Womens Health Diagnostics Market by Diagnostic Devices, Application, End Use and Region – Global Market Size Estimation, Industry-Wide Analysis, Competitive Landscape Assessment & Long-Term Forecast to 2032
Overview
Womens Health Diagnostics Market size was valued at USD 37.59 Bn. in 2025 and the total Womens Health Diagnostics revenue is expected to grow by 8.5% from 2026 to 2032, reaching nearly USD 66.54 Bn.
Womens Health Diagnostics Market Overview:
Women's health is a branch of medicine that primarily focuses on the treatment and diagnosis of illnesses and ailments that have an impact on a woman's health. Breast cancer, ovarian cancer, cervical cancer, menopause, and pregnancy are just a few examples of the many problems specific to women that are detected by screening, testing, or diagnosis in women's health. The identification of disorders affecting women's health is possible using a variety of tests.
The use of medical diagnostic tools including mammography, imaging and monitoring systems, biopsy instruments, and others helps doctors screen for and diagnose diseases by identifying cancer, clearing obstructions, and taking pictures of interior organs. Breast cancer testing, cervical cancer testing, prenatal genetic screening & carrier testing, pregnancy & ovulation testing, and ovarian cancer testing are all part of the diagnostic process. To avoid difficulties and enhance the quality of life, tests for screening and identification of different illnesses are required. Health tests are essential for maintaining good health as well as preventing and treating major disorders.
The global demand for different health diagnostic tests has grown substantially during the last decade. Currently, the market is being pushed primarily by increased awareness of different health-related problems, the growing incidence of chronic and lifestyle disorders, and the high prevalence of infectious diseases such as HIV and hepatitis in women worldwide. Increased use of point-of-care diagnostic testing, as well as an increase in the number of diagnostic and imaging facilities, are expected to promote the growth of this market during the forecast period. However, the high cost of diagnostic equipment, tests, and procedures, as well as a scarcity of experienced laboratory personnel and inadequate reimbursement for diagnostic tests, are expected to limit this market's growth to some extent.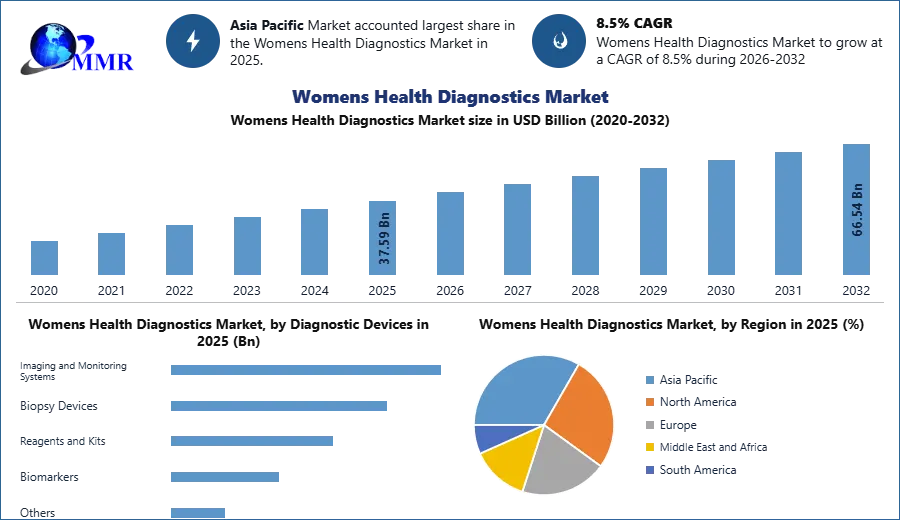 To know about the Research Methodology :- Request Free Sample Report
To know about the Research Methodology :- Request Free Sample Report
Report Scope:
A quantitative analysis of the market's drivers, restraints, trends, and projections is provided by the research on cold storage construction. Porter's five forces analysis shows how buyers and suppliers can develop their supplier-buyer networks and make decisions that are motivated by profit. With the use of extensive analysis, market size, and segmentation, the current Womens Health Diagnostics Market potential can be determined. Investors will gain a thorough understanding of the industry's future from the study, which also includes the factors expected to affect the company either favorably or unfavorably.
