Cancer Monoclonal Antibodies Market Size by Antibody Type, Application, End User, Region – Segment-Level Market Assessment, Growth Opportunity Analysis, Competitive Mapping & Forecast to 2032
Overview
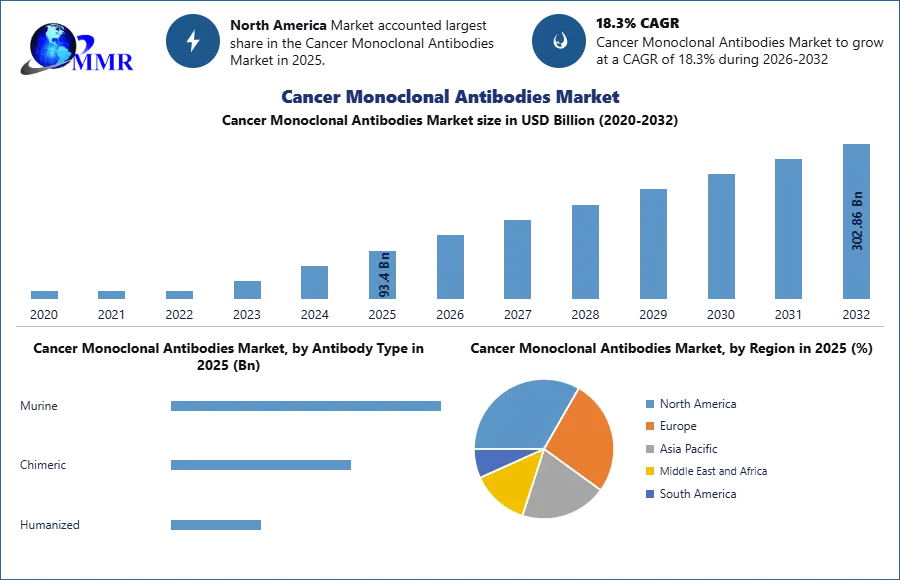
Global Cancer Monoclonal Antibodies Market size was valued at USD 93.4 Bn in 2025 and total Cancer Monoclonal Antibodies Market revenue is expected to reach USD 302.86 Bn by 2032, at a CAGR of 18.3% over the forecast period.
Cancer Monoclonal Antibodies Market Overview:
Monoclonal antibodies are laboratory-produced immune proteins engineered to specifically bind to tumour-specific antigens (e.g., HER2, PD-1, CD20). They combat cancer by stopping the development of tumours directly, flag off cancer cells for immunity destruction, or block immune posts. Monoclonal antibody-based immunotherapy is a major element of cancer therapy besides surgery, chemotherapy and radiation. Immunotherapy is running the global cancer monoclonal antibody market with increasing prevalence of cancer worldwide, progress in biotechnology and increasing demand for individual and targeted cancer remedies.
According to the American Society of Clinical Oncology, by the end of 2025, there will be a projected 2.1 million diagnosed cancer cases and 619,360 deaths in the U.S. giving rise to Global Cancer Monoclonal Antibodies Market. North America Cancer Monoclonal Antibody market accounted largest part in health care due to large patient population. The leading players in the Global Cancer Monoclonal Antibodies Market are Bristol-Myers Squib, Merc, Rosh, Novartis and Biocon. Hospitals and clinics dominate the primary end-users, about 80% of cancer monoclonal antibodies administer therapy and treat clinical adoption through treatment protocols.
To know about the Research Methodology :- Request Free Sample Report
Cancer Monoclonal Antibodies Market Dynamics:
Growing Incidence of Cancer to Boost the Cancer Monoclonal Antibodies Market
The incidence of cancer is increasing globally, which is driving the demand for cancer monoclonal antibodies. According to the World Health Organization (WHO), cancer is one of the leading causes of death worldwide, accounting for an estimated 9.6 million deaths in 2018. Rising healthcare expenditure as Governments and healthcare organizations are increasing their spending on cancer treatment and research, which is driving the growth of the cancer monoclonal antibodies market.
Advances in Biotechnology to Drive the Cancer Monoclonal Antibodies Market
Advanced biotechnology has enabled the development of more targeted and effective monoclonal antibodies for cancer treatment. This has increased the efficacy and safety of cancer monoclonal antibodies, which has led to their widespread adoption. Increasing demand for personalized medicine of Cancer monoclonal antibodies can be tailored to specific genetic mutations, making them a key Indication of personalized medicine. As the demand for personalized medicine increases, so does the demand for cancer monoclonal antibodies.
