Antisense and RNAi Therapeutics Market Size – Industry Structure Evaluation, Demand Drivers Analysis, Regional Growth Analysis and Identification, Competitive Positioning Review & Global Market Size Forecast to 2032
Overview
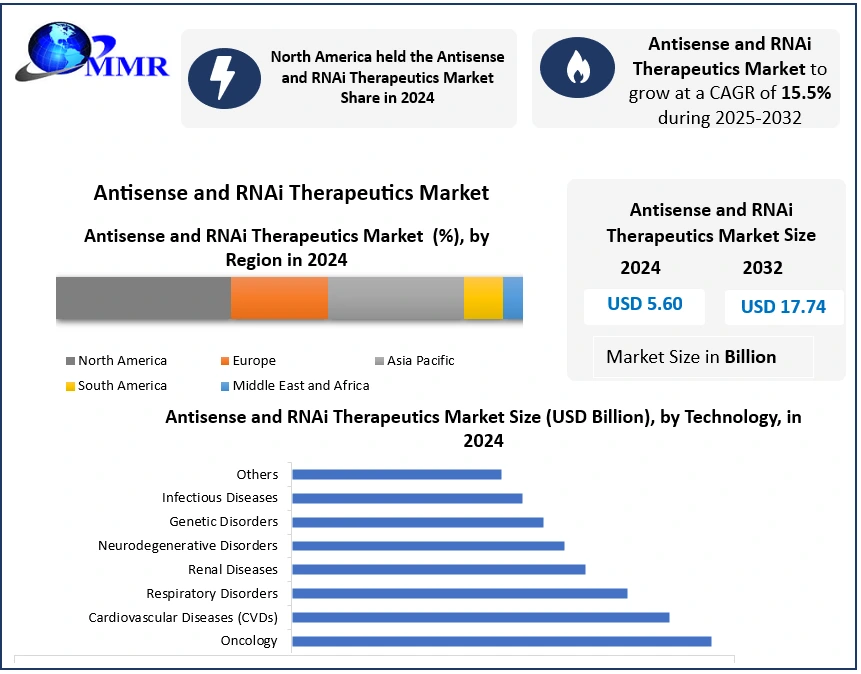
The Global Antisense and RNAi Therapeutics Market size was valued at USD 5.60 Bn in 2024, and the total Antisense and RNAi Therapeutics Market revenue is expected to grow by 15.5% from 2025 to 2032, reaching nearly USD 17.74 Bn.
Antisense and RNAi Therapeutics Market Overview
Antisense and RNA interference (RNAi) therapeutics are potential new gene-silencing technologies that target and regulate the expression of specific genes in disease processes. The licensure goal of these therapeutics is to inhibit the production of disease-causing protein targets, effectively interfering with a functional protein or protein target through targeting messenger RNA (mRNA) that causes disease targets, whether before or after transcription. Delivering therapeutic RNA (ASOs, siRNA, and miRNA) that has high specificity and durability. The increasing awareness of gene-silencing therapeutics is booming, especially within the Antisense and RNAi Therapeutics Market, as these technologies target rare genetic disorders, cancer, neurodegenerative diseases, and viral infections.
 To know about the Research Methodology:- Request Free Sample Report
To know about the Research Methodology:- Request Free Sample Report
The largest pharma and biotech companies are investing in next-generation delivery systems (lipid nanoparticles, GalNAc conjugates, or polymeric vectors) for antisense and RNAi drugs to increase the precision, safety, and bioavailability of therapeutics. The approval and widespread launch of dystrophy gene targeting drugs such as Spinraza (ASO), Onpattro (siRNA), and Givlaari (siRNA) have led to a significant increase in the total approved number of drugs for antisense gene silencing globally and have built a strong pipeline for growth and investor interest.
The precision advances in delivery technologies, historical prevalence rates of chronic and rare diseases, an increase in clinical trial activity, and collaborations between biotech firms and large pharmaceutical companies. Key players in this Antisense and RNAi Therapeutics Market include Ionis Pharmaceuticals, Alnylam Pharmaceuticals, Arrowhead Pharmaceuticals, Sarepta Therapeutics, and Silence Therapeutics, companies pioneering gene-silencing research and commercial applications. Technological innovation, regulatory support, and the trend toward personalized and precision medicine are transforming the Antisense and RNAi Therapeutics Market into a vital component of future drug development globally.
