Clinical Trial Management System Market – Industry Structure Evaluation, Demand Drivers Analysis, Regional Growth Analysis and Identification, Competitive Positioning Review & Global Market Size Forecast to 2032
Overview
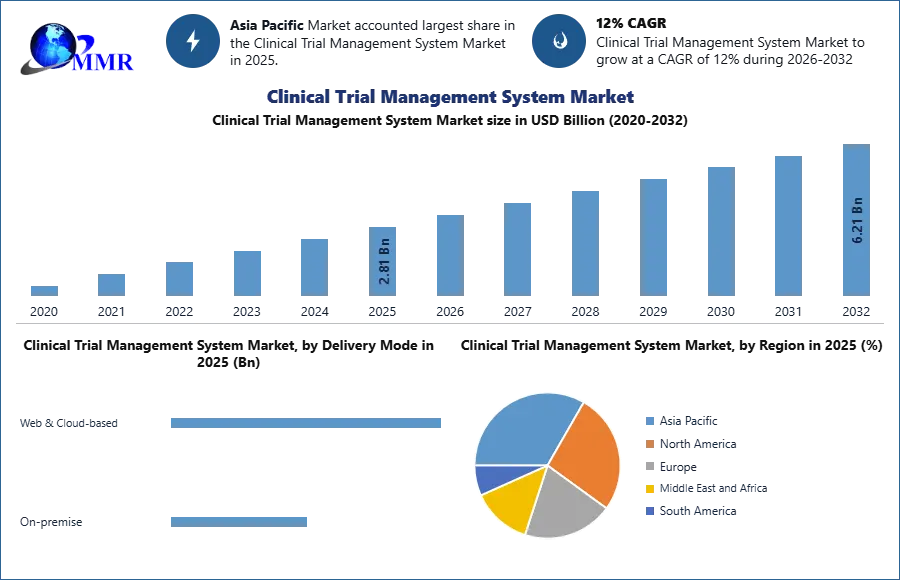
Clinical Trial Management System Market was valued at USD 2.81 Bn. in 2025. The Global Clinical Trial Management System Market size is estimated to grow at a CAGR of 12 % over the forecast period.
Clinical Trial Management System Market Overview:
The report explores the Clinical Trial Management System market’s segments (Type, Component, Delivery Mode, and End-User). Data has been provided by market participants, and regions (North America, Asia Pacific, Europe, Middle East & Africa, and South America). This market report provides a thorough analysis of the rapid advances that are currently taking place across all industry sectors. Facts and figures, illustrations, and presentations are used to provide key data analysis for the historical period from 2020 to 2025. The report investigates the Clinical Trial Management System market’s drivers, limitations, prospects, and barriers. This MMR report includes investor recommendations based on a thorough examination of the Clinical Trial Management System market’s contemporary competitive scenario.
To know about the Research Methodology :- Request Free Sample Report
Clinical Trial Management System Market Dynamics:
The significant growth in the number of clinical trials and the availability of advanced CTMS solutions:
The life science industry is annually witnessing a global increase in the number of clinical trials. The growth in the number of clinical trials can be attributed to factors such as the high prevalence of chronic diseases, the expiry of blockbuster drugs, the obtainability of government funds for clinical trials, and fierce competition in the pharmaceutical industry. In current years, leading players have launched many CTMS solutions in the market that boast superior performance to their traditional counterparts. These products are cost-effective, easy to use, provide effective patient safety and regulatory compliance, and enhance the financial management capabilities of users. They enable organizations to implement a full-featured CTMS without the complex configuration and heavy capital investment associated with traditional solutions.
Investment in actual digital infrastructure and agility in technological acceptance:
The need to fulfill stringent regulations has been vital to expediting the acceptance of new technology in research. Though the supply of technology has been increasing and the regulation of innovative methods is easing, pharmaceutical companies have been slow to use emerging technologies because of the doubt prevailing around this space and a highly fragmented supply market. Lately, pharma companies have augmented expenditure on AI and big data analytics, given their transformative supremacy over the R&D process and cost savings. Fear of competition in the market and the need for business transformation and agility are key forces driving huge investments in R&D technologies.