Growing Focus on Precision Medicine to Drive the Cancer Monoclonal Antibodies Market
Precision medicine involves tailoring treatments to individual patients based on their specific genetic and molecular characteristics. This approach is becoming increasingly important in cancer treatment and is driving the development of monoclonal antibodies that target specific molecular pathways in individual patients.
Development and Approval Timelines to Restrain the Cancer Monoclonal Antibodies Market
Developing and approving monoclonal antibodies can be time-consuming, which can delay their availability to patients. Additionally, the regulatory approval process can be complex and unpredictable, increasing the cost of development and delaying the time to market.
High Cost of Treatment to Restrain the Cancer Monoclonal Antibodies Market
Cancer monoclonal antibodies are expensive, which can limit their accessibility to patients, particularly in developing countries. The high cost of treatment can also put a strain on healthcare systems and limit reimbursement by insurance companies. Safety concerns for Monoclonal antibodies; can cause side effects, including infusion reactions, cytokine release syndrome, and immune-related adverse events. These side effects can limit the use of these drugs in some patients and may impact their market adoption.
Limited efficacy in Certain Cancer Types to Restrain the Cancer Monoclonal Antibodies Market
While monoclonal antibodies have shown efficacy in the treatment of certain cancer types, they may not be effective in all cancer types. This limitation may impact their market adoption and sales.
Combination therapies to Create Opportunities in the Cancer Monoclonal Antibodies Market
Combining monoclonal antibodies with other treatments, such as chemotherapy and radiation therapy, is a promising approach that could improve treatment outcomes and reduce side effects. This approach is being studied extensively in clinical trials and could lead to the development of new combination therapies in the future.
Emerging markets to Create Opportunities in the Cancer Monoclonal Antibodies Market
Emerging markets, particularly in Asia, offer significant growth opportunities for the cancer monoclonal antibodies market. These markets have large populations and increasing access to healthcare, which is expected to drive demand for cancer treatments.
Cancer Monoclonal Antibodies Market Segment Analysis:
Based on Antibody Type, Cancer Monoclonal Antibodies Market is segmented into Humanized, chimeric, Murine antibody. Humanized antibody segment dominated the monoclonal antibody market, accounting for 65% of all medical monoclonal antibodies in clinical use in 2025 due to their customized security and efficacy. Unlike chimeric (25%shares) or marine (<10%) antibodies, humanized monoclonal antibodies reduce immunogenecity (80%reduce anti -drug antibodies), while making them ideal for chronic remedies such as PD -1 inhibitors (of PD -1 inhibitors) while maintaining target specificity. Clinical data suggests that humanized monoclonal antibodies have a low side effect (eg, cytokine release syndrome).
Based on Application, the Cancer Monoclonal Antibodies Market is segmented into Blood cancer, Breast cancer, Lung cancer, etc. Blood cancer is the major segment in the monoclonal antibody market, inspired by better clinical results and comprehensive medical adoption. Monoclonal antibodies such as Ritximab (Anti-CD20) and darutumumab (anti-CD38) display reaction rates of more than 60% in hematologic deformities, while compared to 40–50% for solid tumours. These treatments have been integrated into 80% of the first-rich regimens for non-Hodgkin lymphoma and multiple myeloma, which reflects their important role in the treatment protocol.
Cancer Monoclonal Antibodies Market Regional Analysis:
North America is dominated in the Cancer Monoclonal Antibodies Market in 2025 and expected to dominate during forecast period
North America is currently the largest market for cancer monoclonal antibodies, driven by high healthcare spending and a large patient population. The region is home to several key market players, including Roche, Bristol-Myers Squibb, and Merck. Europe is also a significant market for cancer monoclonal antibodies, with countries such as Germany, France, and the UK driving growth. The Asia Pacific region is expected to experience the fastest growth in the coming years, driven by rising healthcare spending, increasing patient awareness, and expanding access to healthcare.
The region is home to several large pharmaceutical companies, including Takeda, Daiichi Sankyo, and Astellas Pharma, which are investing in the development of new cancer monoclonal antibodies. Latin America and the Middle East and Africa are also expected to contribute to the growth of the market, driven by increasing demand for cancer treatment and improving healthcare infrastructure. Overall, the cancer monoclonal antibodies market is expected to see continued growth across all regions, driven by ongoing research and development, increasing patient demand, and expanding access to healthcare.
