieux was highlighted as a key move to commercialize a rapid point-of-care immunoassay platform. | This integration accelerates the deployment of high-sensitivity cardiac biomarkers in emergency settings, drastically reducing the turnaround time for critical triage decisions. |
| 26 February 2026 | Roche Diagnostics | Roche confirmed the global delivery of 30 billion diagnostic tests in the 2025 fiscal year, driven by high demand for its automated cardiac immunoassay systems. | The massive scale of testing underscores the market's shift toward high-throughput laboratory consolidation to manage the rising global burden of cardiovascular diseases. |
| 15 October 2025 | GlobalRPH / Industry Clinics | A clinical assessment revealed that the 2025 adoption of high-sensitivity cardiac troponin I (hs-cTnI) protocols led to an absolute 5.6% reduction in unnecessary hospital admissions. | The use of these high-precision markers directly improves emergency department throughput and optimizes healthcare resource allocation by effectively ruling out acute cardiac events faster. |
| 12 September 2025 | Roche | Roche announced primary results from its TSIX Study Program, validating the clinical performance of the new Elecsys Troponin T hs Gen 6 (sixth-generation) assay. | The launch of this next-generation assay sets a new benchmark for early myocardial injury detection, enabling clinicians to identify cardiac damage at significantly lower concentrations. |
| 20 August 2025 | MDPI / Diagnostic Research | A major study published in 2025 confirmed the high diagnostic potential of a multi-marker panel including GDF-15, sST2, and H-FABP for heart failure prognosis. | This development supports the industry transition toward multiplex biomarker panels, providing a more comprehensive risk profile than traditional single-marker troponin tests. |
| 22 January 2025 | Abbott Laboratories | The U.S. FDA cleared Abbott's Alere NT-proBNP assay for use on the Alinity i system to aid in the diagnosis and risk stratification of heart failure. | The clearance expands the cardiac testing menu on integrated laboratory platforms, enhancing workflow efficiency for high-volume hospital laboratories. |
Cardiac Biomarkers Market Dynamics
The burgeoning prevalence of cardiovascular diseases worldwide stands as a primary driver behind the growth of the cardiac biomarkers market.
As heart-related ailments persist as leading causes of mortality, the necessity for accurate diagnostic tools like cardiac biomarkers intensifies. According to the World Health Organization (WHO), cardiovascular diseases account for approximately 17.9 million deaths globally each year, representing about 31% of all global deaths. Moreover, continuous advancements in biomarker technology enhance their precision, sensitivity, and specificity, thereby bolstering their efficacy in diagnosing various heart conditions. For instance, the development of high-sensitivity troponin assays has revolutionized the diagnosis of acute myocardial infarction, allowing for earlier detection and intervention.
The cardiac biomarkers market have several constraints as the considerable cost associated with biomarker tests and diagnostic equipment, particularly inhibitory in regions with limited healthcare budgets these factors affect the growth of cardiac biomarkers Market. According to a report by the American Heart Association, the estimated direct and indirect costs of cardiovascular diseases and stroke in the United States alone total USD 351.2 billion annually. Furthermore, stringent regulatory requirements pose hurdles, often delaying the introduction of new biomarkers into the market. Additionally, in certain developing regions, inadequate awareness and healthcare infrastructure impede widespread adoption, exacerbating disparities in access to cardiovascular care.
Emerging Trends in the Cardiac Biomarkers Market
In tandem with advancements, the cardiac biomarkers market witnesses the emergence of transformative trends. The growing inclination towards personalized medicine, where treatment plans are tailored based on individual biomarker profiles, ushered in a new era of precision diagnostics and therapies. According to a study published in the Journal of the American College of Cardiology, personalized medicine approaches have shown promising results in improving outcomes for patients with cardiovascular diseases by targeting specific biomarkers. Concurrently, integration with artificial intelligence (AI) presents immense potential, as machine learning algorithms analyze biomarker data to deliver more accurate and predictive diagnostic insights.