Hence, pharma companies must recognize outward innovation through supplier benchmarking in each of these categories and be involved in early engagement through co-development to stay competitive.
The availability of limited funding is limiting the growth of the market:
Small and mid-sized customer facilities often face financial restraints for clinical trials due to limited private funding, rigorous regulatory mandates, and financial outlook. This also includes ineffective site selection, poor study design & trial execution, safety issues, and dropouts due to practical or financial issues. Furthermore, the time and capital required to complete a trial increase at each phase. The total cost of a Phase III failure includes the cost of all previous phases, plus the time that could have been used to trial a different drug. Each failed trial contributes to the rising costs of biopharma R&D.
Although R&D spending in the life science industry is rising, start-ups and SMEs still face budget constraints, which affect their decision to purchase CTMS solutions. Due to uncertainties in product approvals, undue delays in product approvals, and long gestation periods, private investors usually prefer established firms to invest in rather than start-ups and SMEs
Clinical Trial Management System Market Trends:
Investment in actual digital infrastructure and agility in technological adoption is quickly growing. The need to fulfill stringent regulations has been crucial to expediting the adoption of new technology in research. Though the supply of technology has been increasing and the regulation of advanced methods is easing, pharmaceutical companies have been slow to use emerging technologies because of the uncertainty prevailing around this space and a highly fragmented supply market.
Recently, pharma companies have increased expenditure on AI and big data analytics, given their transformative supremacy over the R&D process and cost savings. Fear of rivalry in the market and the need for business transformation and agility are key forces driving vast investments in R&D technologies. Later, pharma companies must recognize outward innovation through supplier benchmarking in each of these categories and be involved in early engagement through co-development to stay competitive.
The scarcity of skilled professionals for handling sophisticated digital solutions in research teams is restraining the growth of the CTMS market. With severe time constraints and increasing cost cuts, CROs and pharmaceutical companies are reluctant to invest in training their research employees. Hence, a huge gap exists between the available and required skilled manpower in the clinical research industry, restricting the adoption and utilization of complex software solutions in clinical trials. The development of user-friendly software solutions could act as a key growth opportunity for software vendors in this market.
Clinical Trial Management System Market Segment Analysis:
By delivery mode, the clinical trial management system is segmented into the Web & Cloud-based, and On-premise. The Web & Cloud-based segment dominated the largest market share in 2025. As these are the most preferred CTM systems. They help minimize costs associated with system security, backups, upgrades, and uptime consistency. Furthermore, web & cloud-based systems support the storage of large amounts of data as well as centralization of data, which facilitates access to data from any location. These factors are expected to drive growth during the forecast period.
This segment is also anticipated to exhibit the fastest growth during the forecast period. This owing to the increased adoption of AI, analytics, and machine learning algorithms. The advanced automation thus contributes to the increased utility of web & cloud-based solutions. The technology comprises three services such as Platform as a Service (PaaS), Infrastructure as a Service (IaaS), and Software as a Service (SaaS). Cloud-based technology offers greater study control to CTMS, wherein the clinical trial managers can manage location and other research study requirements in real-time. Another advantage is access to data from any device such as mobile, workstations, laptops, and tablets through software.
By End-User, the clinical trial management system is segmented into Pharmaceutical and Biotechnology Firms, Medical Device Firms, and Others. As the CTMS plays a vital role in drug discovery by improving workflows, reducing costs, and speeding up the overall clinical trial process. The pharmaceutical and biotechnological firms segment inhabits the largest revenue share in the CTMS market. This growth can be attributed to the rising acceptance of CTMS end-users to store data, sync records, and manage the large number of clinical studies conducted every year.
The others segment is anticipated to grow the fastest. It comprises contract research organizations (CROs), academic research organizations, and others. The growth of this segment is attributable to the increased outsourcing of clinical trials as well as a rise in the number of decentralized clinical trials. Clarion for instance offers solutions for all trial models: site-based, decentralized, and hybrid clinical trials. It generates rich, diverse, and reliable evidence via its 30 facilities spread across 9 countries in North America, Europe, and the Asia Pacific.
