Liquid Biopsy Market by Product, Technology, Application, End User and Region – Global Market Size Estimation, Industry-Wide Analysis, Competitive Landscape Assessment & Long-Term Forecast to 2032
Overview
Liquid Biopsy Market size was valued at USD 3.73 Billion in 2025 and the total revenue is expected to grow at a CAGR of 20.9% from 2025 to 2032, reaching nearly USD 14.07 Billion.
Liquid Biopsy Market Overview:
Liquid biopsy is an easy, quick, non-invasive, and repeatable sampling technique that can reflect changes in tumor gene expression profiles and offer a solid foundation for customized treatment and cancer early detection. The focus in recent years has also been on early cancer detection, tracking the development of tumors, evaluating therapy response and clinical prognosis, and identifying recurrent and refractory cancers.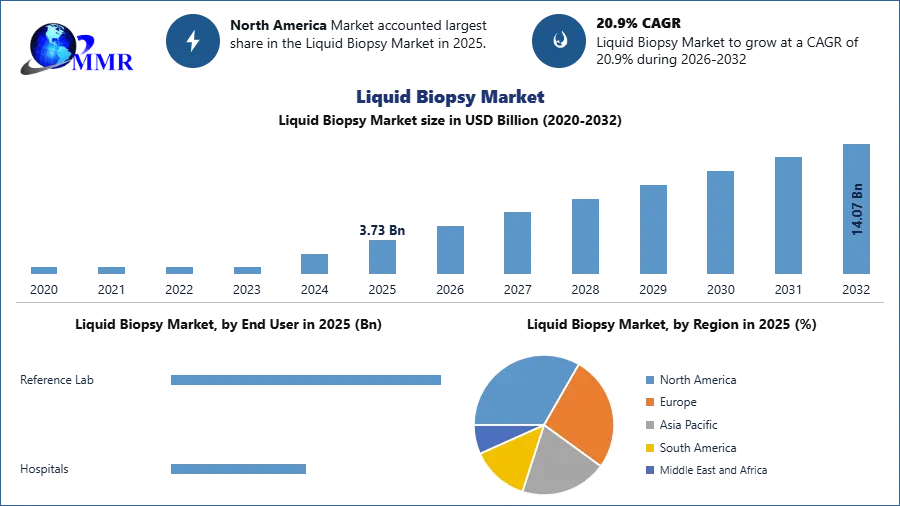 To know about the Research Methodology:-Request Free Sample Report
To know about the Research Methodology:-Request Free Sample Report
Furthermore, due to technological advancements in both practicality and turnaround time, liquid biopsy has recently attracted a lot of attention as a non-invasive substitute procedure for tissue biopsy in patients with cancer.
2025 is considered as a base year to forecast the market from 2026to 2032. 2025’s market size is estimated on real numbers and outputs of the key players and major players across the globe. Past five years' trends are considered while forecasting the market through 2032.
Liquid Biopsy Market Dynamics:
Liquid Biopsy Market Drivers:
Rising incidence and prevalence of cancer: The number of persons who have cancer has significantly increased on a world basis. Environmental factors, cigarette use, infectious diseases like Hepatitis B and C, as well as lifestyle changes, are some of the causes of the increased incidence of cancer. Cancer is the second-leading cause of mortality worldwide, according to the WHO. According to estimates from the International Agency for Research on Cancer (IARC), there would be 19.3 million new cases of cancer and 9.96 million cancer-related deaths worldwide in 2025.
Compared to conventional cancer diagnostic methods, liquid biopsy has various advantages, including lower cost, early prognosis, therapy monitoring, tumor heterogeneity identification, acquired drug resistance, and patient comfort (by eliminating the need for surgery). As a result, the liquid biopsy will probably become more popular.
Liquid Biopsy Market Restraints:
The lower sensitivity of certain liquid biopsies: The quantities of ctDNA of any particular cancer mutation may be very low in the plasma of a cancer patient, especially after treatment or surgery, making the detection of ctDNA in liquid biopsies technically tricky. According to sampling statistics, there may be less than one detectable copy of the cancer mutation-containing ctDNA in any one patient's plasma sample.
As a result, even though ctDNA is present in the plasma but at a low level, it might not be picked up in the patient sample. This affects the informative value of the liquid biopsy tests for cancer by producing false-negative results where ctDNA is present but not identified. False negatives can occasionally result in a tumor's recurrence not being discovered at an early stage.
