Breast Cancer Liquid Biopsy Market Size by Product Type, Circulating Biomarker, Application, End-User, Region, Industry-Wide Analysis, Competitive Landscape Assessment & Long-Term Forecast to 2032
Overview
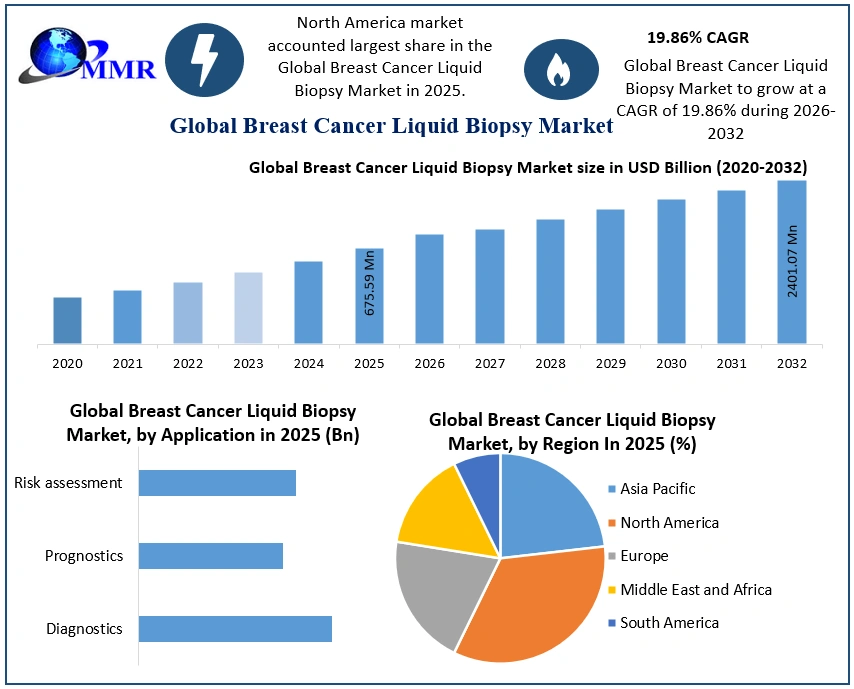
The Breast Cancer Liquid Biopsy Market size was valued at USD 675.59 Million in 2025 and the total Breast Cancer Liquid Biopsy revenue is expected to grow at a CAGR of 19.86% from 2026 to 2032, reaching nearly USD 2401.07 Million by 2032.
Breast Cancer Liquid Biopsy Market Overview
Liquid biopsy is a non-invasive diagnostic technique used for the diagnosis of breast cancer which involves the analysis of various biomarkers, such as circulating tumor cells (CTCs), cell-free DNA (cfDNA), and other components present in the bloodstream. This differs from the conventional biopsy which involves extraction of tissue specimens via invasive procedures, liquid biopsy presents an alternative less intrusive approach for detecting and monitoring breast cancer. In breast cancer, the utility of liquid biopsy is that it gives insights regarding genetic mutations and alteration in addition to molecular profile on circulating tumor cells present in the blood.
However, this approach is especially effective in evaluating the heterogeneity of tumors and defining therapeutic targets as well as treatment evaluation and minimal residual disease detection. Liquid biopsy is set to revolutionize the breast cancer diagnostics field by providing a more dynamic and real-time view of disease, leading to personalized interventions based on temporally targeted intervention.
In the case of breast cancer, liquid biopsies are rapidly becoming a widely used diagnostic practice, as they offer several advantages. The main strengths represents their non-invasive nature, which enables extracting important data without performing invasive tissue biopsies. The main components of liquid biopsies are ctDNA and CTCs, which are provided in the bloodstream. In the case of breast cancer, these biomarkers contain molecular information unique to each tumor manifestation and allow studying mutations and other alterations that play a significant role in carcinogenesis. The capacity to identify these genetic alterations brings invaluable information on tumor heterogeneity and more profound knowledge of the disease itself.