Recent Industry Developments (2025–2026)
| Exact Date | Company | Development | Impact |
|---|---|---|---|
| 13 November 2025 | Sandoz | The FDA approved PERTUZUMAB-DPZB (POHERDY), marking the first-ever interchangeable biosimilar for pertuzumab (Perjeta) in treating HER2-positive breast cancer. | This development is expected to significantly lower treatment costs and increase patient access to critical HER2-targeted therapies globally. |
| 14 November 2025 | Merck & Co. Inc | Merck completed the $9.2 billion acquisition of Cidara Therapeutics, specifically targeting their Fc-conjugate platform for improved pharmacokinetics. | The deal strengthens Merck's oncology pipeline by integrating next-generation antibody engineering technologies designed for longer-lasting therapeutic effects. |
| 19 November 2025 | Johnson & Johnson | The FDA granted approval for DARATUMUMAB AND HYALURONIDASE-FIHJ (DARZALEX FASPRO) specifically for patients with high-risk smoldering multiple myeloma. | This expands the addressable market for anti-CD38 therapies into earlier stages of blood cancer, potentially delaying disease progression. |
| 06 January 2026 | Day One Biopharmaceuticals | Day One Biopharmaceuticals acquired a novel Antibody Drug Conjugate (ADC) candidate from Mersana Therapeutics in a deal valued at $285 million. | The acquisition highlights the growing trend of ADC specialization as a core growth driver within the broader monoclonal antibody market. |
| 26 January 2026 | Mirum Pharmaceuticals | Mirum Pharmaceuticals finalized an $820 million agreement with Bluejay Therapeutics to acquire a human monoclonal antibody (IgG isotype) platform. | This strategic move diversifies Mirum’s portfolio with biological therapies capable of highly specific immune system modulation. |
| 11 February 2026 | AstraZeneca | AstraZeneca received Swissmedic approval for the combination of Savolitinib and Tagrisso for MET-amplified non-small cell lung cancer (NSCLC). | The approval validates the clinical efficacy of combination antibody-inhibitor regimens in addressing resistant cancer mutations. |
Cancer Monoclonal Antibodies Market Key Trends
• Precision Strike (AI-Powered Antibody Optimization): AI-designed monoclonal antibodies now predict tumour escape mutations, boosting efficacy (e.g., BioNTech’s CAR-T/ monoclonal antibodies combos).
• Stealth Mode (Immunogenicity Elimination): Fully humanized scaffolds (e.g., Xencor’s X monoclonal antibodies tech) cut anti-drug antibodies by 80%.
• Double Agents (Bispecific Dominance): 60% of Phase III trials now test bispecifics (e.g., J&J’s Teclistamab vs. Regeneron’s Linvoseltamab).
Cancer Monoclonal Antibodies Market Scope: Inquire before buying
| Cancer Monoclonal Antibodies Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | 93.4 USD Billion |
| Forecast Period 2026-2032 CAGR: | 18.3% | Market Size in 2032: | 302.86 USD Billion |
| Segments Covered: | by Antibody Type | Murine Chimeric Humanized |
|
| by Application | Blood Cancer Breast Cancer Lung Cancer Others |
||
| by End User | Hospitals and Clinics Research & Academic Institutes/Laboratories Others |
||
| by Medication Type | Rituximab Trastuzumab Bevacizumab Cetuximab Pembrolizumab Nivolumab Ipilimumab Others |
||
Global Cancer Monoclonal Antibodies Market, by region
North America (United States, Canada and Mexico)
Europe (United Kingdom, France, Germany, Italy, Spain, Sweden, Russia, Rest of Europe)
Asia Pacific (China, Japan, South Korea, India, Australia, Malaysia, Thailand, Vietnam, Indonesia, Philippines, Rest of APAC)
Middle East and Africa (South Africa, GCC, Nigeria, Egypt, Turkey, Rest of MEA)
South America (Brazil, Argentina, Colombia, Chile, Peru, Rest of South America)
Key Players / Competitors Profiles Covered in Brief in Global Cancer Monoclonal Antibodies Market Report in Strategic Perspective:
- F. Hoffmann-La Roche Ltd
- Merck & Co. Inc
- Bristol Myers Squibb Company
- Johnson & Johnson
- Amgen Inc
- AstraZeneca plc
- Eli Lilly and Company
- Pfizer Inc
- Regeneron Pharmaceuticals Inc
- Sanofi S.A
- Novartis AG
- GlaxoSmithKline plc
- AbbVie Inc
- Daiichi Sankyo Company
- Sandoz International GmbH
- Celltrion Inc
- Genmab A/S
- Innovent Biologics Inc
- Samsung Biologics
- Biocad
- Boehringer Ingelheim
- Seagen Inc
- MacroGenics Inc
- Immunomedics
- Shanghai Henlius Biotech