The AI in the healthcare market, which was valued at $4.9 billion in 2020, is projected to reach USD 45.2 billion by 2026, reflecting the significant investment and interest in AI-driven solutions. Moreover, ongoing research endeavors focus on the discovery of novel biomarkers that offer comprehensive insights into cardiovascular health, promising further innovation in the field. As such, collaborations between pharmaceutical companies, diagnostic firms, and research institutions are accelerating the development and commercialization of new biomarkers, underscoring the dynamic nature of the cardiac biomarkers market.
For example, recent studies have identified new biomarkers like microRNAs and long non-coding RNAs, which could provide deeper insights into heart disease mechanisms.
This growth underscores the dynamic nature of the Cardiac Biomarkers Market, driven by advancements in technology and the continuous pursuit of improved diagnostic methods. Furthermore, collaborations between pharmaceutical companies, diagnostic firms, and research institutions are accelerating the development and commercialization of new biomarkers, further propelling market growth.
Cardiac Biomarkers Market Segment Analysis
In 2025, the Type segment shows the highest demand for Troponin biomarkers in the cardiac biomarkers market. Troponin testing is widely recognized as the gold standard for diagnosing myocardial infarction due to its high sensitivity and specificity in detecting heart muscle damage. BNP and NT-proBNP biomarkers are also experiencing strong demand, particularly for diagnosing and monitoring congestive heart failure, as they provide important insights into cardiac stress and ventricular dysfunction. CK-MB and Myoglobin remain important for early detection and confirmation of cardiac events, although their use has gradually declined with the increasing preference for more precise biomarkers such as troponin. Emerging biomarkers, including ischemia-modified albumin and heart-type fatty acid binding protein, are gaining attention in research and specialized diagnostics, but their adoption remains comparatively limited.
Based on Application, Myocardial Infarction is the most dominant and demanded segment in 2025. Cardiac biomarkers play a critical role in the early diagnosis and management of heart attacks, enabling rapid clinical decision-making and improving patient outcomes. Acute Coronary Syndrome is another significant application area where biomarker testing helps physicians differentiate between various cardiac conditions and determine appropriate treatment strategies. Congestive Heart Failure testing is also expanding rapidly as the global burden of chronic cardiovascular diseases increases, leading to higher demand for BNP and NT-proBNP tests. Atherosclerosis and other cardiovascular disorders represent smaller but growing segments, supported by the increasing emphasis on early disease detection and preventive healthcare.
Based on End Use, Laboratory Testing holds the largest share of the cardiac biomarkers market in 2025. Hospitals and diagnostic laboratories rely heavily on centralized laboratory facilities equipped with advanced immunoassay analyzers to conduct accurate and high-volume cardiac biomarker testing. These facilities provide reliable diagnostic results essential for emergency and routine cardiac care. However, Point of Care Testing is the fastest-growing segment due to its ability to deliver rapid results directly at the patient’s bedside or in emergency departments. The growing demand for immediate diagnosis in critical cardiac events, along with technological advancements in portable diagnostic devices, is driving the expansion of point-of-care testing solutions.
Cardiac Biomarkers Market Regional Insight
Cardiac Biomarkers Market Scope: Inquire Before Buying
Key Players /Competitor Profiles Covered in the Global Cardiac Biomarkers Market Report from a Strategic Perspective.
- F. Hoffmann-La Roche Ltd.
- Siemens Healthineers AG
- Thermo Fisher Scientific Inc.
- Bio-Rad Laboratories Inc.
- Danaher Corporation
- bioMérieux SA
- Randox Laboratories Ltd.
- Becton Dickinson and Company
- QuidelOrtho Corporation
- Creative Diagnostics
- Life Diagnostics Inc.
- PerkinElmer Inc.
- Tosoh Corporation
- Trinity Biotech
- Ortho Clinical Diagnostics
- Luminex Corporation
- Beckman Coulter Inc.
- Boditech Med Inc.
- BG Medicine Inc.
- Response Biomedical Corp.
- Diazyme Laboratories Inc.
- AgPlus Diagnostics Ltd.
- LSI Medience Corporation
- Signosis Inc.