The usefulness of liquid biopsies lies in their ability to provide a rapidly accurate assessment of not only the state but also how well a particular therapy is working. This real-time monitoring helps in modifying treatment approaches, maximizing optimal therapeutic intervention, and minimizing major adverse effects, thus propelling the breast cancer liquid biopsy market revenue. In addition, the same tests are used in identifying MRD after surgical procedures or other treatments that are more reliable and accurate factors to consider the risk of disease relapse.
To know about the Research Methodology:- Request Free Sample Report
Breast Cancer Liquid Biopsy Market Dynamics
Overcoming Limitations of Traditional Biopsy Techniques
Traditional tissue biopsies involve the surgical removal of a portion of the tumor or affected tissue for analysis. While these biopsies have been fundamental in cancer diagnosis, they pose certain challenges, including invasiveness, potential complications, and the inability to capture the full heterogeneity of the tumor. Liquid biopsy addresses these limitations by offering a minimally invasive and repeatable approach to obtaining molecular information about breast cancer. Liquid biopsy's non-invasive nature significantly reduces the physical and psychological burden on patients, making it a more attractive option, especially for those who are not suitable candidates for surgical procedures.
This is particularly relevant in breast cancer, where routine screening and monitoring are crucial due to the high prevalence of the disease and the potential for recurrence. The simplicity of drawing a blood sample makes liquid biopsy a convenient and accessible tool for monitoring patients throughout their cancer journey, facilitating regular assessments without the need for repeated invasive procedures.
Additionally, liquid biopsy provides a more comprehensive view of tumor heterogeneity. Breast cancer is known for its molecular and histological diversity, with different regions of a tumor exhibiting distinct genetic characteristics. Traditional biopsies cannot capture this heterogeneity adequately, potentially leading to incomplete information for treatment decision-making. Liquid biopsy offers a complete tumor's genetic landscape with an enhanced ability to capture tumor heterogeneity and understand the evolution of the disease over time.
Early Detection and Personalized Treatment Approaches
Early detection of breast cancer is crucial for improving patient outcomes, as interventions at an early stage often lead to more effective treatments and higher survival rates. Liquid biopsy, particularly the analysis of circulating tumor DNA (ctDNA) and circulating tumor cells (CTCs), provides a non-invasive means to identify genetic alterations and biomarkers associated with breast cancer. By detecting breast cancer-related mutations in blood samples, liquid biopsy allows for the identification of tumors at an earlier stage than traditional imaging or clinical examinations. This has significant implications for the timely initiation of treatment strategies, potentially increasing the success of therapeutic interventions and propelling the breast cancer liquid biopsy market growth.
Moreover, the genomic information obtained through liquid biopsy contributes to personalized treatment options. Each breast cancer patient has a unique genetic makeup, and understanding the specific genetic mutations guides clinicians in selecting targeted therapies. Liquid biopsy enables the continuous monitoring of genomic alterations over time, allowing for real-time adjustments to treatment plans based on the evolving genetic profile of the tumor. This dynamic approach to cancer management contrasts with traditional methods, offering a more precise and tailored therapeutic strategy. As the field of oncology increasingly emphasizes personalized medicine, the ability of liquid biopsy to detect genetic variations and inform targeted treatments positions and offers key players with breast cancer liquid biopsy market opportunity.
Breast Cancer Liquid Biopsy Market Segment Analysis:
Based on Application, the circulating cell-free DNA (cfDNA) segment accounted for the largest Breast Cancer Liquid Biopsy Market share and is expected to remain dominant during the forecast period. Circulating cell-free DNA (cfDNA) offers a non-invasive and easily accessible source of genetic material for detecting cancer-related mutations. Additionally, cfDNA is released into the bloodstream as a result of cell death or apoptosis, providing a real-time and dynamic reflection of the tumor's genetic profile. Further, the circulating nature of cfDNA allows for the continuous monitoring of cancer progression and treatment response, offering a longitudinal perspective on the disease. This is crucial for clinicians to make informed decisions regarding personalized treatment plans and adjustments.