The research offers a complete understanding of the market for those investors. The study contains scenarios for the Womens Health Diagnostics Market from the past and present, along with projected market numbers. The report's thorough analysis of important competitors, including market leaders, followers, and new entrants, covers every aspect of the market. The research contains strategic profiles of the top market participants, a full examination of their key competencies, and their company-specific plans for the introduction of new products, growth, partnerships, joint ventures, and acquisitions. With its clear portrayal of competitive analysis of significant companies by product, pricing, financial condition, product portfolio, growth strategies, and regional presence in the domestic as well as the local market, the research acts as an investor's guide.
Womens Health Diagnostics Market Dynamics:
Market Drivers:
Increasing disease concerns
The prevalence of chronic and lifestyle-related illnesses is expected to rise, there is an increase in imaging and diagnostic facilities, more people use point-of-care and quick diagnostic tests, and there are more partnerships and collaborations across stakeholders. The prevalence of these disorders is rising, necessitating the development of accurate diagnostic tools and assays, which is propelling the development of the women's health diagnostics market throughout the forecasted time frame. Breast cancer is the most frequent disease in women and accounts for one in four cancer diagnoses in women worldwide as of December 2020, according to the Global Cancer Observatory (Globocan) statistics. Women are also frequently diagnosed with thyroid, lung, cervical, and colorectal malignancies.
Moreover, the market for women's health diagnostics is growing as a result of the growing incidence of sexually transmitted illnesses among women. For instance, a 2020 April update to an Avert article titled "Women and Girls, HIV and AIDS" said that women make up more over half of all HIV-positive individuals globally. It also mentions that young women (10 to 24 years old) have a double the risk of contracting HIV compared to young males of the same age. AIDS-related infections are still the number one killer of women between the ages of 15 and 49, according to the survey. Owing to the high burden of disease and death, there is a significant need for diagnostic tools and procedures, which in turn fuels the growth of the market under investigation.
With increasing women's health awareness and illness load, there is a continuing demand for imaging and diagnostics facilities throughout the world. The majority of diagnostic and imaging tests are conducted in centers, the market is likely to grow. According to a United Nations, Food and Drug Administration (USFDA) page titled "Mammography Quality Standards Act and Program (MQSA) National Statistics" updated in November 2021, there are 23,590 certified mammography units in the United States as of November 1st, 2021. There are 13,125 authorized Full Field Digital Mammography and Computed Radiography Units and 10,458 Digital Breast Tomosynthesis Units out of the 13,125 total. It also stated that there are 8,718 approved facilities as of October 2020, with the number expected to rise to 8,733 by November 2021. The market is growing due to the abundance of facilities in the United States.
Women have a high frequency of infectious illnesses
POC tests, for example, are routinely used to detect HIV in patients. A quick POC HIV test allows clinicians to offer patients with results and HIV status during the same visit. It does not need pre- and post-test patient counseling, as is customary in standard laboratory analysis. Women are more vulnerable to other sexually transmitted diseases (STDs) and associated long-term consequences, in addition to HIV. STDs are responsible for more than half of all avoidable infertility cases in the United States. Most sexually transmitted infections can be passed on to a fetus or child, sometimes fatally. Rapid POC testing can help with the treatment of infectious illnesses including HIV, UTIs, and TB, especially in underdeveloped nations where prompt medical care is difficult to come by and healthcare infrastructure is antiquated.
Market Restraints:
Expensive diagnostic imaging equipment and procedures
The majority of hospitals in underdeveloped countries cannot afford expensive diagnostic imaging equipment. MRI equipment typically costs between USD 1.5 million and USD 2.0 million, whilst CT machines are priced between USD 1.0 million and USD 1.5 million. Healthcare institutions like hospitals and diagnostic centers are hesitant to make large investments in pricey machinery due to the high cost of these systems and the limited financial resources of hospitals. When purchasing these pricey systems, healthcare institutions sometimes rely on third-party payers (such as Medicare, Medicaid, or commercial health insurance plans) to cover the expenses of the diagnostic and screening operations carried out utilizing this equipment.