Liquid Biopsy Market Opportunities:
The growing significance of companion diagnostics: Companion diagnostics are tests or assays used to help doctors decide what course of treatment to provide patients based on how well they respond to therapy. The rapid and cost-effective production of safer pharmaceuticals with improved therapeutic efficacy can fundamentally modify the drug development process and accelerate the commercialization of medication candidates. The market for companion diagnostics has significant growth potential due to the rise in demand for expensive specialized therapies and safer medications. The market for liquid biopsies is seeing prospects for growth as companion diagnostics gain prominence.
Liquid Biopsy Market Challenges:
Unclear reimbursement scenario: The procedure of paying for pricey molecular testing and the knowledge of the several specialists needed to interpret test results and create treatment programs are convoluted and uneven. This is particularly problematic with more expensive modern methods like liquid biopsy. The price of molecular testing services is frequently unknown to doctors, pathologists, and patients until they are paid or billed. Both patients and medical professionals find the confusion disturbing.
Additionally, payers' biomarker coverage policies have complex organizational structures that make them challenging to comprehend. While some only pay for the most fundamental tests or those related to certain clinical disorders and genes, others pay for a wide variety of testing. It is frequently more difficult to evaluate and keep track of different coverage policies due to this significant variety.
Liquid Biopsy Market Segment Analysis:
Based on Technology, In terms of revenue, the Multi-gene-parallel Analysis (NGS) category accounted for over 73 % of the global market in 2023, and it is expected that it will continue to dominate over the forecast period. NGS technology may identify a wide range of mutations that might contribute to carcinogenesis as well as identify potential resistance mechanisms that might have emerged from pre-existing clones following therapy.
Recent rapid advancements in NGS technology have resulted in a significant cost reduction for high-accuracy sequencing. This approach enables the screening of unidentified variations and is employed in focused panels for the precise and extremely sensitive detection of ctDNA mutations. For instance, stage 1 or stage 2 lung cancer patients can be identified by NGS with a MAF of roughly 0.1%.
Important operational companies have also released cutting-edge products, driving the market. For instance, Twist Bioscience Corp. introduced the Twist NGS Methylation Detection System in February 2021 for epigenetic research and liquid biopsy cancer analysis. Additionally, Biodesix, Inc. announced in April 2021 that it would expand its line of molecular testing by introducing a blood-based 52-gene NGS test.
Based on the Biomarker, The circulating Nucleic Acids Segment is dominating the market with a 36.67% share. The section is driven by the numerous uses of circulating tumor DNA (ctDNA) in liquid biopsies of cancer. Recent advancements in ctDNA-based liquid biopsy analysis have opened up new possibilities for cancer molecular diagnosis and surveillance. It can accurately predict tumour development, the prognosis, and help with targeted therapy.
Liquid biopsies are being used by translational cancer researchers to identify ctDNA from tumours. Future liquid biopsy applications for cancer detection have a significant chance to use the detection of ctDNA as a possible biomarker. ctDNA has been made available as an alternate source to invasive methods for molecular profiling of tumour DNA in cancer patients.
The fastest CAGR is expected for Exosomes/Microvesicles through the forecast period. Exosomes are being more widely used as liquid biopsies in oncology clinical diagnostics, and the arrival of novel products also contributes to the segment's growth.
At the American Urological Association's (AUA) 2022 Annual Meeting, miR Scientific presented findings for its miR Sentinel Prostate Cancer Test in September of that year. A single urine sample is used in this exosome-based liquid biopsy test, which has a 90% accuracy rate for detecting prostate cancer.
Liquid Biopsy Market Regional Insights:
The North America region is dominating the liquid biopsy market with a 51% share. Due to higher investments and the existence of numerous biotechnology businesses producing the tests, the U.S. dominates the regional market. Liquid biopsy is supported by several organizations, including the American Society of Clinical Oncology (ASCO). In consequence, it is expected that this will accelerate sales growth in this area.
Since tests that have received FDA approval may be utilized there, Canada has followed the United States in adopting liquid biopsy procedures. Additionally, the industry is highly competitive, which is expected to fuel sales growth throughout the forecast period. a rise in government funding and investments for the creation of liquid biopsy testing.
For instance, BioMark Diagnostic Solutions, Inc. received USD 825,000 in financing in June 2021 to develop the liquid biopsy assay for lung cancer screening. Consortium for Industrial Research and Innovation in Medical Technology grants and Spark funds from the Canadian Cancer Society, Brain Canada Foundation, and Canadian Institutes of Health Research - Institute of Cancer Research provide a sizable portion of the funding.