Antisense and RNAi Therapeutics Market Dynamics
Rapid advancement in drug delivery technologies to drive the Antisense and RNAi Therapeutics Market Growth
RNA molecules such as antisense oligonucleotides (ASOs) and small interfering RNAs (siRNAs) are inherently unstable and susceptible to enzymatic degradation, making effective delivery systems critical for their success. Innovations such as lipid nanoparticles (LNPs), ligand conjugation, and polymer-based carriers have significantly advanced the Antisense and RNAi Therapeutics Market by enabling these molecules to cross cell membranes, evade nuclease degradation, and selectively accumulate in disease-specific tissues with high precision. For example, LNP technology is also pivotal in the mRNA COVID-19 vaccines, has become central to delivering siRNA therapies for liver and rare genetic diseases. These delivery advancements reduce off-target effects and enhance in vivo half-life, thereby improving both clinical outcomes and regulatory approval prospects. The targeted delivery systems expand the therapeutic reach of RNA-based drugs beyond the liver to tissues such as muscle, brain, and tumors, unlocking new opportunities in neurology, oncology, and rare diseases.
High Cost and Complexity of Development to Restrain the Antisense and RNAi Therapeutics Market Growth
Developing these therapies involves expensive R&D cycles, intricate manufacturing processes, and long, high-risk clinical trials, with each stage requiring specialized expertise and stringent regulatory compliance. Delivery challenges, despite technological improvements, still pose risks, as ensuring consistent biodistribution, minimizing off-target effects, and achieving safe systemic delivery remain scientifically demanding. Even approved RNA-based drugs, such as patisiran and nusinersen, come with high price tags, exceeding USD 100,000 per patient annually, limiting patient access and placing strain on healthcare systems. These cost and complexity issues deter broader adoption, restrict availability to niche indications, and slow Antisense and RNAi Therapeutics Market penetration, ultimately tempering the otherwise strong growth trajectory.
Antisense and RNAi Therapeutics Market Segment Analysis
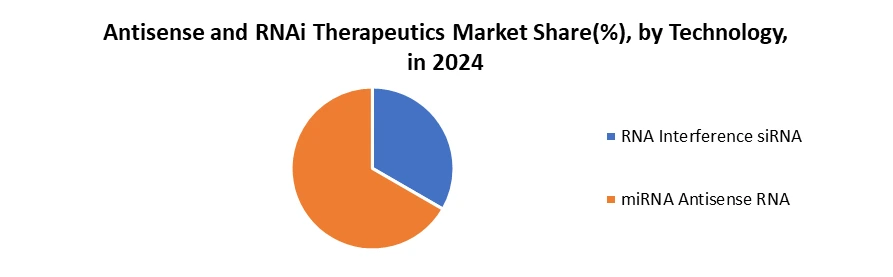
By Technology, Antisense RNA technology holds the dominant position in the Antisense and RNAi Therapeutics market, due to its maturity, proven clinical success, and broad applicability. Antisense oligonucleotides (ASOs) have been in development for decades, with several FDA- and EMA-approved drugs already on the Antisense and RNAi Therapeutics Market for conditions such as spinal muscular atrophy, Duchenne muscular dystrophy, and various cancers. Their mechanism of action, binding to complementary messenger RNA (mRNA) sequences to prevent protein translation or alter splicing, provides a high level of specificity while allowing flexible targeting across a wide range of diseases. RNA interference technologies such as siRNA and miRNA are relatively newer, with fewer approved therapies and a more limited track record. They have chemical modifications, such as phosphorothioate backbones and 2′-sugar modifications, that enhance stability and bioavailability, often removing the need for complex delivery vehicles. This reduces manufacturing complexity and can shorten development timelines in the Antisense and RNAi Therapeutics Market, accelerating the delivery of effective gene-silencing treatments. ASOs are designed and optimized quickly in response to emerging therapeutic targets, giving them a speed-to-market edge.