The versatility of cfDNA in capturing genetic alterations, such as mutations and variations, contributes to its efficacy in identifying specific biomarkers indicative of breast cancer. Moreover, ongoing advancements in molecular technologies and next-generation sequencing have significantly improved the sensitivity and specificity of cfDNA analysis, enabling the detection of low-frequency mutations with high accuracy. These technological advancements have driven the clinical utility of cfDNA in early cancer detection, monitoring treatment response, and identifying potential resistance mechanisms.
Based on End User, the hospitals and physician laboratories segment dominated the Breast Cancer Liquid Biopsy Market in terms of revenue, as they serve as primary points of care for patients undergoing diagnosis, treatment, and monitoring for breast cancer. These facilities often have well-established infrastructure, including skilled medical personnel, diagnostic equipment, and laboratory facilities, making them readily accessible for patients seeking cancer-related services. Moreover, hospitals and physician laboratories are equipped to handle a wide range of medical procedures, including liquid biopsy testing, within the same facility where patients receive other aspects of their care. This integration streamlines the diagnostic process, enabling efficient coordination between different stages of patient management, from initial screening to treatment planning and follow-up assessments. Additionally, the presence of multidisciplinary teams comprising oncologists, pathologists, radiologists, and other specialists in these settings facilitates comprehensive patient care and decision-making based on liquid biopsy results.
Further, hospitals and physician laboratories often have established relationships with healthcare providers and referral networks, which contribute to a variety of patient samples for liquid biopsy testing. This consistent volume of specimens enhances the economies of scale and operational efficiency of testing procedures, thereby driving down overall costs and making liquid biopsy services more financially available within these settings.
Breast Cancer Liquid Biopsy Market Regional Insights:
North America dominated the market in 2025 and is expected to hold the largest Breast Cancer Liquid Biopsy Market share over the forecast period. The region has a well-established healthcare infrastructure, advanced diagnostic technologies, and a robust research and development ecosystem. Major economies like the United States and Canada have been at the forefront of adopting innovative medical technologies, including liquid biopsy for breast cancer detection. Additionally, the high prevalence of breast cancer in North America has driven the demand for more accurate and non-invasive diagnostic methods, such as liquid biopsy, which offers the potential for early detection and monitoring of the disease.
For instance, according to the Breast Cancer Research Foundation, an estimated 297,790 women were diagnosed with breast cancer in the U.S., making it the most common cancer in American women. Further, collaborations between research institutions, biotechnology companies, and healthcare providers in North America have facilitated advancements in liquid biopsy technologies, contributing to the region's leadership in this market. Regulatory support for precision medicine and diagnostics has also played a crucial role in fostering the growth of the Breast Cancer Liquid Biopsy Market in North America.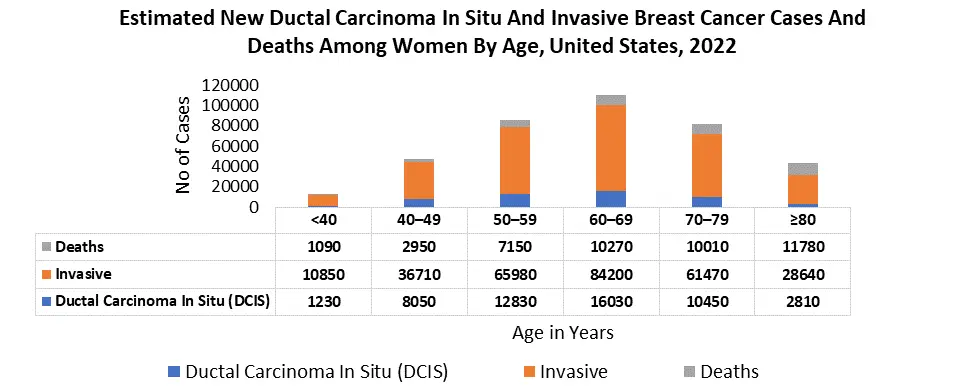
Breast Cancer Liquid Biopsy Market Scope: Inquire before buying