Market Opportunities:
Growing treatment techniques
The increase in the chemotherapy industry provides several opportunities for growth. Infection is more likely in breast cancer patients. It is a kind of cancer that requires early treatment to prevent infection from spreading. Chemotherapy and other treatment modalities have gained popularity in recent years. Chemotherapy has been shown to impede the spread of cancer cells. For the majority of cancer patients, it is standard therapy. During the forecasted period, more patients receive chemotherapy. Additionally, new market innovations result from an increase in investment in this industry. During the forecast period, the market research for male breast cancer increased. The breast cancer industry expand as a result of the medical sector's technical development. Positive changes result from more technological developments in cancer therapy and gadgets. These chances for growth lead to a high rate of market revenue and growth.
Market Challenges:
More people using diagnostic imaging devices
Many developing-country and hospitals are unable to invest in diagnostic imaging technology due to high costs, low reimbursement rates, and fiscal restrictions. Owing to the strong demand for diagnostic imaging treatments in these countries, hospitals that cannot afford to invest in new imaging systems choose to use reconditioned ones. Refurbished systems are less expensive than new systems, typically costing 40% to 60% of the original purchase price.
As a result, numerous industry leaders are increasingly marketing reconditioned gadgets via various schemes. Some notable worldwide refurbishment initiatives advocating the use of reconditioned diagnostic imaging systems include Siemens' Medical Proven Excellence Program, GE Healthcare's Gold Seal Program, and Philips' Diamond Select Program.
Womens Health Diagnostics Market Segment Analysis:
Based on Application, Cancer testing, infectious disease testing, osteoporosis testing, pregnancy & fertility testing, and prenatal testing are the application segments of the market. Cancer dominated the market in 2017 as a result of the advent of technologically superior hybrid imaging tools with higher precision. For example, in July 2017, Quest Diagnostics bought the Texas laboratories Med Fusion and Clear Point in order to grow its laboratory business for the identification of cancer in women.
Various partnerships among major companies, governments, and other organizations to improve patient access to diagnostics for various ailments are expected to drive market expansion. For example, in November 2017, BGI and the Lunenfeld-Tanenbaum Research Institute agreed to collaborate on the development of a genomics-based diagnostic tool for the early identification of women at risk of preterm delivery and other pregnancy issues.
UNITAID and Population Services International began implementing self-testing in 2015 as part of the four-year Self-Testing Africa Research (STAR) initiative. From 2015 to 2017, the STAR initiative provided 750,000 self-test kits across three countries: Zambia, Malawi, and Zimbabwe.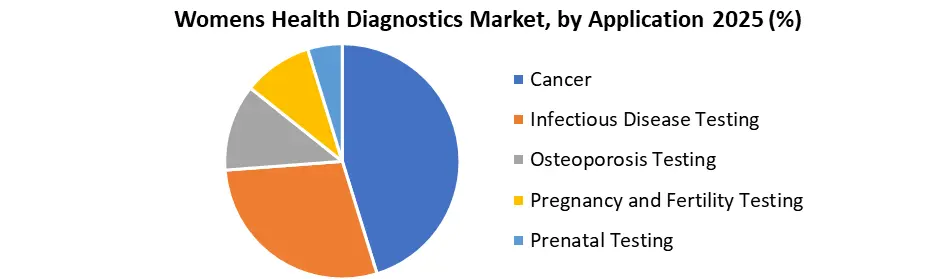 Based on the End Use, In 2022, hospitals and diagnostics centers held the highest proportion of the womens health diagnostics market. The sector is expected to maintain its market position through 2025 since they are typically the first option for patients for diagnosis of cancer, infectious disorders, and osteoporosis, among other things. The home category is expected to develop rapidly throughout the projected period as market participants increase their expenditures on commercializing portable and point-of-care devices. Furthermore, rising awareness of home diagnostic devices is expected to drive industry growth.
Based on the End Use, In 2022, hospitals and diagnostics centers held the highest proportion of the womens health diagnostics market. The sector is expected to maintain its market position through 2025 since they are typically the first option for patients for diagnosis of cancer, infectious disorders, and osteoporosis, among other things. The home category is expected to develop rapidly throughout the projected period as market participants increase their expenditures on commercializing portable and point-of-care devices. Furthermore, rising awareness of home diagnostic devices is expected to drive industry growth.