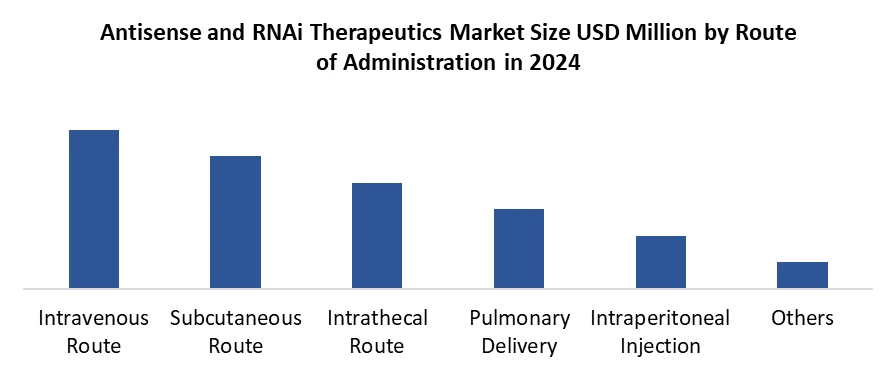
By Route of Administration, Intravenous administration remains the dominant route for antisense and RNAi therapeutics, capturing the largest Antisense and RNAi Therapeutics Market share due to its efficiency in delivering drugs systemically with predictable pharmacokinetics. IV delivery enables immediate entry of the therapeutic agent into the bloodstream, allowing for rapid distribution to target organs, particularly the liver, which is a major site for RNA-based drug action. This route is especially critical for therapies where high bioavailability and precise dosing control are essential, driving Antisense and RNAi Therapeutics Industry growth. While alternative delivery methods such as subcutaneous, intrathecal, and pulmonary routes are gaining attention, especially with innovations like GalNAc conjugates for subcutaneous delivery, these approaches are still emerging and generally used for niche indications or patient-specific needs. IV administration benefits from a well-established healthcare infrastructure, regulatory familiarity, and clinical protocols, which streamline both development and commercialization.
Antisense and RNAi Therapeutics Market Regional Analysis
North America dominated the Antisense and RNAi Therapeutics Market in 2024 and is expected to continue its dominance over the forecast period. The region benefits from an active pipeline, with numerous clinical trials demonstrating promising safety and efficacy results for both antisense oligonucleotides (ASOs) and RNA interference (RNAi) therapeutics. Many biotech firms based in the U.S. have allocated substantial resources toward advancing RNAi-based therapies, with several candidates now entering late-stage clinical development or nearing regulatory approval.
Government agencies, including the FDA and NIH, actively support innovation by streamlining clinical trial processes and providing funding initiatives aimed at accelerating the development of novel gene-silencing treatments. These efforts not only encourage new entrants but also foster collaborations between academia, biotech startups, and pharmaceutical giants. The anticipated commercialization of multiple antisense and RNAi therapies in the coming years is expected to significantly boost the Antisense and RNAi Therapeutics Industry growth in North America. The region’s advanced healthcare infrastructure, patient awareness, and reimbursement frameworks contribute to faster adoption once therapies are approved. Overall, North America’s robust ecosystem positions it as the global hub for innovation, research, and commercialization within the Antisense and RNAi Therapeutics Market.
Antisense and RNAi Therapeutics Market Competitive Landscape
The Antisense and RNAi Therapeutics Market is competitive and innovation-driven, growing on the heels of innovations in gene-silencing technologies, efficient delivery platforms, and extensive clinical applications due to the growing range of rare, genetic, and chronic diseases. With a strong pipeline of commercially approved drugs and strategic partnerships that leverage relative benefits, market leaders Ionis Pharmaceuticals (United States) and Alnylam Pharmaceuticals (United States) continue to dominate the market.
For example, Ionis had revenues of approximately USD 800 million in 2024, almost entirely through the continued success of its pipeline drug Spinraza, which Biogen has partnered with Ionis on, along with other opportunities with Roche. Alnylam's, leveraging its FDA-approved RNA therapies in Onpattro, Givlaari, and Amvuttra, with previous partnerships with Takeda and now Eli Lilly. Additionally, both Ionis and Alnylam are leveraging new delivery systems such as GalNAc conjugation and lipid nanoparticles to create a more precise possibility around targeting and safety. Emerging players such as Arrowhead Pharmaceuticals, Silence Therapeutics, and Dicerna Pharmaceuticals are animating this competitive landscape by pushing RNA-based drug development into next-gen modalities, which have established a ground for future partnerships with larger pharmaceutical companies.
Antisense and RNAi Therapeutics Market Developments
• In 2025, Alnylam presented Phase III HELIOS-B trial results showing that vutrisiran treatment led to meaningful improvements in cardiac function, including both systolic and diastolic performance. It also showed a 28% reduction in all-cause mortality and recurrent cardiovascular events overall, and a 33% reduction for patients on monotherapy.
• February 2025 – Ionis Pharmaceuticals announced positive Phase II data for its antisense candidate ION224, targeting metabolic liver disease. The therapy achieved significant reductions in liver fat content and key metabolic biomarkers, supporting advancement to Phase III trials.
• January 2025 – Sarepta Therapeutics reported FDA acceptance of its Biologics License Application (BLA) for SRP-5051, an antisense therapy for Duchenne muscular dystrophy amenable to exon 51 skipping. Priority review designation was granted, with a decision expected in late 2025.
• July 2024 – Silence Therapeutics launched a Phase I clinical trial for SLN124, an siRNA therapeutic for rare hematological disorders such as beta-thalassemia and myelodysplastic syndrome. Early clinical data indicated reductions in disease biomarkers and improved patient hematologic profiles.