| Global Breast Cancer Liquid Biopsy Market | |||
|---|---|---|---|
| Report Coverage | Details | ||
| Base Year: | 2025 | Forecast Period: | 2026-2032 |
| Historical Data: | 2020 to 2025 | Market Size in 2025: | USD 675.59 Mn. |
| Forecast Period 2026 to 2032 CAGR: | 19.86% | Market Size in 2032: | USD 2401.07 Mn. |
| Segments Covered: | By Product Type | Kits and Consumables Instruments |
|
| By Circulating Biomarker | Circulating Tumor Cells (CTCs) Circulating Cell-free DNA (cfDNA) Extracellular Vesicles (EVs) Others Circulating Biomarker |
||
| By Application | Diagnostics Prognostics Risk assessment |
||
| By End-User | Reference Laboratories Hospitals and Physician Laboratories Others |
||
Breast Cancer Liquid Biopsy Market, by Region
North America (United States, Canada, and Mexico)
Europe (UK, France, Germany, Italy, Spain, Sweden, Austria and Rest of Europe)
Asia Pacific (China, South Korea, Japan, India, Australia, Indonesia, Malaysia, Taiwan, and Rest of APAC)
Middle East and Africa (South Africa, GCC, Egypt, and Rest of ME&A)
South America (Brazil, Argentina, and Rest of South America)
Breast Cancer Liquid Biopsy Market, Key Players:
1. F. Hoffmann-La Roche Ltd (Switzerland)
2. QIAGEN (Germany)
3. Biocept Inc (United States)
4. Menarini Group (Italy)
5. Guardant Health AMEA (United States)
6. Fluxion Biosciences Inc (United States)
7. Thermo Fisher Scientific Inc. (United States)
8. Neogenomics Laboratories, Inc. (United States)
9. Myriad Genetics, Inc. (United States)
10. ANGLE plc (United Kingdom)
11. Epic Sciences (United States)
12. Illumina, Inc. (United States)
Frequently Asked Questions:
1. What is Liquid biopsy used for breast cancer?
Ans: Liquid biopsy for breast cancer involves the analysis of circulating biomarkers, such as circulating tumor DNA (ctDNA), circulating tumor cells (CTCs), and microRNAs, in a patient's blood.
2. Which regions show dynamic growth in the breast cancer liquid biopsy market?
Ans: The breast cancer liquid biopsy market experiences dynamic growth in North America, Europe, Asia-Pacific, and Latin America. Notably, North America, especially the United States, has been a significant contributor to market revenue.
3. Why is liquid biopsy significant in breast cancer diagnosis?
Ans: Liquid biopsy is significant in breast cancer diagnosis due to its non-invasive nature, enabling the detection of circulating tumor DNA or cells in the blood, offering an accessible and less discomforting alternative to traditional tissue biopsies. It allows for early detection, real-time monitoring of treatment response, and identification of genetic alterations, providing valuable insights for personalized treatment strategies in breast cancer patients.
4. Why is North America a key player in the breast cancer liquid biopsy market?
Ans: North America is a key player in the breast cancer liquid biopsy market due to its robust healthcare infrastructure, advanced research facilities, and high adoption of innovative medical technologies. The region's well-established regulatory framework and significant investments in cancer research contribute to its leadership in developing and commercializing liquid biopsy technologies for breast cancer detection and monitoring.
5. Which end-user segment has the largest market share in the breast cancer liquid biopsy market?
Ans: In 2025, the Hospital & Physician Laboratories segment held the largest market share, due to widespread clinical adoption, diagnostic convenience, and integrated healthcare systems, fostering efficient implementation of this non-invasive technology in patient care and breast cancer diagnostics.