Regional Insights:
North America is expected to have a significant market share due to the high prevalence of cancer among women, increased incidence of lifestyle-related illnesses, high adoption of technologically improved goods, new launches, and government efforts. The rise is linked to an increase in the incidence of malignancies in women, such as breast and ovarian cancer, as well as other chronic illnesses, as well as the existence of important market participants in the region. According to the Breast Cancer Facts and Statistics report from July 2022, women in the United States are expected to be diagnosed with 287,850 new instances of invasive breast cancer and 51,400 new cases of non-invasive (in situ) breast cancer in 2022. As a result, the rising occurrence of breast cancer is predicted to stimulate demand for women's health diagnoses, driving the market growth.
A strong market presence by leading market players delivering technologically sophisticated products for women's health diagnostics is expected to drive regional expansion. For example, Lia will debut the world's first flushable and biodegradable pregnancy test in March 2021. Lia's pregnancy tests are manufactured from plant fibers and are completely plastic-free, so they biodegrade in just ten weeks. Better healthcare infrastructure, improved knowledge of women's health issues, and higher female labor-force involvement are all expected to boost the regional market growth.
The regulatory authority's permission is also aiding the region's market growth. Seno Medical Instruments Inc., for example, received premarket clearance (PMA) from the US Food & Drug Administration (FDA) in January 2021 for its Imagio Breast Imaging System from the Center for Devices and Radiological Health (CDRH). It is a revolutionary diagnostic breast cancer imaging tool that will assist clinicians in better distinguishing between benign and malignant breast lesions by employing non-invasive optoacoustic ultrasound (OA/US) technology to deliver information on worrisome breast lesions. Such approvals are propelling the sector forward. As a result of the aforementioned reasons, the North American womens health diagnostics market is predicted to rise significantly throughout the forecast period.
Recent Industry Developments
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 13 January 2026 | AOA Dx | Launched the AKRIVIS GD™, a first-of-its-kind multi-omics liquid biopsy for early ovarian cancer detection. | This development addresses a critical diagnostic gap in one of the deadliest cancers affecting women today. |
| 17 July 2025 | Kalorama Information | Reported that Non-invasive prenatal testing (NIPT) reached an 8% CAGR milestone following new global clinical recommendations. | The widespread adoption is transforming maternal-fetal medicine by making genetic screening standard for all pregnancies. |
| 15 May 2025 | Redcliffe Labs | Introduced Femcliffe, a specialized diagnostic platform tailored for women’s health across puberty, fertility, and pregnancy. | The platform improves healthcare accessibility by offering curative and preventive testing in a single ecosystem. |
| 10 April 2025 | Neuberg Diagnostics | Partnered with Magnum Imaging to launch the Women’s Health Card, providing low-cost access to Pap Smear screening. | This initiative lowers financial barriers for high-risk cervical cancer screenings among underserved female populations. |
Womens Health Diagnostics Market Scope: Inquire before buying
| Womens Health Diagnostics Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 37.59 USD Billion |
| Forecast Period 2026-2032 CAGR: | 8.5% | Market Size in 2032: | 66.54 USD Billion |
| Segments Covered: | by Diagnostic Devices | Imaging and Monitoring Systems Biopsy Devices Reagents and Kits Biomarkers Others |
|
| by Application | Breast Cancer Testing Cervical Cancer Testing Ovarian Cancer Testing Infectious Disease Testing Osteoporosis Testing Pregnancy & Fertility Testing Prenatal Testing |
||
| by End User | Hospitals Diagnostic & Imaging Centers Home Care Settings |
||
by Region
North America (United States, Canada and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan and Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A)
South America (Brazil, Argentina Rest of South America)
Key Players / Competitors Profiles Covered in Brief in Global Womens Health Diagnostics Market Report in Strategic Perspective:
- Quest Diagnostics Incorporated
- Hologic, Inc.
- PerkinElmer, Inc.
- Cardinal Health, Inc
- General Electric Company
- Becton, Dickinson and Company
- Abbott Laboratories
- Guided Therapeutics, Inc.
- F. Hoffmann-La Roche AG
- Siemens Healthineers AG
- FUJIFILM Holdings Corporation
- bioMérieux SA
- Bio-Rad Laboratories, Inc.
- Danaher Corporation
- Agilent Technologies, Inc.
- Thermo Fisher Scientific Inc.
- Koninklijke Philips N.V.
- Qiagen N.V.
- Sysmex Corporation
- Illumina, Inc.
- Myriad Genetics, Inc.
- Eurofins Scientific
- Natera, Inc.
- Hologlogic
- LabCorp (Laboratory Corporation of America Holdings)