Clinical Trial Management System Market Regional Insights:
Asia Pacific region emerged as the dominant regional market share accounting for 32.4% in 2025. This is because of the major factors driving the growth of the Asia Pacific market include increasing government funding to support clinical trials, the presence of less stringent regulatory guidelines as compared to developed nations, a large patient base, faster rate of patient recruitment for clinical trials than mature nations, low operating costs for conducting clinical trials, a shortage of trial volunteers in Europe and North America, and the growing number of pharmaceutical companies and CROs in the region.
North America region is expected to be the second dominant region in this market. The large share of the North American region can be attributed to the presence of key companies the and rising adoption of technology in R&D. Favorable regulatory policies and increasing investment by the pharmaceutical companies can also be attributed to this regional growth. In November 2022, a clinical endpoint technology leader— ERT merged with Bioclinica to form Clarion. This enabled the company to technology-based based therapeutic area solutions to meet clinical trial objectives.
Recent Industry Developments (2025–2026)
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 10 June 2025 | Medpace | Medpace partnered with Voximetry to integrate AI-powered dosimetry tools into its clinical trial management workflows. | This integration enhances safety assessments in radiopharmaceutical trials through patient-specific radiation dose calculations. |
| 15 April 2025 | Veeva Systems | Veeva announced the Veeva SiteVault CTMS, a specialized system for research sites that integrates with eISF and eConsent. | This enables sites to manage trials comprehensively within a single platform, improving operational efficiency for investigators. |
| 01 March 2025 | Jeeva Clinical Trials | The company launched a unified AI-driven CTMS through its new CRO Partnership Program. | The system is designed to accelerate trials in therapeutic areas such as oncology and dermatology via automation. |
The objective of the report is to present a comprehensive analysis of the global Clinical Trial Management System Market to the stakeholders in the industry. The past and current status of the industry with the forecasted market size and trends are presented in the report with the analysis of complicated data in simple language. The report covers all the aspects of the industry with a dedicated study of key players that include market leaders, followers, and new entrants.
PORTER, PESTEL analysis with the potential impact of micro-economic factors of the market have been presented in the report. External as well as internal factors that are supposed to affect the business positively or negatively have been analyzed, which will give a clear, futuristic view of the industry to the decision-makers.
The reports also help in understanding the Clinical Trial Management System Market dynamic and structure by analyzing the market segments and projecting the Clinical Trial Management System Market size. Clear representation of competitive analysis of key players by Design, price, financial position, product portfolio, growth strategies, and regional presence in the Clinical Trial Management System Market make the report investor’s guide.
Clinical Trial Management System Market Scope: Inquire before buying
| Clinical Trial Management System Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 2.81 USD Billion |
| Forecast Period 2026-2032 CAGR: | 12% | Market Size in 2032: | 6.21 USD Billion |
| Segments Covered: | by Type | Enterprise Site |
|
| by Component | Software Service |
||
| by Delivery Mode | Web & Cloud-based On-premise |
||
| by End User | Pharmaceutical and Biotechnology Firms Medical Device Firms Others |
||
Clinical Trial Management System Market by Region:
North America (United States, Canada and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Vietnam, Taiwan, Bangladesh, Pakistan and Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, Nigeria and Rest of ME&A)
South America (Brazil, Argentina Rest of South America)
Key Players / Competitors Profiles Covered in Brief in Global Clinical Trial Management System Market Report in Strategic Perspective:
- Oracle Corporation
- Dassault Systèmes (Medidata Solutions)
- Veeva Systems Inc.
- IQVIA Holdings Inc.
- Parexel International Corporation (Calyx)
- IBM Corporation (Merative)
- Advarra, Inc.
- ArisGlobal LLC
- Bio-Optronics
- Datatrak International, Inc. (Fountayn)
- MasterControl Inc.
- Clario
- SimpleTrials
- RealTime Software Solutions, LLC
- Ennov
- Anju Software Inc.
- Medrio Inc.
- Clinion
- Castor
- Cloudbyz
- Viedoc
- Crucial Data Solutions
- PharmaSeal International Ltd.
- Suvoda LLC
- MedNet Solutions, Inc.