• 2024 – Silence Therapeutics, headquartered in Berlin, signed a strategic collaboration agreement with AstraZeneca to jointly develop new siRNA therapeutics for cardiovascular and metabolic disorders. The deal includes over USD 2 billion in potential upfront and milestone payments, underscoring the growing strategic importance of RNAi-based approaches.
Antisense and RNAi Therapeutics Market Trends
| Trend | Description | Examples |
| Delivery Technology Advancements | New delivery systems improve targeted delivery to specific organs while minimizing off-target effects. | GalNAc conjugates and lipid nanoparticles (LNPs) enable targeted delivery to the liver and extra-hepatic tissues, enhancing efficacy and safety. |
| Strategic Partnerships and Licensing Deals | Collaborations with large pharmaceutical companies provide funding, co-development support, and faster commercialization pathways. | Examples include Ionis with Roche, and Alnylam with Regeneron. |
| Greater Focus on Chronic and Common Disease States | Expansion of therapeutic targets beyond rare diseases to prevalent conditions, creating larger market potential. | Common diseases targeted include hypertension, hypercholesterolemia, and Alzheimer’s disease. |
Antisense and RNAi Therapeutics Market Scope: Inquire before buying
| Global Antisense and RNAi Therapeutics Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2024 | Forecast Period: | 2025-2032 |
| Historical Data: | 2019 to 2024 | Market Size in 2024: | USD 5.60 Bn. |
| Forecast Period 2025 to 2032 CAGR: | 15.5% | Market Size in 2032: | USD 17.74 Bn. |
| Segments Covered: | by Technology | RNA Interference siRNA miRNA Antisense RNA |
|
| by Route of Administration | Intravenous Route Subcutaneous Route Intrathecal Route Pulmonary Delivery Intraperitoneal Injection Others |
||
| by Application | Oncology Cardiovascular Diseases (CVDs) Respiratory Disorders Renal Diseases Neurodegenerative Disorders Genetic Disorders Infectious Diseases Others |
||
Antisense and RNAi Therapeutics Market, by Region
North America (United States, Canada, Mexico)
Europe (United Kingdom, France, Germany, Italy, Spain, Sweden, Russia, the Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Philippines, Thailand, Vietnam, Rest of Asia Pacific)
Middle East and Africa (MEA) (South Africa, GCC, Nigeria, Rest of MEA)
South America (Brazil, Argentina, Colombia, Chile, Rest of South America)
Antisense and RNAi Therapeutics Market,Key Players are
North America
1. Alnylam Pharmaceuticals – United States
2. Ionis Pharmaceuticals – United States
3. Biogen Inc. – United States
4. Sarepta Therapeutics – United States
5. Arrowhead Pharmaceuticals – United States
6. Avidity Biosciences – United States
7. Dyne Therapeutics – United States
8. Stoke Therapeutics – United States
9. Regulus Therapeutics – United States
10. Arbutus Biopharma – Canada
Europe
1. Novartis AG – Switzerland
2. GSK plc (GlaxoSmithKline) – United Kingdom
3. Silence Therapeutics – United Kingdom/Germany
4. Roche – Switzerland
5. AstraZeneca – United Kingdom/Sweden
6. Sanofi – France
7. Bayer – Germany
8. Boehringer Ingelheim – Germany
9. Secarna Pharmaceuticals – Germany
10. ProQR Therapeutics – Netherlands
Asia-Pacific
1. Benitec Biopharma Ltd – Australia
2. OliX Pharmaceuticals – South Korea
3. Sirnaomics – Hong Kong/China
4. Suzhou Ribo Life Science – China
5. Shanghai GenePharma – China
6. Nippon Shinyaku – Japan
7. Takeda Pharmaceutical – Japan
8. Daiichi Sankyo – Japan
9. Wave Life Sciences – Singapore
10. Bioneer – South Korea
Frequently Asked Questions:
1. Which region has the largest share in the Antisense and RNAi Therapeutics Market?
Ans: The North America region held the highest Antisense and RNAi Therapeutics Market share in 2024.
2. What is the growth rate of the Antisense and RNAi Therapeutics Market?
Ans: The Global Antisense and RNAi Therapeutics Market is growing at a CAGR of 15.5 % during the forecasting period 2025-2032.
3. What is the scope of the market report?
Ans: The Global Antisense and RNAi Therapeutics Market report helps with the PESTEL, Porter's, Recommendations for Investors & Leaders, and market estimation of the forecast period.
4. What is the study period of this market?
Ans: The Global Antisense and RNAi Therapeutics Market is studied from 2024 to 2032.