The objective of the report is to present a comprehensive analysis of the global Liquid Biopsy Market to the stakeholders in the industry. The past and current status of the industry with the forecasted market size and trends are presented in the report with the analysis of complicated data in simple language. The report covers all the aspects of the industry with a dedicated study of key players that include market leaders, followers, and new entrants.
PORTER, PESTEL analysis with the potential impact of micro-economic factors of the market have been presented in the report. External as well as internal factors that are supposed to affect the business positively or negatively have been analyzed, which will give a clear futuristic view of the industry to the decision-makers.
The reports also help in understanding the Liquid Biopsy Market dynamic, structure by analyzing the market segments and projecting the Liquid Biopsy Market size. Clear representation of competitive analysis of key players by Product type, price, financial position, product portfolio, growth strategies, and regional presence in the Liquid Biopsy Market make the report investor’s guide.
Liquid Biopsy Industry Ecosystem
Liquid Biopsy Market Recent Industry Developments (2025–2026)
| Date | Company | Development | Impact |
|---|---|---|---|
| 19 February 2026 | GRAIL | The company released topline results from the NHS-Galleri trial, involving over 142,000 participants, confirming the efficacy of its multi-cancer early detection (MCED) test. | The study demonstrated a substantial reduction in Stage IV cancer diagnoses and a four-fold higher cancer detection rate compared to standard screening. |
| 15 January 2026 | Illumina | The company unveiled its Billion Cell Atlas and strategic expansion into AI-driven liquid biopsy workflows during the J.P. Morgan Healthcare Conference. | This initiative accelerates minimal residual disease (MRD) testing and drug discovery by mapping biological pathways at an unprecedented scale. |
| 09 December 2025 | Guardant Health | The company launched FPG 360, an in-house liquid biopsy testing service at Policlinico Gemelli in Italy, utilizing Guardant360 CDx technology. | This partnership enables local access to comprehensive genomic profiling for Italian cancer patients, eliminating the need to ship samples abroad. |
| 05 December 2025 | Natera | The company completed the acquisition of Foresight Diagnostics for approximately $275 million to bolster its oncology portfolio. | The acquisition is designed to enhance Signatera’s sensitivity and expand the company's reach into the hematologic cancer detection market. |
| 20 November 2025 | Abbott | The company announced a definitive agreement to acquire Exact Sciences for an enterprise value of approximately $23 billion. | The deal positions the company at the forefront of the $120 billion cancer diagnostics market, integrating advanced liquid biopsy and MCED technologies. |
| 10 September 2025 | Exact Sciences | The company commercially launched Cancerguard, a first-of-its-kind multi-cancer early detection blood test. | Available as a laboratory-developed test for $689, it targets over 50 cancer types and expands non-invasive screening options for high-risk populations. |
Liquid Biopsy Market Scope: Inquire before buying
| Liquid Biopsy Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 3.73 USD Billion |
| Forecast Period 2026-2032 CAGR: | 20.9% | Market Size in 2032: | 14.07 USD Billion |
| Segments Covered: | by Product | Assay Kits Instruments |
|
| by Technology | Multi-gene-parallel Analysis (NGS) Single Gene Analysis (PCR Microarrays) |
||
| by Application | Cancer Non- Cancer |
||
| by End User | Reference Lab Hospitals |
||
Liquid Biopsy Market, by Region
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan and Rest of APAC)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe)
North America (United States, Canada and Mexico)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A)
South America (Brazil, Argentina Rest of South America)
Key Players
1. F. Hoffmann-La Roche Ltd. (Switzerland)
2. Myriad Genetics, Inc. (US)
3. QIAGEN N.V. (Netherlands)
4. Thermo Fisher Scientific, Inc. (US)
5. Guardant Health, Inc. (US)
6. MDxHealth SA (Belgium)
7. Exact Sciences Corporation (US)
8. Illumina Inc. (US)
9. Sysmex Inostics (US)
10. Bio-Rad Laboratories, Inc. (US)
11. Biocept, Inc. (US)
12. NeoGenomics, Inc. (US)
13. ANGLE plc. (UK)
14. Menarini-Silicon Biosystems (Italy)
15. Vortex Biosciences, Inc. (US)
16. Exosome Diagnostics, Inc. (US)
17. Agena Bioscience, Inc. (US)
18. MedGenome Inc. (US)
19. Epigenomics AG (Germany)
20. Personal Genome Diagnostics, Inc. (US).

